Energy, Work, and Heat in Chemical Reactions
Energy is a system's capacity to do work or transfer heat, and tracking how energy flows during chemical reactions is the central idea behind thermochemistry. This section covers the forms energy takes, how work and heat move energy between a system and its surroundings, and the tools used to measure those changes.

Types of Energy
- Kinetic Energy: The energy of motion. Atoms and molecules are constantly moving (translating, vibrating, rotating), and that motion is kinetic energy. The faster they move, the more kinetic energy they have.
- Potential Energy: Energy stored because of position or arrangement. In chemistry, the arrangement of atoms within chemical bonds stores potential energy.
- Chemical Energy: A specific form of potential energy held in chemical bonds. When bonds break and new ones form during a reaction, some of that stored energy can be released or absorbed.
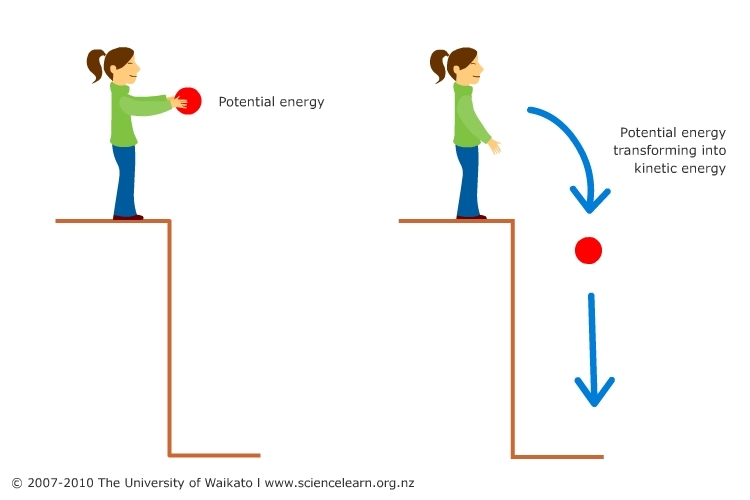
In the left image, a girl stands on a ledge, tightly gripping a ball, representing potential energy. The right image shows the ball dropped, symbolizing the conversion of potential energy to kinetic energy.
Image Courtesy of Sciencelearn.org
Units of Energy
- The SI unit of energy is the joule (J).
- Another common unit is the calorie (cal), where 1 cal = 4.184 J.
- You'll often see kilojoules (kJ) in thermochemistry since the joule is a relatively small unit.
Work and Chemical Reactions
Work (w) is the transfer of energy that occurs when a force moves an object through a distance. In chemistry, the most common type of work is pressure-volume (P-V) work, which happens when a gas expands or is compressed against an external pressure.
The negative sign is built into the equation by convention:
- When a gas expands (), the system does work on the surroundings, so is negative and the system loses energy.
- When a gas is compressed (), the surroundings do work on the system, so is positive and the system gains energy.
Heat Transfer in Reactions
Heat (q) is the transfer of thermal energy between a system and its surroundings due to a temperature difference. Energy always flows from the hotter object to the cooler one.
- Exothermic Reaction (): The system releases heat to the surroundings. The surroundings warm up. Example: Combustion of propane in a gas grill releases about .
- Endothermic Reaction (): The system absorbs heat from the surroundings. The surroundings cool down. Example: Dissolving ammonium nitrate in water feels cold to the touch because the process pulls heat from the water.
