Factors that Affect the Rate of Reactions
Chemical kinetics is all about understanding why some reactions happen in a flash while others take hours, days, or even years. Knowing the factors that control reaction rates gives you the ability to predict and manipulate how fast a reaction proceeds, which is central to everything from industrial manufacturing to biological processes.

🧪 The Role of Concentration in Reaction Rates
A higher concentration of reactants means more particles are packed into the same volume. More particles in the same space means more frequent collisions, and more collisions means a greater chance that reactant particles will successfully react. The result: a faster reaction rate.
Rate Laws
A rate law is a mathematical expression that shows exactly how reactant concentrations affect the speed of a reaction:
- is the rate constant (specific to each reaction at a given temperature)
- and are the molar concentrations of reactants A and B
- and are the reaction orders with respect to each reactant
The reaction orders ( and ) are determined experimentally. They tell you how sensitive the rate is to changes in each reactant's concentration. They are not simply taken from the balanced equation's coefficients.
Effects on Different Phases
Concentration changes affect substances differently depending on their phase. For gases, increasing the pressure in a closed container effectively increases the concentration of gas molecules (same number of moles in a smaller volume), which speeds up the reaction. For solids, concentration isn't really adjustable the same way, so surface area becomes the relevant factor instead (more on that below).
Practice Question
If you double the concentration of reactant A in a reaction where A is first-order and B is zeroth-order, by what factor does the reaction rate increase?
Explanation: For a first-order reactant, the rate is directly proportional to its concentration. Doubling doubles the rate. Since B is zeroth-order, changes in have no effect on the rate. The overall rate increases by a factor of 2.
🤨 How Temperature Affects Reaction Rates
Raising the temperature speeds up nearly all reactions for two reasons:
- Molecules move faster (higher average kinetic energy), so they collide more frequently.
- A larger fraction of those collisions now have enough energy to overcome the activation energy barrier, making a greater proportion of collisions effective.
Both effects combine to produce a significantly faster reaction rate. As a rough rule of thumb, increasing the temperature by about 10°C can roughly double the rate of many reactions.
Arrhenius Equation
The Arrhenius equation quantifies the relationship between temperature and the rate constant:
- = the frequency factor (related to how often molecules collide with proper orientation)
- = activation energy (the minimum energy needed for a successful collision)
- = the gas constant ()
- = temperature in Kelvin
Notice the negative exponent: as increases, becomes less negative, so gets larger, and increases. A higher means a faster reaction.
Industrial Implications
Temperature control is critical in industrial chemistry. Running a reaction too hot can cause dangerous runaway reactions or unwanted side products. Running it too cool wastes time and money. Engineers carefully optimize temperature to balance safety, product yield, and efficiency.
🏃🏾♂️ Catalysts
A catalyst speeds up a reaction by providing an alternative reaction pathway with a lower activation energy. The catalyst itself is not consumed and emerges chemically unchanged at the end of the reaction.
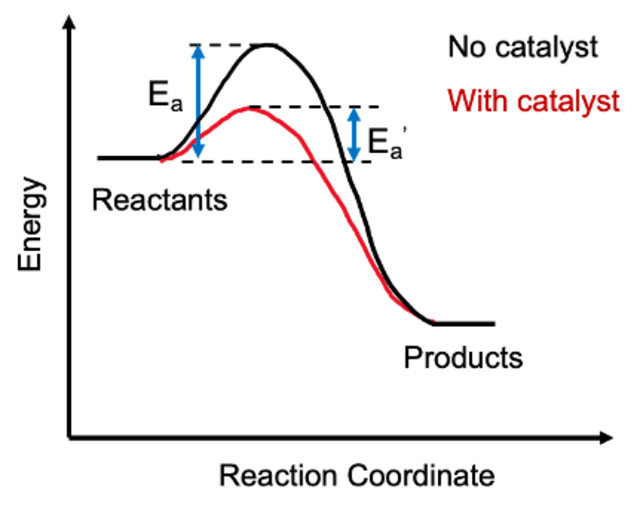
Image displaying how catalysts speed up enzymatic reactions.
Image Courtesy of Wikimedia Commons.
Homogeneous vs Heterogeneous Catalysts
- Homogeneous catalysts exist in the same phase as the reactants (e.g., an acid catalyst dissolved in an aqueous reaction mixture).
- Heterogeneous catalysts exist in a different phase from the reactants (e.g., a solid platinum surface catalyzing a gas-phase reaction).
Enzymes are biological catalysts (proteins) with highly specific active sites that bind to particular molecules called substrates. This specificity is why enzymes typically catalyze only one reaction or a narrow set of reactions.
Activation Energy: Barrier to Reaction
Activation energy () is the minimum energy that colliding particles must possess to successfully react and form products. Think of it as an energy hill that reactants must climb over to reach the product side.
- If is low, a large fraction of collisions have enough energy to react, so the reaction is fast.
- If is high, only a small fraction of collisions are energetic enough, so the reaction is slow.
At the top of this energy hill is the transition state, a brief, high-energy arrangement where old bonds are partially broken and new bonds are partially formed. From there, the system either proceeds to products or falls back to reactants.
Catalysts work by lowering , which means more collisions can successfully reach the transition state at any given temperature.
