Electrochemical Cells
Electrochemical cells convert between chemical energy and electrical energy using redox reactions. Understanding how these cells work is central to electrochemistry, from explaining how batteries generate power to why electrolysis can extract pure metals from solutions. This guide covers galvanic and electrolytic cells, standard electrode potentials, and the Nernst equation.

Principles of Galvanic and Electrolytic Cells
Electrochemical cells are devices where redox reactions either produce or consume electrical energy. The two main types are galvanic cells and electrolytic cells.
Galvanic (Voltaic) Cells
Galvanic cells convert chemical energy into electrical energy through spontaneous redox reactions. Every battery you've ever used is based on this principle. Here are the key components:
- Anode — the negative electrode where oxidation occurs (electrons are lost). The anode material acts as the reducing agent. Electrons flow away from the anode through the external circuit.
- Cathode — the positive electrode where reduction occurs (electrons are gained). The cathode material acts as the oxidizing agent, accepting electrons that arrive through the external circuit.
- Electrolyte — a solution containing dissolved ions. It allows ion movement between the two half-cell compartments, which is necessary to maintain charge balance as electrons flow through the wire.
- External circuit — the conductive wire connecting the anode to the cathode. The flow of electrons through this wire is the electrical current we harness.
- Salt bridge — connects the two half-cell solutions and allows ions to migrate between compartments. Without it, charge would build up in each compartment and the reaction would stop almost immediately.
A helpful mnemonic: An Ox, Red Cat (Anode = Oxidation, Reduction = Cathode). This applies to both galvanic and electrolytic cells.
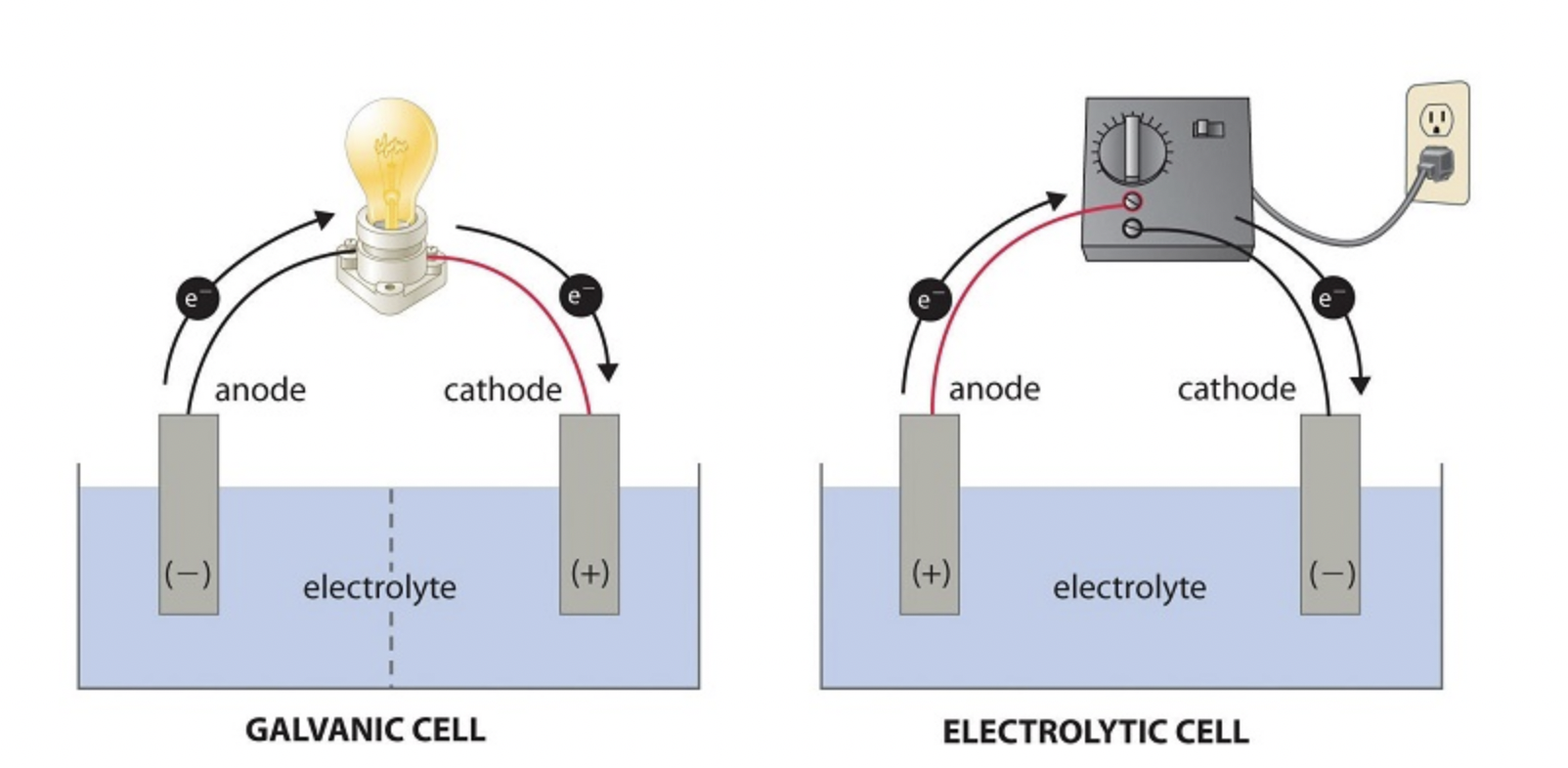
Image depicting a side-by-side comparison diagram showing the setup of both a galvanic cell and an electrolytic cell with labeled parts.
