Applications of Electrochemistry
Electrochemistry connects the redox reactions you've been studying to real-world technology. Batteries, fuel cells, corrosion prevention, and electroplating all rely on the same principles of electron transfer, half-reactions, and cell potentials you've already learned. This section covers how those principles show up in practice.

🔋 Batteries: Powering Our Daily Lives
Primary and Secondary Batteries
A primary battery is designed for one-time use and cannot be recharged. Once the reactants are consumed, the cell is dead. Alkaline batteries (like the ones in a TV remote) are a common example. The zinc anode is gradually oxidized, and once it's used up, the reaction can't be reversed.
A secondary battery can be recharged by running current through it in the reverse direction, restoring the original reactants. Your phone's lithium-ion battery is a secondary battery. During charging, an external power source forces electrons back, reversing the spontaneous redox reaction.
🤔 Think About It: Why recycle batteries instead of throwing them away? Many batteries contain heavy metals (like cadmium, lead, or mercury) that can leach into soil and groundwater. Recycling also recovers valuable materials like lithium and cobalt, which are expensive and energy-intensive to mine.
Battery Chemistry
Every battery converts chemical energy into electrical energy through spontaneous redox reactions. At the anode, oxidation occurs (electrons are released). At the cathode, reduction occurs (electrons are accepted). Electrons flow from anode to cathode through an external circuit, and that flow of electrons is the electric current you use.
The electrolyte between the electrodes allows ions to migrate, completing the internal circuit. A separator keeps the electrodes from touching (which would short-circuit the cell).
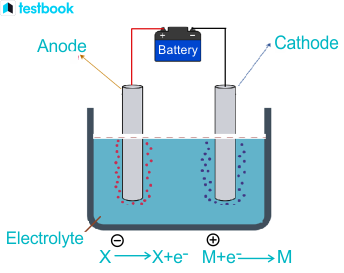
Image showing electron flow in a battery from anode to cathode with labels for each part of the cell including electrolyte and separator.
Image courtesy of Testbook.
Applications of Batteries
- Consumer Electronics: Mobile phones, laptops, and cameras all depend on portable battery power.
- Electric Vehicles: Large lithium-ion battery packs power electric cars, reducing dependence on fossil fuels. A typical EV battery pack operates at around 400 V and stores 60–100 kWh of energy.
- Grid Energy Storage: Large-scale battery systems store energy from renewable sources like solar and wind, releasing it when demand is high or generation is low.
⛽ Fuel Cells: Clean Energy Conversion
Working Principle of Fuel Cells
A fuel cell converts chemical energy directly into electrical energy without combustion. In a hydrogen fuel cell, the overall reaction is:
Here's how it works step by step:
-
Hydrogen gas () enters the anode side and is oxidized:
-
Electrons travel through an external circuit to the cathode, generating electric current.
-
Protons () migrate through a membrane to the cathode.
-
At the cathode, oxygen reacts with the protons and electrons:
-
The only byproduct is water.
Because there's no combustion, fuel cells can achieve higher efficiencies than internal combustion engines (up to ~60% vs. ~25–30%).
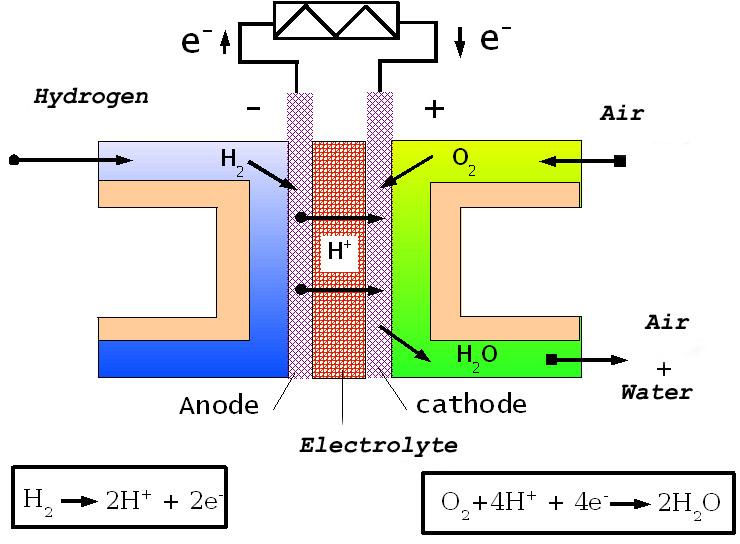
Image showing the components of a fuel cell including the flow of hydrogen and oxygen with arrows indicating electron flow and water as output.
