Functional Groups and Reactions

Introduction to Functional Groups
Functional groups are specific arrangements of atoms within organic molecules that determine how those molecules behave in chemical reactions. The carbon backbone of an organic molecule is relatively unreactive on its own. The functional group is where the chemistry actually happens: it controls reactivity, physical properties, and how the compound interacts with other substances.
Identifying Common Functional Groups
A functional group is a specific combination of atoms (and bond types) attached to a carbon skeleton that gives the compound its characteristic chemical properties. Molecules with the same functional group tend to undergo similar reactions, which is why recognizing these groups is so useful.
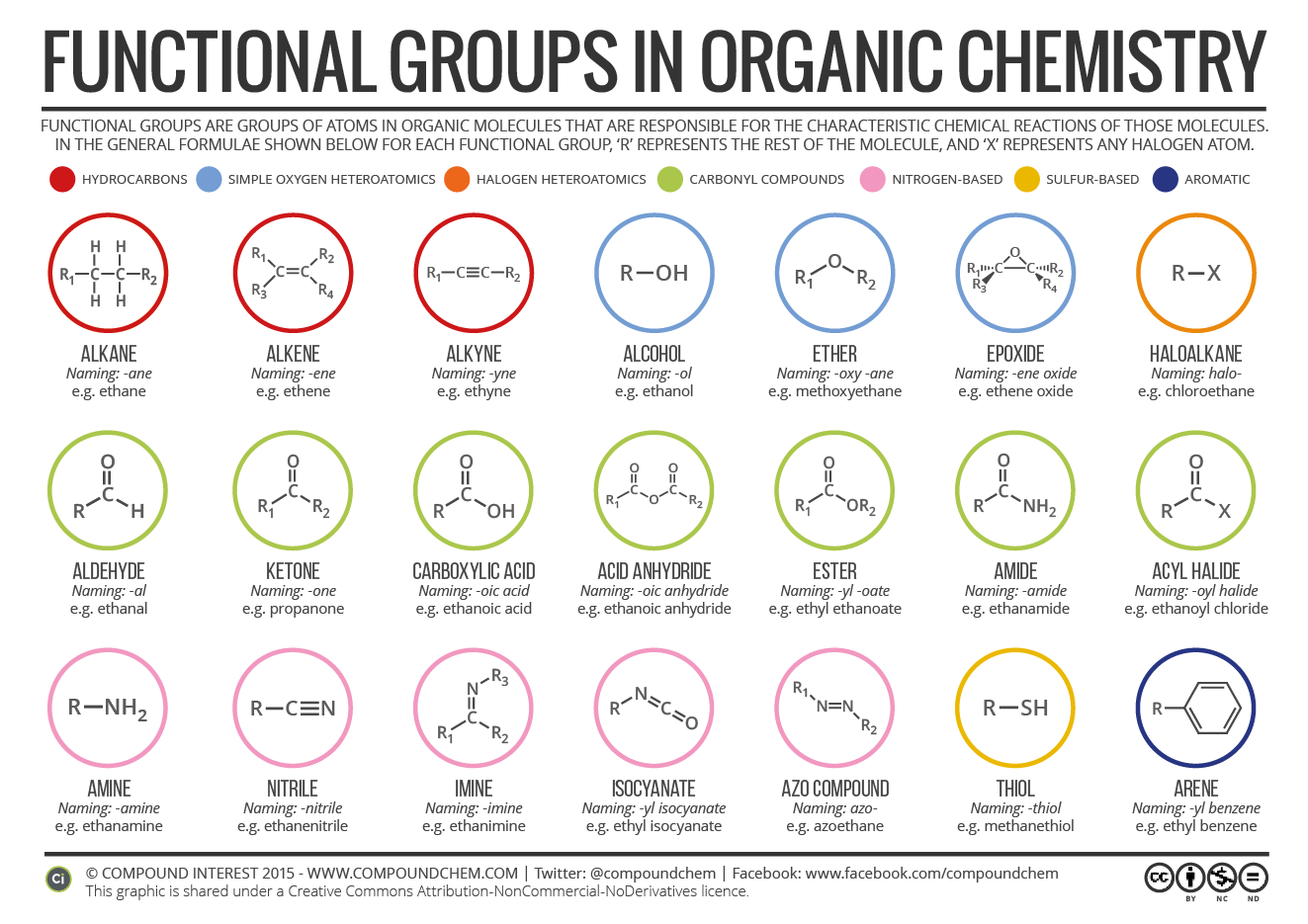
Image showing all of the functional groups within organic chemistry.
