Oxidation and Reduction
Oxidation and reduction describe how electrons transfer between atoms during chemical reactions. These concepts are the foundation of electrochemistry, and you'll need a solid grasp of them to understand batteries, corrosion, and many biological processes covered later in this unit.

🔄 Understanding Oxidation and Reduction
🔍 Defining Oxidation and Reduction
Every redox reaction involves one species losing electrons and another gaining them. These two processes always happen together.
- Oxidation: An atom loses electrons. Its oxidation state increases (becomes more positive).
- Reduction: An atom gains electrons. Its oxidation state decreases (becomes more negative).
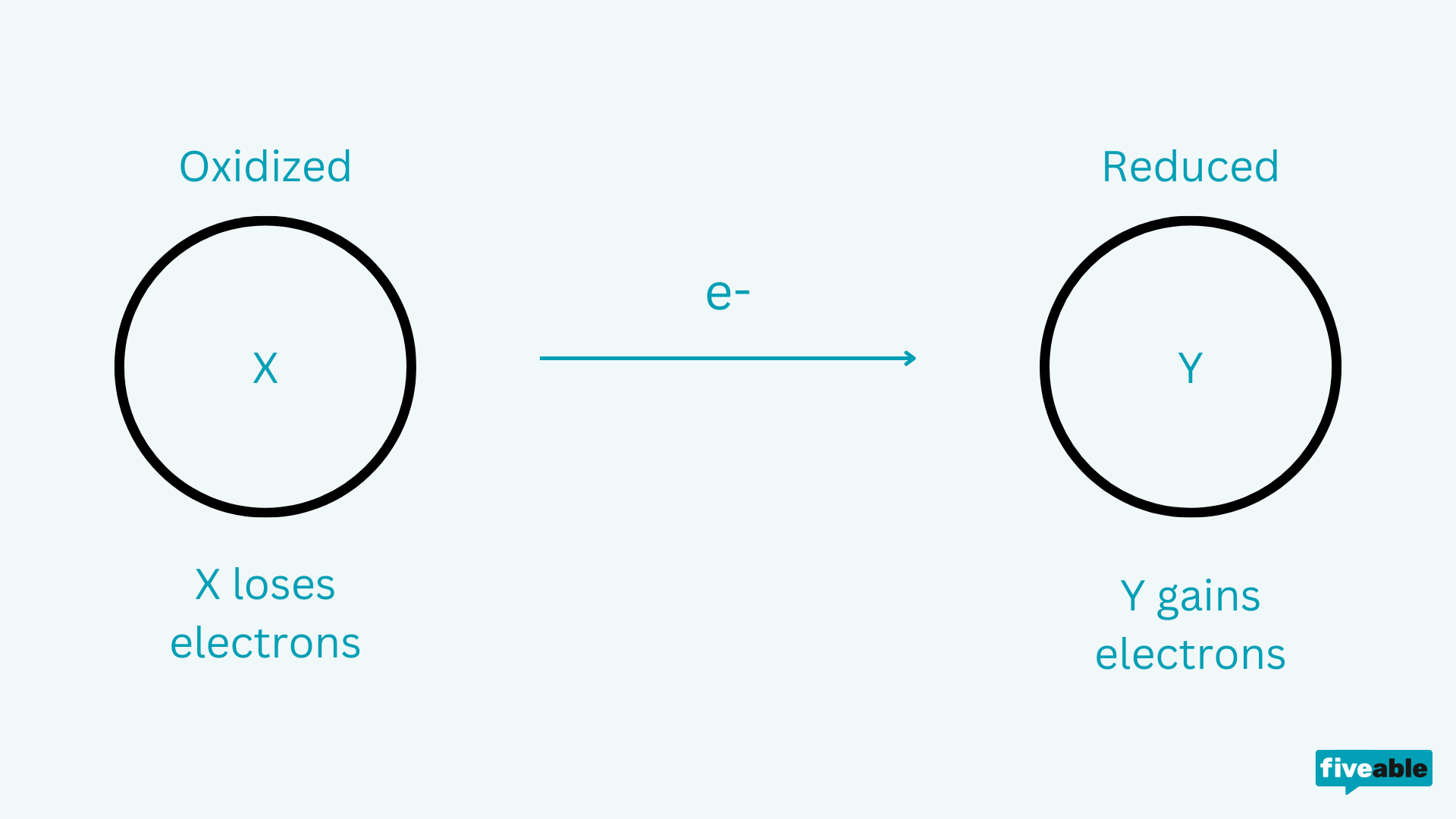
Image representing oxidation and reduction.
Image courtesy of Vibhi
- The classic mnemonic here is OIL RIG:
- OIL: Oxidation Is Losing (electrons)
- RIG: Reduction Is Gaining (electrons)
⚖️ Changes in Oxidation State
An oxidation state (or oxidation number) is a bookkeeping tool that tracks how many electrons an atom has gained or lost relative to its elemental form. Tracking these numbers is how you identify what's being oxidized and what's being reduced.
