Types and Solubility Factors
A solution is a homogeneous mixture of two or more substances. The solute is the substance being dissolved, and the solvent is the substance doing the dissolving. Solubility is the maximum amount of solute that can dissolve in a given amount of solvent at a specific temperature and pressure, often expressed in grams of solute per 100 g of solvent or in moles per liter (molarity).
These concepts show up everywhere in chemistry, from predicting reaction outcomes to understanding biological and industrial processes.

🏷️ Types of Solutions
Solutions aren't limited to liquids. They can exist in any phase, depending on the states of the solute and solvent.
Gaseous Solutions
Gaseous solutions are homogeneous mixtures of two or more gases uniformly distributed throughout one another. The atmosphere is a classic example (, , , , etc.).
- Dalton's Law of Partial Pressures governs gas mixtures: the total pressure equals the sum of each gas's partial pressure.
Each is the partial pressure of an individual gas. In an ideal gas mixture, a gas's partial pressure is proportional to its mole fraction.
- Henry's Law describes gas solubility in liquids: the solubility of a gas in a liquid is directly proportional to the partial pressure of that gas above the liquid. This is why a sealed soda bottle stays carbonated under high pressure but goes flat once opened.
- Gas molecules mix rapidly and spontaneously due to their high kinetic energy.
- Temperature effect on gases: Increasing temperature decreases gas solubility. Higher kinetic energy makes it easier for dissolved gas molecules to escape back into the gas phase. Think of how a warm soda goes flat faster than a cold one.
Liquid Solutions
Aqueous Solutions
The solvent is water. These are the most common solutions you'll encounter in chemistry.
- Aqueous solutions containing dissolved ions (electrolytes) can conduct electricity.
- When ionic compounds dissolve, water molecules surround each ion, forming hydration shells. The partially negative oxygen end of water orients toward cations, and the partially positive hydrogen end orients toward anions.
- Dissolved solutes affect the solution's physical properties (density, boiling point, freezing point) compared to pure water. You'll explore these colligative properties more in later sections.
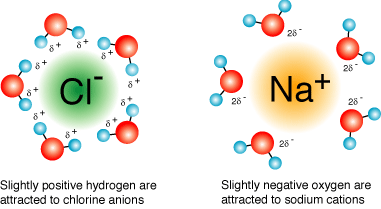
Aqueous solution with ions surrounded by hydration shells.
Image Courtesy of Socratic
Non-aqueous Solutions
The solvent is any liquid other than water, such as ethanol, acetone, or hexane. These solvents are typically organic compounds that are liquid at room temperature.
- Miscibility (the ability of two liquids to mix) depends on polarity. Polar solvents dissolve polar solutes well; nonpolar solvents dissolve nonpolar solutes well. This is the "like dissolves like" principle.
- Non-aqueous solutions generally have lower electrical conductivity than aqueous ionic solutions because fewer free ions are present.
Solid Solutions
Solid solutions form when one substance is uniformly distributed throughout another in the solid phase. Alloys are the most familiar examples: brass (copper + zinc) and steel (iron + carbon).
- Formed by melting the component metals together and then cooling the mixture so the atoms arrange into a uniform solid structure.
- Like all solutions, solid solutions have a solubility limit for how much solute the solid solvent can incorporate.
- Alloying often enhances properties like strength, hardness, or corrosion resistance compared to the pure metals alone.
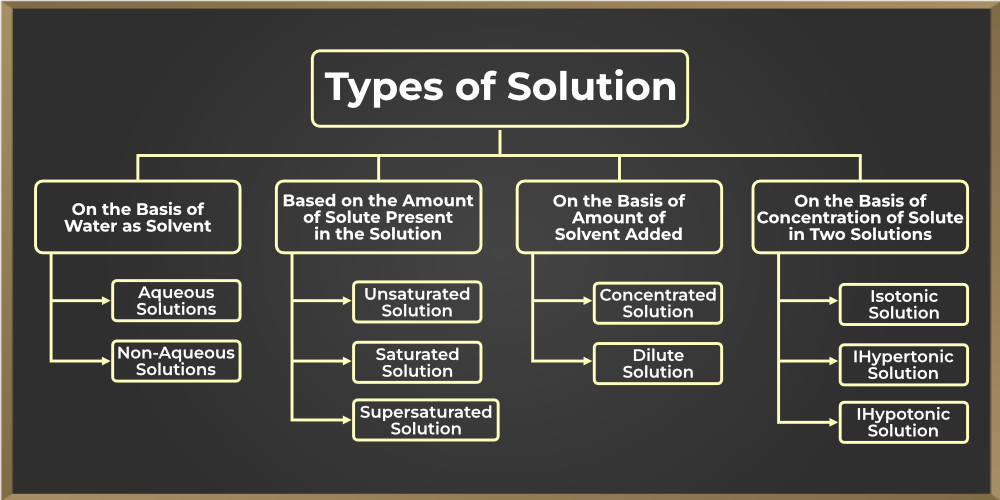
Different types of solutions
Image Courtesy of GeeksforGeeks
🌟 Factors Affecting Solubility
Three main factors determine how much solute can dissolve in a given solvent.
Temperature
For most solid and liquid solutes, higher temperatures increase solubility. Extra thermal energy helps break apart solute particles and increases interactions with solvent molecules.
For gases, the opposite is true: higher temperatures decrease solubility. Gas molecules gain kinetic energy and escape the liquid more easily.
Solubility curves (graphs of solubility vs. temperature) let you predict exactly how much of a solid will dissolve at a given temperature. Most curve upward for solids, but a few (like ) actually decrease with temperature.
Pressure
Pressure has a negligible effect on the solubility of solids and liquids. But for gases dissolved in liquids, pressure matters a lot.
Henry's Law states:
where is the concentration of dissolved gas, is the Henry's Law constant (specific to each gas-solvent pair), and is the partial pressure of the gas above the solution. Double the pressure, double the amount of gas that dissolves.
Nature of Solute and Solvent
The guiding principle is "like dissolves like."
- Polar solvents (like water) dissolve polar solutes and ionic compounds. Water dissolves because its polar molecules can stabilize the separated and ions.
- Nonpolar solvents (like hexane) dissolve nonpolar solutes. Fats and oils dissolve readily in nonpolar organic solvents but not in water.
This comes down to intermolecular forces. Dissolution occurs when solute-solvent attractions are strong enough to overcome solute-solute and solvent-solvent attractions.
🧪 Saturation & Supersaturation