Image Credits: Compound Interest
-
Alkanes (Saturated Hydrocarbons)
- Contain only carbon-carbon single bonds (). Because all bonds are single, alkanes are relatively unreactive and serve as the baseline hydrocarbon structure.
-
Alkenes
- Contain at least one carbon-carbon double bond (). The double bond is a site of higher electron density, making alkenes more reactive than alkanes, especially in addition reactions.
-
Alkynes
- Contain at least one carbon-carbon triple bond (). Even more reactive than alkenes due to the concentration of electron density in the triple bond.
-
Alcohols (Hydroxyl Group)
- Contain a hydroxyl group () bonded to a carbon. The group is polar and can form hydrogen bonds, which significantly affects solubility and boiling point.
-
Amines
- Contain a nitrogen atom bonded to one or more hydrogens and/or carbon groups (, , or ). The lone pair on nitrogen makes amines act as bases.
-
Ethers
- Feature an oxygen atom bonded between two carbon groups (). Ethers are relatively unreactive and are often used as solvents.
-
Aromatics
- Built around a benzene ring (), a six-carbon ring with delocalized electrons. This delocalization makes the ring unusually stable, so aromatics tend to undergo substitution reactions rather than addition.
-
Aldehydes
- Contain a carbonyl group () at the end of a carbon chain (). Aldehydes can be oxidized to carboxylic acids or reduced to primary alcohols.
-
Ketones
- Contain a carbonyl group () bonded to two carbon groups within the chain. Ketones can be reduced to secondary alcohols but are harder to oxidize than aldehydes.
-
Carboxylic Acids
- Contain both a carbonyl and a hydroxyl group (). The combination allows the hydrogen to be donated as , making these compounds acidic. They react to form esters and amides.
-
Esters
- Contain a carbonyl group bonded to an oxygen that connects to another carbon (). Esters can undergo hydrolysis, breaking back into a carboxylic acid and an alcohol.
Properties of Organic Compounds
The functional groups present in an organic compound directly shape its physical and chemical behavior. Polar functional groups like or introduce stronger intermolecular forces, while nonpolar groups like keep intermolecular forces weak. This single idea explains most of the property trends below.
Physical Properties
Understanding physical properties helps you predict how a compound will behave under different conditions, from lab distillations to biological systems.
- Boiling and Melting Points: Functional groups that can hydrogen bond (, , ) raise boiling and melting points significantly. For example, ethanol (, bp 78°C) boils much higher than dimethyl ether (, bp −24°C) despite having the same molecular formula, because ethanol's group forms hydrogen bonds. Halogen-containing groups raise these points through stronger dipole-dipole interactions and London dispersion forces.
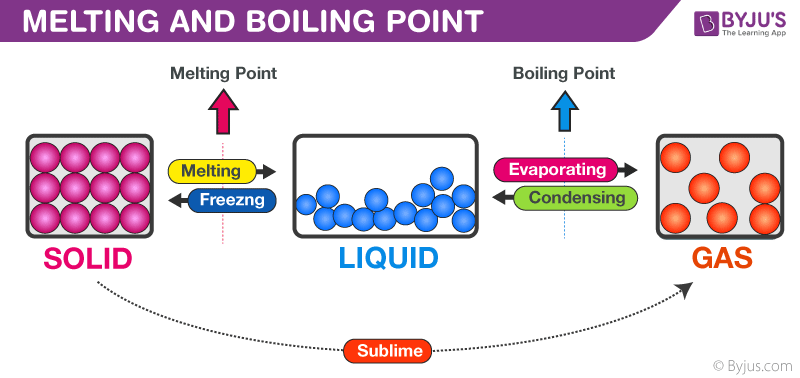
Image explaining melting and boiling points.
Image Credits: BYJU'S
- Solubility: "Like dissolves like" is the guiding rule. Polar functional groups (, ) increase solubility in polar solvents like water because they can interact with water molecules through hydrogen bonding or dipole-dipole forces. Nonpolar groups (, long hydrocarbon chains) make compounds more soluble in nonpolar solvents like hexane. As the carbon chain gets longer, even an alcohol becomes less water-soluble because the nonpolar portion dominates.