Image courtesy of Chemistry LibreTexts.
Electrolytic Cells
Electrolytic cells do the opposite of galvanic cells: they use electrical energy from an external power source to drive non-spontaneous reactions. This process is called electrolysis.
Because the reaction wouldn't happen on its own, you have to supply a voltage that exceeds the minimum required to force the reaction forward. The electrode labels flip in terms of sign: in an electrolytic cell, the anode is positive and the cathode is negative (the external source pushes electrons into the cathode and pulls them from the anode).
Comparison: Galvanic vs. Electrolytic Cells
Here's what sets these two apart:
- Reaction type: Galvanic cells run on spontaneous reactions (). Electrolytic cells force non-spontaneous reactions ().
- Energy conversion: Galvanic cells convert chemical → electrical energy. Electrolytic cells convert electrical → chemical energy.
- Electron flow: Electrons flow from anode to cathode in both types, but in galvanic cells this happens naturally, while in electrolytic cells it's driven by an external voltage source.
- Electrode signs: In a galvanic cell, the anode is negative and the cathode is positive. In an electrolytic cell, the anode is positive and the cathode is negative.
Practical applications:
- Galvanic cells: Batteries in phones, cars, watches, flashlights.
- Electrolytic cells: Electroplating jewelry with silver or gold, refining aluminum from ore, producing chlorine gas industrially.
Think of galvanic cells as providing electricity, and electrolytic cells as consuming electricity to make chemistry happen.
Standard Electrode Potentials (E°)
Standard electrode potentials ( values) measure how strongly a species tends to gain electrons (be reduced) under standard conditions: 1 M concentration for solutions, 1 atm pressure for gases, and 25°C (298 K).
These values are always written as reduction potentials. The standard hydrogen electrode (SHE) is assigned and serves as the reference point for all other half-reactions.
The electrochemical series ranks half-reactions by their values. Species with more positive values are more easily reduced (stronger oxidizing agents). Species with more negative values are more easily oxidized (stronger reducing agents).
Calculating Cell Potential
To find the standard cell potential for a galvanic cell:
Both values used in this equation are the standard reduction potentials from a reference table. You don't flip the sign of the anode's value before plugging it in; the subtraction takes care of that.
If is positive, the reaction is spontaneous under standard conditions. If it's negative, the reaction is non-spontaneous.
The Nernst Equation
Under non-standard conditions (different concentrations, pressures, or temperatures), the actual cell potential shifts from . The Nernst equation lets you calculate this adjusted potential:
Where:
- = cell potential under non-standard conditions
- = standard cell potential
- = universal gas constant (8.314 J·mol⁻¹·K⁻¹)
- = temperature in Kelvin
- = number of moles of electrons transferred in the balanced equation
- = Faraday's constant (96,485 C/mol)
- = reaction quotient (same form as the equilibrium expression, but using current concentrations)
At 298 K, this simplifies to a form you'll use most often:
The key idea: as products build up ( increases), the cell potential decreases. When (equilibrium), and the cell is "dead."
Experimental Setup & Observations
When you set up electrochemical cells in lab, watch for these common observations:
- Metal deposition on the cathode during electrolysis (e.g., copper plating out of a solution).
- Gas evolution at electrodes (e.g., hydrogen bubbles at the cathode or oxygen bubbles at the anode during water electrolysis).
- Color changes in the electrolyte as ion concentrations shift.
Measure voltage with a voltmeter across the two electrodes. Comparing your measured voltage to the calculated is a great way to see how real-world conditions (impure solutions, resistance in wires) cause deviations from theoretical values.
Guided Practice Questions:
-
Calculate the standard cell potential for a galvanic cell with a zinc anode (, ) and a copper cathode (, ). Is this reaction spontaneous?
Answer:
Using :
Since is positive, the reaction is spontaneous under standard conditions.
-
For an electrolytic process using a silver cation solution (), if we apply a voltage greater than its standard reduction potential of , will silver metal plate out onto the cathode? Explain.
Answer:
Yes. In electrolysis, the external voltage must exceed the minimum potential needed to drive the non-spontaneous reaction. By applying more than , you supply enough energy to reduce ions to solid atoms, which deposit onto the cathode surface. This is the basis of silver electroplating.
-
Calculate the cell potential for a voltaic cell based on the following half-reaction and conditions:
Given: , , ,
Answer:
Step 1: Determine the reaction quotient. For this half-reaction,
Step 2: Apply the Nernst equation at 298 K. Since one electron is transferred ():
Step 3: Calculate
The cell potential is higher than standard because the product concentration () is below standard, which shifts the reaction forward (Le Chatelier's principle in action).