Chemical Equilibrium
Chemical equilibrium describes the state of a reversible reaction where the forward and reverse reactions occur at equal rates, so the concentrations of reactants and products stay constant over time. Understanding equilibrium is central to predicting how reactions behave and how conditions like temperature and pressure can shift the outcome.

🔄 The Dynamic Nature of Chemical Equilibrium
What is Chemical Equilibrium?
In many reactions, products can react to re-form the original reactants. This makes the reaction reversible. At chemical equilibrium, the forward reaction (reactants → products) and the reverse reaction (products → reactants) proceed at the same rate.
The key word here is dynamic. At equilibrium, both reactions are still happening constantly. The concentrations just aren't changing because the two rates are equal. Nothing has "stopped."
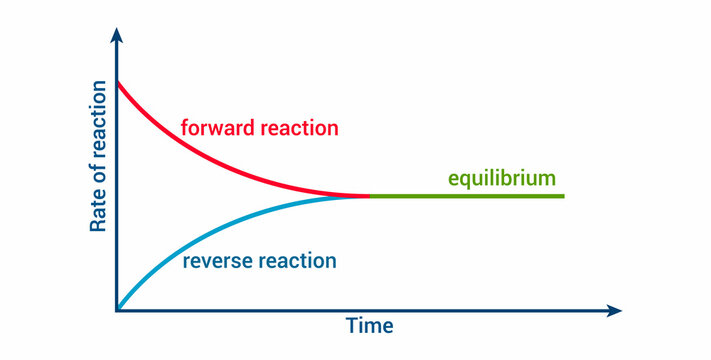
Image displaying reaction rates and the state of equilibrium.
Image Courtesy of Adobe Stock
Closed System Requirement
Chemical equilibrium can only be established in a closed system, meaning no matter enters or leaves. If the system were open, products (especially gases) could escape, and the reverse reaction could never keep pace with the forward reaction. 🔒
🔢 Calculating and Interpreting the Equilibrium Constant
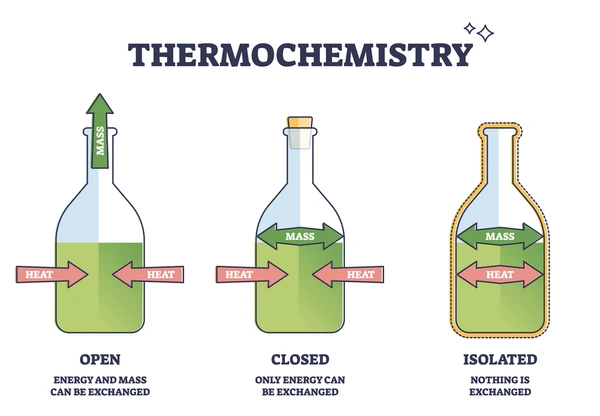
Image showing the difference between open, closed, and isolated systems.
Image Courtesy of Shutterstock
What is an Equilibrium Constant?
The equilibrium constant () is a ratio that compares the concentrations (or pressures) of products to reactants at equilibrium, each raised to the power of its stoichiometric coefficient. For a given reaction at a given temperature, is always the same value regardless of starting amounts.
Types of K
For a general balanced equation:
- uses molar concentrations (mol/L):
- uses partial pressures (atm), and applies only to gaseous equilibria:
Note: Pure solids and pure liquids are not included in equilibrium expressions because their concentrations are constant.
How to Calculate K
- Write the balanced chemical equation.
- Identify the stoichiometric coefficients; these become the exponents in the expression.
- Plug in the equilibrium concentrations (for ) or partial pressures (for ). Don't use initial values.
- Calculate the ratio of products over reactants.
High vs. Low K Values
- : At equilibrium, products are present in greater amounts than reactants. The reaction "favors products."
- : At equilibrium, reactants dominate. The reaction "favors reactants."
- : Neither side is strongly favored; significant amounts of both reactants and products are present.
The farther is from 1 in either direction, the more strongly the equilibrium lies toward that side.
Temperature and K
Temperature is the only factor that changes the value of . (Changing concentration or pressure shifts the position of equilibrium but doesn't change itself.)
- If increases with rising temperature, the forward reaction is endothermic (heat acts like a reactant).
- If decreases with rising temperature, the forward reaction is exothermic (heat acts like a product).