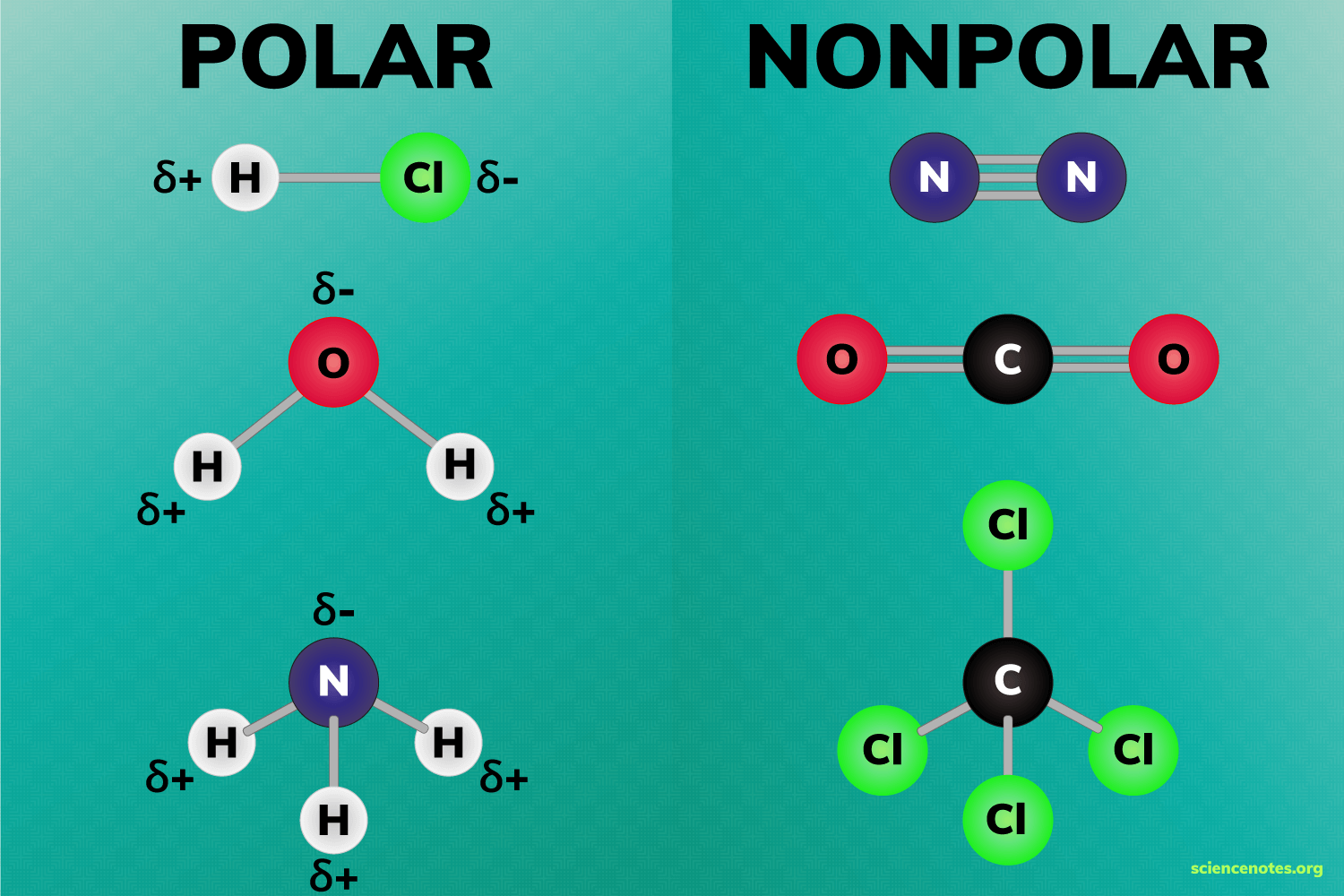
Image displaying the difference between polar and non-polar molecules.
Image Credits: Science Notes
- Optical Activity: Some organic molecules have chiral centers, which are carbon atoms bonded to four different groups. A chiral molecule and its mirror image (called enantiomers) are non-superimposable, much like your left and right hands. Enantiomers rotate plane-polarized light in opposite directions, which is what we measure as optical activity.