Image shows types of solutions.
Image Courtesy of Toppr
Unsaturated Solutions
An unsaturated solution contains less solute than the maximum the solvent can hold at that temperature. You could still dissolve more solute into it.
Saturated Solutions
A saturated solution holds the maximum amount of dissolved solute at a given temperature. At this point, the rate of dissolution equals the rate of crystallization, creating a dynamic equilibrium. Any additional solute you add will simply sit undissolved.
Supersaturated Solutions
A supersaturated solution contains more dissolved solute than should be possible under normal conditions. This is typically achieved by dissolving solute in a hot solvent and then cooling very slowly and carefully.
Supersaturation is an unstable state. Disturbing the solution, such as adding a tiny seed crystal or even scratching the container wall, provides nucleation sites that trigger rapid crystallization. The excess solute crashes out of solution until the system reaches its normal saturation point. Sodium acetate "hot ice" is a classic demonstration of this.

Image showing crystallization.
Image Courtesy of SolutionInn
✏️ Practice Questions
-
What happens to the solubility of most solid substances as temperature increases?
Explanation: Solubility typically increases. The added thermal energy helps disrupt interactions holding the solid together, allowing more solute particles to be pulled apart and surrounded by solvent molecules.
-
Describe what happens when you add more sugar into a saturated sugar-water solution at room temperature.
Explanation: The extra sugar will not dissolve. It accumulates at the bottom of the container as a solid because the solution has already reached its maximum capacity for dissolved sugar at that temperature.
-
Explain how pressure affects the dissolution of a gas using Henry's Law.
Explanation: Henry's Law () states that at constant temperature, the concentration of dissolved gas is directly proportional to its partial pressure above the liquid. Increasing the pressure forces more gas molecules into solution until a new equilibrium is reached.
-
Predict what happens to a supersaturated sodium acetate solution when a seed crystal is added.
Explanation: The seed crystal provides nucleation sites that disrupt the unstable supersaturated state. Excess solute rapidly crystallizes out of solution, releasing heat in the process, until the solution reaches its normal saturation concentration.
-
If you have two liquids that do not mix, like oil and water, can you call one the solvent and the other the solute?
Explanation: Not really. Oil and water are immiscible, meaning they don't form a homogeneous mixture. The terms "solute" and "solvent" only apply when one substance fully dissolves into another to form a true solution.