First Law of Thermodynamics and Enthalpy Changes
Energy can't be created or destroyed during chemical reactions; it can only be transferred or converted from one form to another. That's the core idea behind the first law of thermodynamics, and it's the foundation for understanding why reactions release or absorb heat. This section covers how energy moves between systems and surroundings, how enthalpy tracks those energy changes, and how to calculate them using Hess's Law, standard enthalpies of formation, and bond enthalpies.

🌀 First Law of Thermodynamics and Energy Exchange
System and Surroundings
In thermochemistry, you divide the universe into two parts:
- The system: The part you're studying, usually a chemical reaction or physical process.
- The surroundings: Everything outside the system.
A beaker where a reaction occurs is the system. The lab bench, the air, your hands holding the beaker: those are all surroundings. Energy flows between these two parts, but the total energy of system + surroundings stays constant. That's the first law.
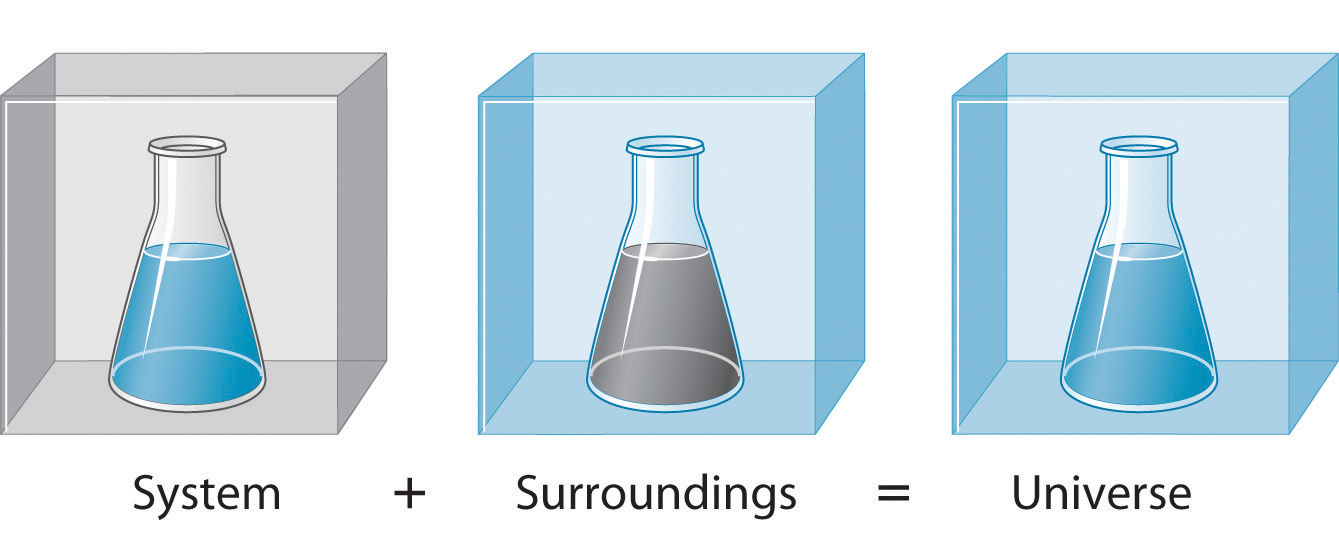
Image showing the relationship between system and surroundings.
Image Courtesy to Chemistry LibreTexts
Heat (q) and Work (w)
Energy moves between the system and surroundings in two ways:
- Heat (q): Energy transferred because of a temperature difference between the system and surroundings.
- Work (w): In chemistry, this usually means pressure-volume work, where gases expand against external pressure or get compressed.
The first law is expressed mathematically as:
where is the change in internal energy of the system. The sign conventions matter here: means heat flows into the system, and means work is done on the system.
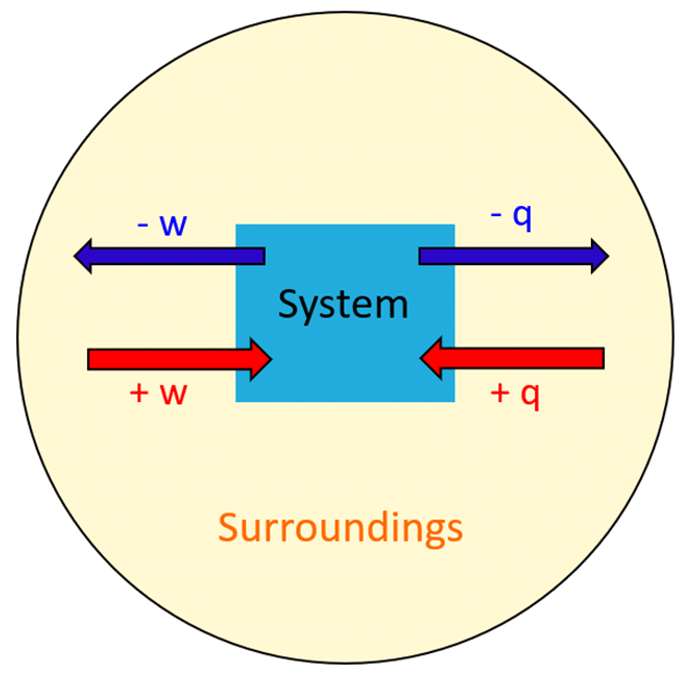
Image displaying the relationship between heat, work, system, and surroundings
Image Courtesy to Chemistry Steps
🔥 Enthalpy Changes in Reactions
Defining Enthalpy (H)
Enthalpy is defined by:
where is internal energy, is pressure, and is volume. You can't measure directly, but you can measure the change in enthalpy (). At constant pressure (which is how most lab reactions run), equals the heat exchanged:
This is why enthalpy is so useful in chemistry: at constant pressure, the enthalpy change is the heat of the reaction.