⚖️ Factors Affecting Chemical Equilibrium
Le Chatelier's Principle
Le Chatelier's Principle states that if a stress is applied to a system at equilibrium, the system will shift in the direction that partially counteracts that stress. The three main stresses are changes in concentration, pressure, and temperature.
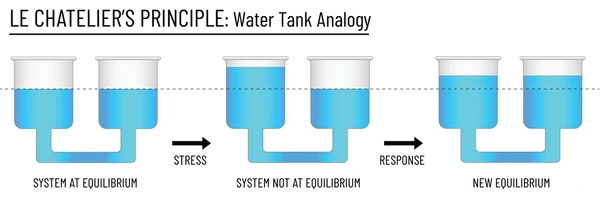
Image displaying Le Chatelier's Principle.
Image Courtesy of Shutterstock
Concentration Changes
- Adding a reactant or product shifts the equilibrium to the opposite side (to consume what was added).
- Removing a reactant or product shifts the equilibrium toward that side (to replenish what was removed).
For example, in , adding more shifts the equilibrium to the right, producing more .
Pressure Changes (Gases Only)
Pressure changes affect equilibria that have unequal moles of gas on each side.
- Increasing pressure (or decreasing volume) shifts equilibrium toward the side with fewer moles of gas.
- Decreasing pressure (or increasing volume) shifts equilibrium toward the side with more moles of gas.
If both sides have the same number of moles of gas, pressure changes have no effect on the equilibrium position.
Temperature Changes
Temperature is unique because it actually changes the value of .
- Increasing temperature favors the endothermic direction (the direction that absorbs heat).
- Decreasing temperature favors the exothermic direction (the direction that releases heat).
A useful trick: write "heat" into the equation as if it were a reactant (endothermic) or product (exothermic), then treat temperature changes like concentration changes.
Catalysts' Role
Catalysts speed up both the forward and reverse reactions equally. They help the system reach equilibrium faster, but they do not change the equilibrium position or the value of .
🌏 Real-World Applications of Chemical Equilibrium
Industrial Syntheses
The Haber process (, exothermic) is a classic example. Engineers use Le Chatelier's Principle to maximize ammonia yield:
- High pressure shifts equilibrium toward (fewer moles of gas on the product side: 2 vs. 4).
- Moderate temperature (~400–500°C) is used as a compromise. Lower temperatures favor the exothermic forward reaction, but the reaction becomes too slow. A catalyst (iron) helps speed things up at this moderate temperature.
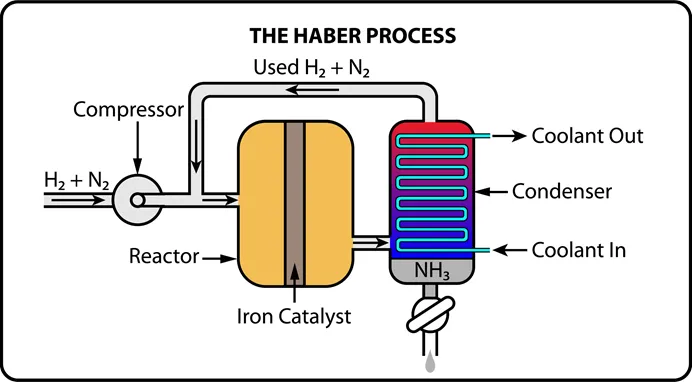
Image showing how the Haber Process works.
Image Courtesy of Shalom Education
Environmental Implications
Atmospheric ozone () exists in a dynamic equilibrium. Human-produced chemicals like CFCs disrupt this equilibrium by accelerating ozone destruction, shifting the balance toward depletion. Understanding equilibrium helps scientists model and predict these environmental changes.
✏️ Chemical Equilibrium Practice Questions
-
The value of for a certain reaction is 0.0012 at room temperature. Does this favor reactants or products?
Explanation: A value of 0.0012 is much less than 1, which means reactants are present in far greater concentrations than products at equilibrium. This reaction favors reactants.
-
Explain what would happen to an exothermic reaction at equilibrium if the temperature is increased.
Explanation: For an exothermic reaction, heat is effectively a product. Increasing the temperature adds heat to the system, so by Le Chatelier's Principle, the equilibrium shifts to the left (toward reactants) to consume the added heat. This means the value of decreases, and more reactants are present at the new equilibrium.