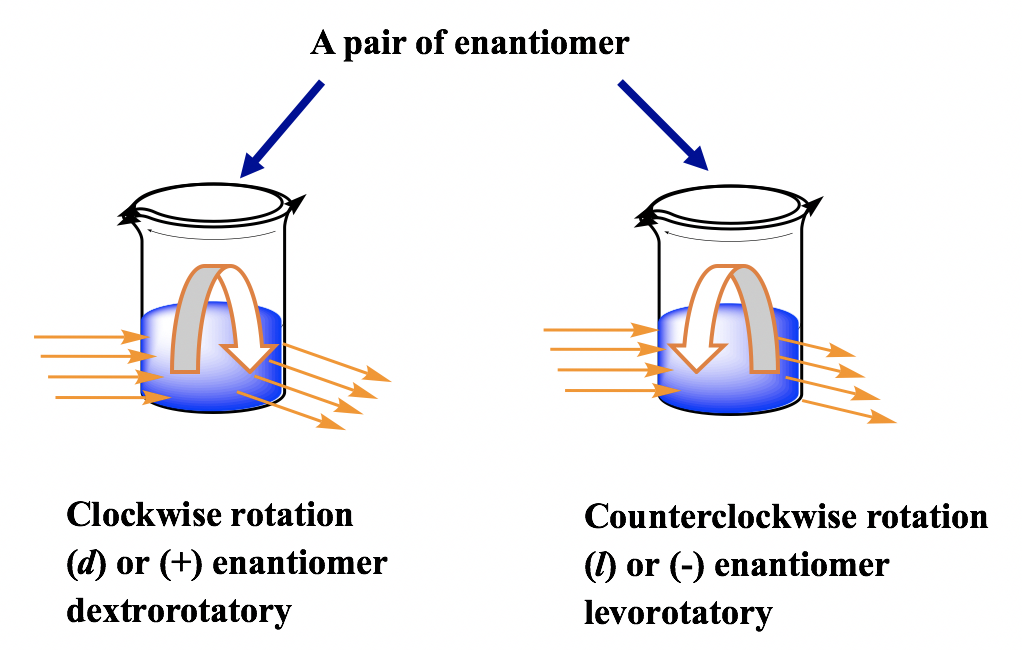
Image showing optical activity within molecules.
Image Credits: Kwantlen Polytechnic University
Chemical Properties
Chemical properties describe how organic compounds react and transform.
- Acidity/Basicity: Functional groups determine whether a compound acts as an acid or a base. Carboxylic acids () are acidic because they can donate the from the portion; the resulting conjugate base is stabilized by resonance across the two oxygens. Amines () are basic because the lone pair on nitrogen can accept a proton.
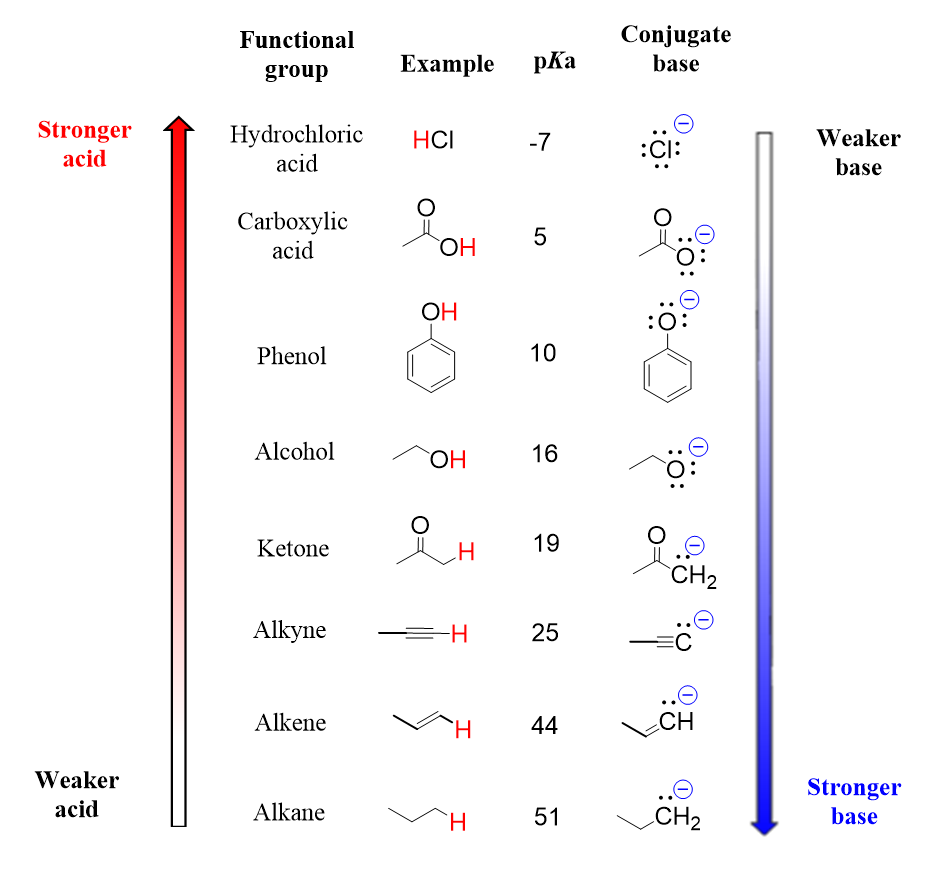
Image displaying the acidity/basicity of various functional groups.