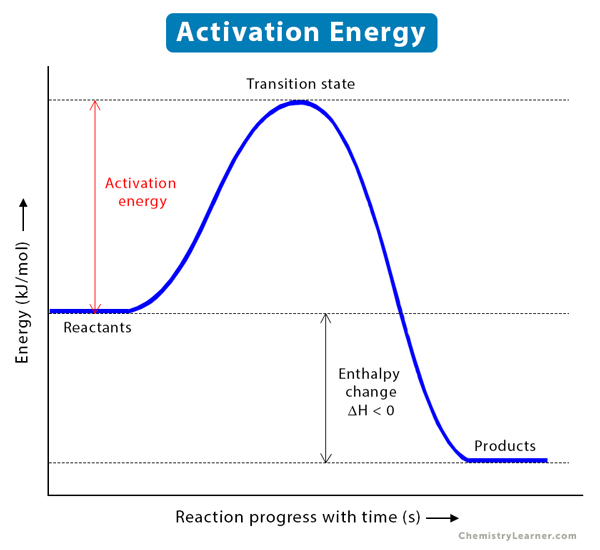
Image displaying the activation energy.
Image Courtesy of Chemistry Learner.
🤓 Factors Affecting Gas and Solid Reactions
Collision Theory
Collision theory states that for a reaction to occur, particles must:
- Collide with each other
- Collide with sufficient energy (at least equal to )
- Collide with proper orientation so that the reactive parts of the molecules are facing each other
All three conditions must be met. A collision that lacks enough energy or has the wrong orientation will simply bounce off without reacting.
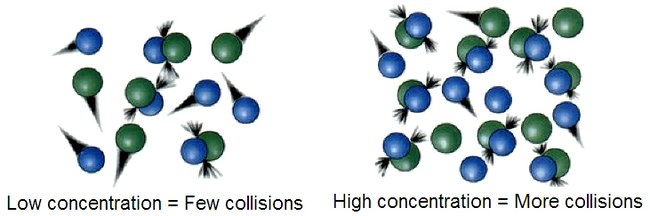
Image showing the effects of the collision theory.
Image Courtesy of Wikipedia.
Pressure's Impact on Gas Reactions
Increasing the pressure on gases in a closed container forces the same number of molecules into a smaller volume. This raises the effective concentration, which increases collision frequency and speeds up the reaction.
Note: Le Chatelier's principle describes how a system at equilibrium responds to stress, which is a separate concept from reaction rate. Pressure affects rate because of increased concentration, not because of Le Chatelier's principle. You'll encounter Le Chatelier's principle more when you study equilibrium later in this unit.

Image showing the impact of pressure on gas reactions.
Image Courtesy of Chem4Kids.
Surface Area for Solids
For reactions involving solids, only the particles on the surface are exposed to collisions with other reactants. Increasing the surface area exposes more particles at once. Crushing or grinding a solid into smaller pieces dramatically increases the surface area available for reaction, which speeds things up significantly.
This is why powdered sugar dissolves faster than a sugar cube, and why dust explosions can occur in grain elevators when fine particles are suspended in air.
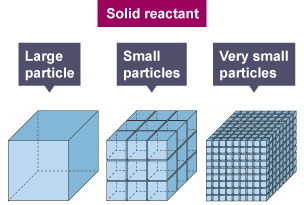
Image showing the effect of surface area in a solid reactant.
Image Courtesy of BBC.
🌿 Nature of Reactants & Products
🧬 Chemical Nature's Role
Not all reactants are created equal. Ionic compounds dissolved in solution tend to react faster because their ions are already dissociated and free to interact. Covalent compounds often react more slowly because covalent bonds must be broken, which requires more energy input.
Bond strength matters too. Molecules with stronger bonds generally react more slowly because more energy is needed to break those bonds. The overall molecular structure and complexity of the reactants also play a role in determining how easily collisions lead to products.
☀️ Light-Driven Reactions
Some reactions require light energy (photons) to proceed. These are called photochemical reactions. The energy of the light depends on its wavelength: shorter wavelengths (like UV light) carry more energy per photon than longer wavelengths (like visible light).
For example, UV light can break bonds in certain monomers to initiate polymerization, and it can also decompose molecules like hydrogen peroxide. The intensity (brightness) of the light also matters, since more photons per second means more molecules can be activated.
Image showing the effect of light-driven reactions on chemistry.
Image Courtesy of PBS.
❓ Conceptual Question
What might happen to a photochemical reaction's speed if you switch from ultraviolet light to visible light?
Explanation: UV photons carry more energy than visible light photons. If a reaction requires a certain minimum photon energy to break bonds or reach the activation energy, visible light may not provide enough energy per photon. The reaction would likely slow down or might not proceed at all, depending on the specific energy threshold required.
✍🏽 Reaction Mechanisms
Most complex reactions don't happen in a single step. Instead, they proceed through a series of elementary steps that together make up the reaction mechanism. Key concepts within mechanisms include:
-
Initiation - The first step where bonds break or form to start the reaction process.
-
Intermediates - Temporary species that are produced in one step and consumed in a later step. They don't appear in the overall balanced equation, but identifying them reveals how the reaction actually works.
-
Propagation - Steps where intermediate species react to form other intermediates or final products. Each propagation step has its own rate constant.
-
Branching and Side Reactions - Alternative pathways that produce different (often unwanted) products. These can complicate the mechanism and reduce the yield of the desired product.
-
Rate-Determining Step - The slowest elementary step in the mechanism. Because the overall reaction can only go as fast as its slowest step, this step controls the overall reaction rate. Identifying it is the key to understanding and predicting the kinetics of the whole reaction.
⭐️ Conclusion
The major factors that affect reaction rate are concentration, temperature, catalysts, surface area (for solids), pressure (for gases), the nature of the reactants, and light (for photochemical reactions). Each of these connects back to collision theory: they either change how often particles collide, how much energy those collisions carry, or how effectively the collisions are oriented. Keep these connections in mind, and the individual factors will make much more sense as a unified picture.