Types of Reactions
Different reaction types have characteristic values:
- Combustion: A substance reacts with , usually producing and . These are highly exothermic (). For example, burning methane releases about .
- Formation: A compound forms from its elements in their standard states. can be positive (endothermic) or negative (exothermic) depending on the compound.
- Dissolution: A solute dissolves in a solvent. Dissolving ammonium nitrate in water is endothermic (the solution feels cold), while dissolving sodium hydroxide is exothermic (it heats up).
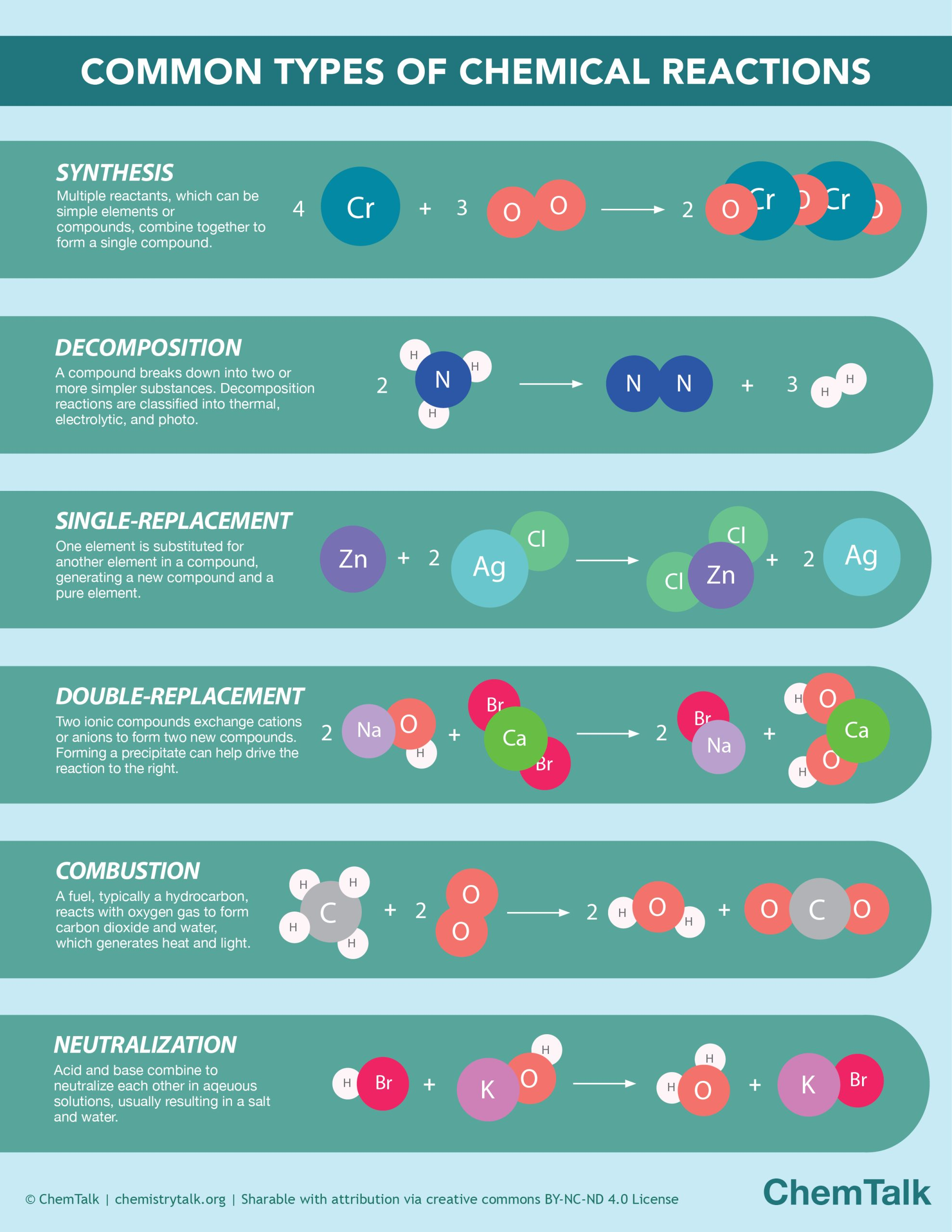
Image displaying the different types of chemical reactions that can occur.