Image Credits: Chemistry Steps
- Stability: Compounds with only single bonds (alkanes) tend to be the most chemically stable because there are no high-energy bonds or reactive sites. Double bonds () and triple bonds () store more energy and are more reactive, making alkenes and alkynes less stable in the presence of reagents that can break those bonds. Resonance stabilization, as seen in aromatic rings, also increases stability.
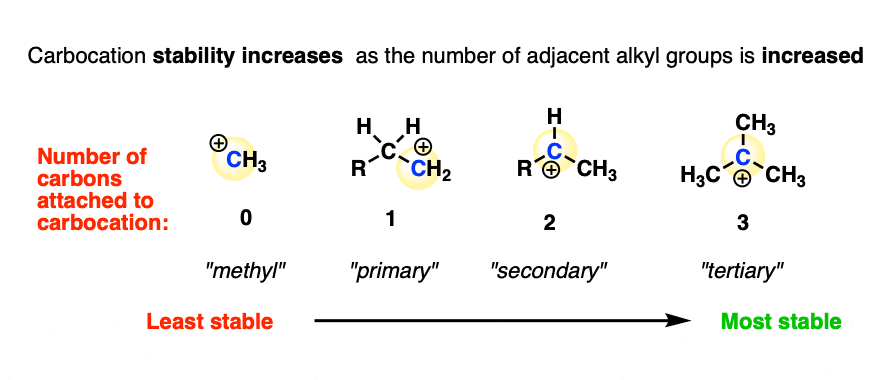
Image explaining stability of organic compounds.
Image Credits: Master Organic Chemistry
Biological Activity
Organic functional groups are central to how molecules behave in living systems, which connects organic chemistry to medicine and biochemistry.
- Pharmacological Properties: The functional groups on a drug molecule determine how it binds to biological targets (like enzymes or receptors), how it's absorbed, and how quickly it's metabolized. For example, adding a hydroxyl group to a drug can increase its water solubility and change how it's distributed in the body.
- Metabolic Pathways: Functional group transformations drive metabolism. Oxidation of alcohols to aldehydes, reduction of ketones, and hydrolysis of esters are all reactions that occur constantly in your cells during the synthesis and breakdown of biomolecules.
Practice Problems
-
Name the functional group in . How does it affect boiling point?
- Functional group: Hydroxyl group (), making this an alcohol.
- Effect: The group can form hydrogen bonds with neighboring molecules. Hydrogen bonds are stronger than the London dispersion forces found in a similarly sized alkane, so the boiling point is significantly higher. Compare ethanol (bp 78°C) to ethane (bp −89°C).
-
Name the functional group in . How does it impact acidity or basicity?
- Functional group: Carbonyl group (), specifically a ketone (the is between two carbon groups).
- Effect: Ketones are neither strongly acidic nor strongly basic. The carbonyl is polar, but there's no easily donated (unlike a carboxylic acid) and no lone pair as available as in an amine. The compound is roughly neutral.
-
Identify the functional group that typically acts as an acid in organic compounds and provide an example.
- Functional group: Carboxyl group ()
- Example: Acetic acid ()
- Explanation: The group donates its relatively easily. After losing the proton, the negative charge is stabilized by resonance between the two oxygen atoms, which is why carboxylic acids are much more acidic than alcohols.