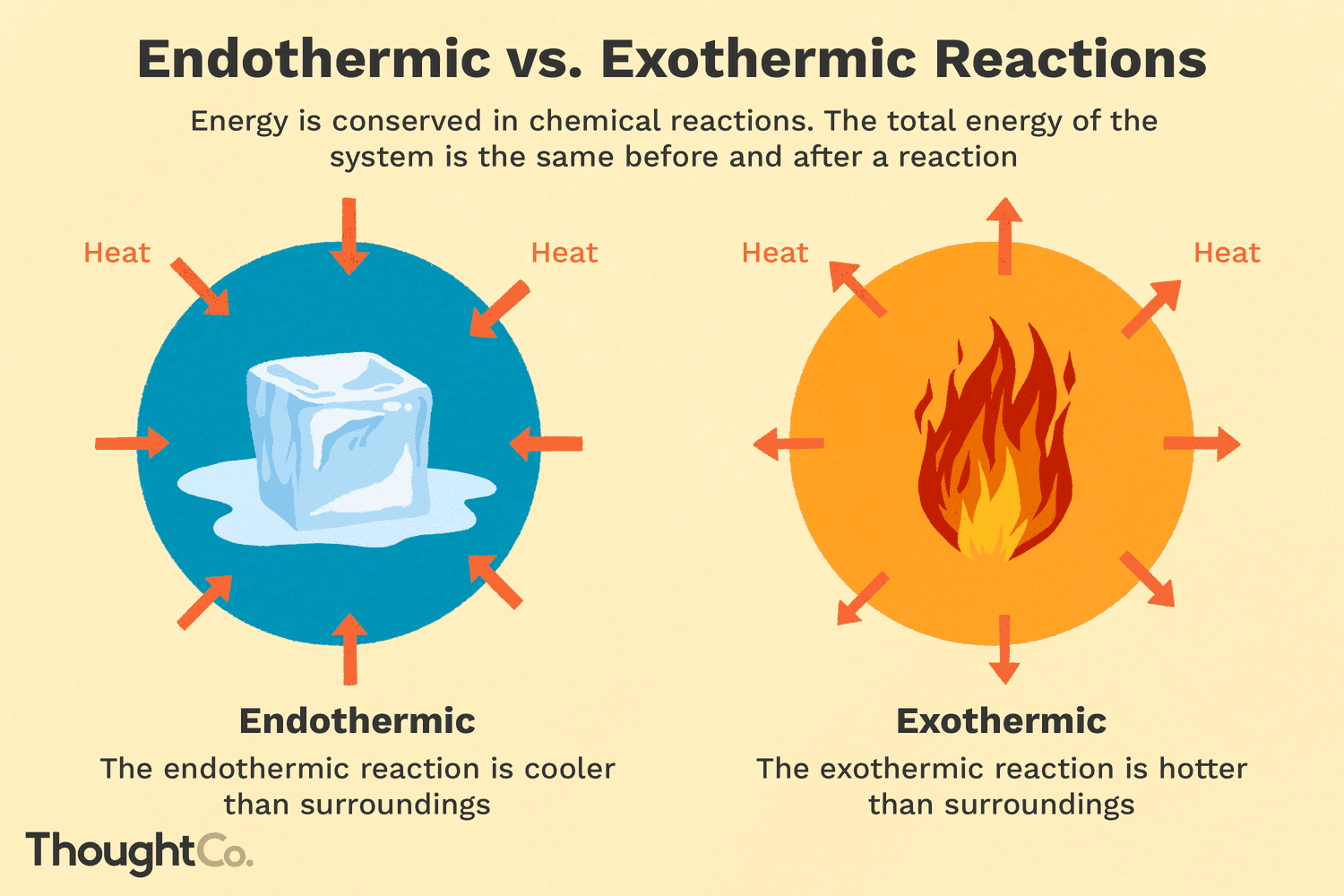
A graphic showing the comparison between endothermic and exothermic reactions, showing energy being absorbed or released during chemical reactions.
Image Courtesy of ThoughtCo
Enthalpy Change (ΔH)
Enthalpy change tells you the net heat absorbed or released at constant pressure:
- If , products are lower in energy than reactants → exothermic.
- If , products are higher in energy than reactants → endothermic.
Internal Energy (U) and the First Law of Thermodynamics
Internal energy (U) is the total energy contained within a system (kinetic + potential energy of all particles). The change in internal energy depends on both heat and work:
This is a statement of the first law of thermodynamics: energy is conserved. The system's energy can only change if heat flows in or out, or if work is done on or by the system.
- : The system gained energy overall.
- : The system lost energy overall.
Relationship Between Work, Heat, and Energy
Pressure-Volume Work (P-V Work)
When a gas expands or contracts against an external pressure, P-V work is done. This is the most relevant type of work in chemistry.