Image courtesy of Wikimedia Commons.
Types of Fuel Cells
- Proton Exchange Membrane Fuel Cells (PEMFCs) operate at relatively low temperatures (~80°C), start up quickly, and are commonly used in vehicles. They use a platinum catalyst to speed up the electrode reactions.
- Solid Oxide Fuel Cells (SOFCs) operate at very high temperatures (~800–1000°C) and are better suited for stationary power generation. Their high operating temperature allows waste heat to be recovered and used, boosting overall efficiency.
Applications of Fuel Cells
- Transportation: Hydrogen fuel cell buses, trains, and cars produce zero tailpipe emissions (only water vapor).
- Backup Power: Hospitals and data centers use fuel cells where reliable, uninterrupted power is critical.
- Spacecraft: NASA has used hydrogen fuel cells since the Gemini program because of their high efficiency and the useful byproduct of drinkable water.
🛡️ Corrosion Prevention: Saving Structures
Mechanisms of Corrosion
Corrosion is the gradual destruction of a metal through electrochemical reactions with its environment. Iron rusting is the classic example. At the molecular level, iron atoms lose electrons (are oxidized):
Those electrons are picked up by oxygen and water at a nearby cathodic site:
The and ions combine and eventually form hydrated iron(III) oxide, which is rust. This is why corrosion speeds up in the presence of water and oxygen, and even faster with salt (which increases the conductivity of the solution, making electron transfer easier).
🧐 Why it matters: Corrosion costs the global economy hundreds of billions of dollars annually. It weakens bridges and infrastructure, shortens the lifespan of vehicles and ships, causes pipeline leaks in the oil and gas industry, and can lead to equipment failure in electronics and food processing.
Corrosion Prevention Techniques
- Sacrificial Anodes (Cathodic Protection): A more reactive metal (like zinc or magnesium) is attached to the structure you want to protect. Because it has a more negative reduction potential, the sacrificial anode oxidizes preferentially, sparing the structural metal. This is why zinc blocks are bolted to ship hulls.
- Protective Coatings: Paint, enamel, or metal plating (like galvanizing steel with zinc) creates a physical barrier between the metal and corrosive agents like water and oxygen.
- Alloying: Mixing metals to form alloys can dramatically improve corrosion resistance. Stainless steel contains chromium (at least 10.5%), which reacts with oxygen to form a thin, stable layer that prevents further oxidation of the iron beneath.
🎨 Electroplating: More Than Just Shiny Objects
Electroplating uses electrolysis to deposit a thin layer of metal onto a conductive surface. It serves both decorative and functional purposes.
Principles of Electroplating
The process works through electrolysis:
-
The object to be plated is connected as the cathode (negative electrode).
-
The plating metal (e.g., silver, chromium, gold) serves as the anode (positive electrode).
-
Both are immersed in an electrolyte solution containing dissolved ions of the plating metal.
-
When current flows, metal ions in solution are reduced at the cathode surface: e.g.,
-
The anode dissolves to replenish the metal ions in solution:
-
Over time, a thin, uniform layer of metal builds up on the cathode.
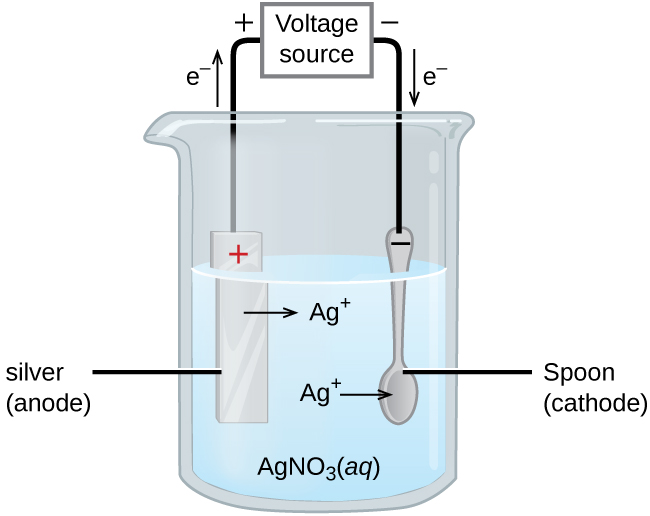
Image is an example of electroplating and a silver-plated tableware is produced in the setup.
Image courtesy of University of Wisconsin Pressbooks.
Applications of Electroplating
- Jewelry and Decorative Items: Gold or silver plating gives objects an attractive finish at a fraction of the cost of solid precious metal.