Image Courtesy to ChemTalk
There are three main methods for calculating enthalpy changes:
- Hess's Law: Combine known reaction steps to find for an overall reaction.
- Standard enthalpies of formation (): Use tabulated values for individual compounds.
- Bond enthalpies: Estimate by accounting for energy needed to break and form bonds.
📊 Calculating Energy Changes
Hess's Law
Hess's Law states that the total enthalpy change for a reaction depends only on the initial and final states, not on the pathway taken. This means you can add up values from multiple steps to find the overall .
How to apply Hess's Law:
- Write the target reaction you need for.
- Identify given reactions with known values that contain the same species.
- Reverse or multiply the given reactions as needed so that when you add them together, intermediates cancel and you're left with the target reaction.
- If you reverse a reaction, flip the sign of its . If you multiply a reaction by a coefficient, multiply its by the same factor.
- Sum all the adjusted values.
Standard Enthalpies of Formation
The standard enthalpy of formation () is the enthalpy change when one mole of a compound forms from its elements in their standard states (25°C, 1 atm). By definition, for any element in its standard state is zero.
To calculate :
- Look up values for every product and reactant from a reference table.
- Apply the equation:
where is the stoichiometric coefficient for each species.
✏️ Practice Problem
Calculate for the formation of water vapor:
Given values:
- (element in standard state)
- (element in standard state)
Applying the formula:
The negative sign tells you this reaction is exothermic: it releases 483.6 kJ of heat per mole of reaction as written.
🔗 Bond Enthalpies
Bond enthalpy is the energy required to break one mole of a particular bond in the gas phase. You can estimate for a reaction by comparing the energy cost of breaking bonds in reactants to the energy released when forming bonds in products.
Steps:
- Draw out the structural formulas so you can identify every bond broken and every bond formed.
- Sum the bond enthalpies for all bonds broken (this is energy absorbed, so it's positive).
- Sum the bond enthalpies for all bonds formed (this is energy released, so it's negative).
- Calculate:
Bond enthalpy calculations give estimates because tabulated values are averages across many different molecules. They work best for gas-phase reactions.
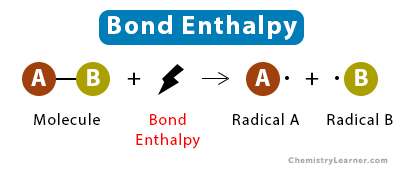
Image showing how bond enthalpy is used in reactions.
Image Courtesy to Chemistry Learner
🌡️ Calorimetry and Measuring Heat Transfer
Calorimetry is the experimental technique for measuring heat exchanged during a reaction or physical change. The key equation is:
where:
- = mass of the substance being heated or cooled (in grams)
- = specific heat capacity (in J/g°C)
- = change in temperature ()
Using a calorimeter:
- Set up the calorimeter and ensure it's insulated so minimal heat escapes to the surroundings.
- Record the initial temperature of the solution or water inside.
- Add the reactants or substance, then stir and monitor the temperature until it reaches a maximum (or minimum).
- Calculate and plug into .
A positive means the solution absorbed heat (endothermic process from the system's perspective), while a negative means the solution released heat. Keep in mind that the heat gained by the water equals the heat lost by the reaction, so .
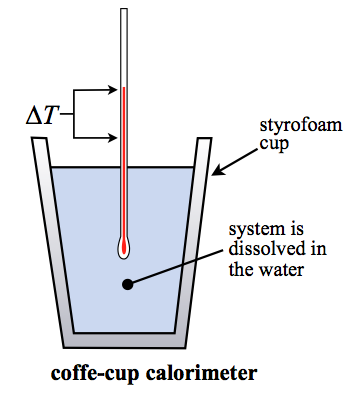
Image displaying how a calorimeter is set up.
Image Courtesy to Ch301
✏️ Practice Problem
You have 100 g of water and want to heat it from 20°C to 50°C. The specific heat capacity of water is 4.18 J/g°C. How much heat is needed?
So 12,540 J (or 12.54 kJ) of heat are needed to raise 100 g of water by 30°C.
✨ Thermochemistry in Real Life
Thermochemical principles show up everywhere in energy production and industry. Burning fossil fuels is a combustion reaction that releases stored chemical energy as heat, which can be converted to electricity or mechanical work. The tradeoff is the production of and other greenhouse gases.
In industrial and engineering contexts, enthalpy data helps with:
- Reactor design: Knowing whether a reaction is exothermic or endothermic determines whether you need to add heat or remove it to keep the process safe and controlled.
- Cost analysis: The energy input or output of a reaction directly affects production costs, so accurate values matter for economic decisions.
- Optimizing conditions: Understanding how enthalpy changes with temperature and pressure helps engineers choose conditions that maximize yield and efficiency.