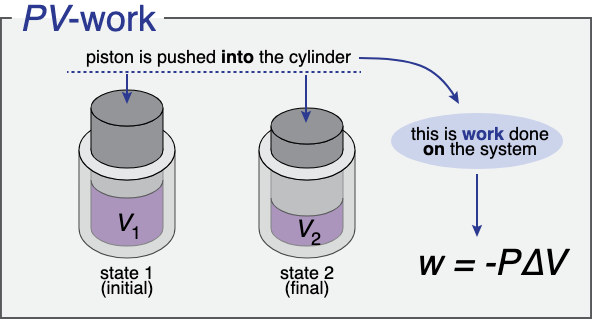
Left cylinder is state 1 (initial) with Volume 1 and before the piston is pushed in, and on right cylinder is state 2 (final) with Volume 2 which is lower than Volume 1 & after piston pushed in. Work is done on the system in this example.
Image Courtesy of Ch301
✏️ Practice Question
A gas in a piston-cylinder system expands against a constant external pressure of 5 atm, and its volume increases by 10 L. Calculate the work.
- Identify the values: , (positive because the gas expands).
- Plug into the formula:
The negative sign tells you the system did work on the surroundings (it expanded), so the system lost energy. To convert to joules, use , giving or about .
Heat Capacity
Heat capacity is the amount of heat needed to raise an object's temperature by 1°C. Specific heat capacity () narrows this down to a per-gram basis, making it useful for comparing different substances.
- = heat absorbed or released (J)
- = mass of the substance (g)
- = specific heat capacity (J/g·°C)
- = change in temperature (°C) =
Water, for example, has a high specific heat capacity of , which is why it takes a lot of energy to heat up and why it's used as the medium in calorimetry experiments.
Enthalpy (H) and Thermochemistry
Enthalpy (H) represents the heat content of a system at constant pressure. Since most reactions in the lab (and in life) happen at atmospheric pressure, enthalpy change is the most practical way to track energy flow in reactions.
Calculating ΔH Using Standard Enthalpies of Formation
Standard enthalpy changes () are measured at standard conditions: 298 K (25°C) and 1 atm. The standard enthalpy of formation for any element in its most stable form is defined as 0 kJ/mol.
✏️ Practice Question
Calculate for the combustion of methane:
Given values:
- :
- :
- :
- :
Solution:
- Sum the formation enthalpies of the products (multiply each by its coefficient):
- Sum the formation enthalpies of the reactants:
- Subtract:
The negative value confirms this is an exothermic reaction. (Note: the correct answer is , not .)
Calorimetry and Measuring Heat Changes
Calorimetry is the experimental technique for measuring heat exchanged during chemical or physical processes. A calorimeter is the insulated device used to do this.
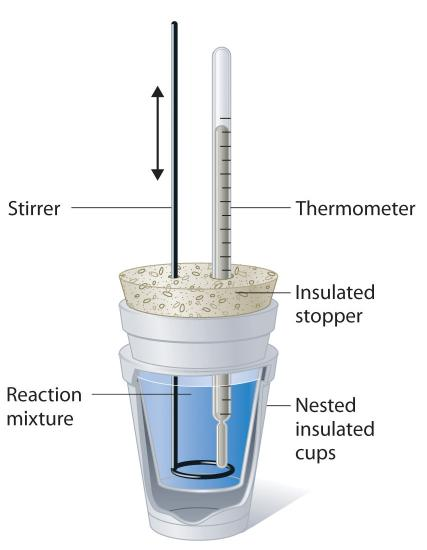
A thermometer stuck in an insulated stopper in a nested insulated cup and being utilized to gauge the temperature of a reaction mixture, allowing us to determine its degree of hotness or coldness.
Image Courtesy of LibreTexts.
Two main types:
- Constant-pressure calorimetry (coffee-cup calorimeter): Measures enthalpy changes () at atmospheric pressure. This is the type you'll most likely use in lab. The key assumption is that no heat escapes the calorimeter, so .
- Bomb calorimetry: Measures changes in internal energy () at constant volume. The reaction takes place in a sealed, rigid container. This is used for combustion reactions.
For both types, you calculate the heat using:
where is the specific heat capacity, is the mass of the solution or water in the calorimeter, and is the measured temperature change.
Closing
The big picture: energy is conserved (first law), and it moves between systems and surroundings as either heat or work. Exothermic reactions release heat (), endothermic reactions absorb it (), and you can measure these changes experimentally through calorimetry. When working through problems, pay close attention to signs, as they tell you the direction of energy flow.