- Industrial Durability: Chromium or nickel plating on tools and machine parts provides a hard surface that resists friction, abrasion, and corrosion.
- Electronics: Thin gold or tin plating on connectors and circuit board contacts improves electrical conductivity and prevents oxidation, keeping connections reliable.
🌿 Environmental & Technological Implications
Clean Energy Technologies
Electrochemistry is central to many clean energy technologies. A few key examples:
- Fuel Cells convert hydrogen and oxygen directly into electricity, with water as the only byproduct (covered in detail above).
- Electrolyzers use electrical energy to split water into hydrogen and oxygen gas: . When powered by renewable electricity, this produces "green hydrogen" that can fuel vehicles or industrial processes with zero carbon emissions.
- Rechargeable Batteries (lithium-ion, etc.) store energy from solar panels and wind turbines for use when the sun isn't shining or the wind isn't blowing.
- Redox Flow Batteries store energy in liquid electrolyte solutions held in external tanks. Their capacity scales with tank size, making them flexible for large grid storage applications.
- Supercapacitors store energy electrostatically (through charge separation at electrode surfaces) rather than through chemical reactions. They charge and discharge very rapidly, making them useful for applications that need quick bursts of power.
Electrochemical Sensors & Monitoring
Electrochemical sensors detect specific chemicals by measuring current or voltage changes that occur when a target substance reacts at an electrode surface. They're used to monitor pollutants and contaminants in air, water, and soil. Their high selectivity (ability to detect one substance among many) and sensitivity make them valuable for environmental monitoring and, increasingly, for medical diagnostics like glucose monitors for diabetes management.
💼 Industrial Applications: Efficient Production Processes
Electrochemical Synthesis
Many important industrial chemicals are produced through electrolysis. For example, the chlor-alkali process electrolyzes brine (concentrated solution) to produce chlorine gas () at the anode and sodium hydroxide () at the cathode. Chlorine is used in water treatment, PVC production, and disinfectants, while sodium hydroxide is essential in paper manufacturing, soap production, and chemical synthesis.
Aluminum is another major product of industrial electrolysis. The Hall-Héroult process reduces aluminum oxide () dissolved in molten cryolite, producing pure aluminum metal. This process consumes enormous amounts of electricity, which is why aluminum smelters are often located near cheap hydroelectric power.
Practice Questions
- Compare primary versus secondary batteries regarding environmental impact.
- Explain why platinum is often used as a catalyst in PEMFCs despite its cost.
- Describe two methods by which corrosion can be prevented on large metallic structures such as bridges.
- How does electroplating improve wear resistance?
- Discuss how advancements in electrochemical sensor technology could positively affect healthcare diagnostics.
Answers:
-
Primary Batteries contain heavy metals (mercury, cadmium, etc.) that can contaminate soil and groundwater if disposed of improperly. Since they're single-use, they also generate more waste per unit of energy delivered. Secondary Batteries have significant environmental costs during manufacturing (mining lithium, cobalt, etc.), but because they're recharged hundreds of times, their resource consumption per use is much lower. They still require proper recycling to recover valuable materials and prevent toxic leaching.
-
Platinum has exceptionally high catalytic activity for both the hydrogen oxidation reaction (anode) and the oxygen reduction reaction (cathode) in PEMFCs. It's also chemically stable under the acidic conditions inside the membrane and maintains its performance over long periods. No other catalyst currently matches platinum's combination of activity, selectivity, and durability for these reactions, which justifies its cost.
-
Two common methods:
- Protective Coatings: Applying paint, epoxy, or galvanized zinc layers creates a physical barrier between the metal and moisture/oxygen, preventing the oxidation reaction from occurring.
- Cathodic Protection: Attaching sacrificial anodes (zinc or magnesium blocks) to the bridge structure. These more reactive metals oxidize preferentially, donating their electrons to the steel and keeping it in a reduced (protected) state.
-
Electroplating deposits a thin layer of a hard metal (such as chromium or nickel) onto a softer substrate. This outer layer acts as a protective barrier against friction and abrasion. The plated surface is harder and more corrosion-resistant than the base material, so the part lasts longer under mechanical stress.
-
Electrochemical sensors offer high sensitivity and selectivity in compact, portable formats. Advances in miniaturization and nanomaterials are enabling point-of-care testing devices that give rapid results without sending samples to a lab. Examples include continuous glucose monitors for diabetes patients, DNA sensors for genetic screening, and immunosensors that detect cancer biomarkers or infectious disease markers at very low concentrations. These tools support earlier diagnosis and more personalized treatment.