Assigning Oxidation Numbers: Follow these rules in order of priority:
- Any element in its pure, uncombined form has an oxidation state of 0 (e.g., , , ).
- Monatomic ions have an oxidation state equal to their charge (e.g., = +1, = −1).
- Oxygen is almost always −2 (exception: peroxides like , where it's −1).
- Hydrogen is +1 when bonded to nonmetals and −1 when bonded to metals (metal hydrides).
- Fluorine is always −1.
- The sum of all oxidation states in a neutral compound equals 0; in a polyatomic ion, it equals the ion's charge.
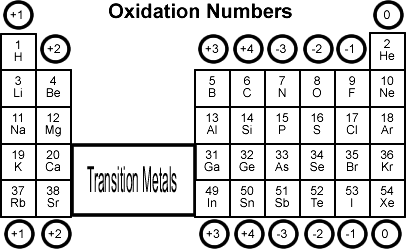
Image representing the periodic table and the oxidation numbers for each group.
Image courtesy of Socratic
Indicating Redox: If an atom's oxidation state increases from reactants to products, it was oxidized. If it decreases, it was reduced. If no oxidation states change, the reaction isn't a redox reaction.
🔎 Identifying Agents in Reactions
Every redox reaction has two key players: the oxidizing agent and the reducing agent. This is the part that trips people up, because the names seem backwards at first.
🦸♂️ Oxidizing Agents
The oxidizing agent is the species that gets reduced itself. It causes oxidation in another species by accepting electrons from it. Common oxidizing agents include , halogens like and , and permanganate ion ().
🦸♀️ Reducing Agents
The reducing agent is the species that gets oxidized itself. It causes reduction in another species by donating electrons to it. Active metals like , , and are strong reducing agents because they lose electrons easily. Molecular hydrogen () can also act as a reducing agent.
📝 Quick trick: find which atom's oxidation number went up. That atom (or the species containing it) is the reducing agent. The atom whose oxidation number went down is part of the oxidizing agent.
Note: was listed as an oxidizing agent in some references, but it most commonly acts as a reducing agent. It can act as an oxidizing agent only with very active metals (forming metal hydrides), which is a less common scenario.
⚡ Role of Electron Transfer in Redox Reactions
Electron transfer is what makes a reaction "redox." There are two useful ways to look at it:
- Direct Transfer: One atom physically passes electrons to another. For example, when a strip of zinc metal is placed in a solution, zinc atoms hand electrons directly to copper ions.
- Half-Reactions: You can split any redox reaction into two parts. The oxidation half-reaction shows only the electron loss, and the reduction half-reaction shows only the electron gain. This makes it much easier to see what's happening and is essential for balancing.
For the zinc-copper example:
- Oxidation half-reaction:
- Reduction half-reaction:
🧮 Balancing Redox Reactions
Redox equations must be balanced for both atoms and charge. Two main methods:
1. The Half-Reaction Method:
- Separate the reaction into oxidation and reduction half-reactions.
- Balance all atoms other than O and H in each half-reaction.
- Balance O by adding ; balance H by adding (in acidic solution) or (in basic solution).
- Balance charge by adding electrons to the appropriate side.
- Multiply each half-reaction by a factor so the electrons lost equal the electrons gained.
- Add the two half-reactions together and cancel anything that appears on both sides.
2. Oxidation Number Method:
- Assign oxidation numbers to every atom in the equation.
- Identify which atoms change oxidation state and by how much.
- Use coefficients to make the total increase in oxidation number equal the total decrease.
- Balance the remaining atoms by inspection.
🌐 Applications of Redox Reactions
Redox reactions show up everywhere:
- 🔋 Batteries and fuel cells: Batteries convert chemical energy to electrical energy through controlled redox reactions. Your phone battery relies on lithium ions shuttling electrons through a circuit.
- 🚿 Water and air treatment: Chlorination of drinking water is an oxidation reaction that destroys pathogens.
- 🌿 Biological systems: Cellular respiration oxidizes glucose to produce ATP, and photosynthesis reduces to build sugars. Both are redox processes.
- 🏭 Industrial processes: Extracting metals from ores (smelting) involves reducing metal oxides. Corrosion of iron (rusting) is oxidation you want to prevent.
💡 Redox Practice Question
Consider the reaction:
a) Determine the oxidation state of each element in both the reactants and products.
b) Identify the species being oxidized and the species being reduced.
c) Name the oxidizing agent and the reducing agent in the reaction.
✏️ Practice Question Solution
a) Oxidation states:
- Reactants:
- Zn: 0 (elemental form)
- Cu in : +2
- S in : +6
- O in : −2
- Products:
- Zn in : +2
- S in : +6
- O in : −2
- Cu: 0 (elemental form)
b) Species being oxidized and reduced:
- Oxidized: Zn goes from 0 to +2 (oxidation state increased, meaning it lost electrons).
- Reduced: Cu goes from +2 to 0 (oxidation state decreased, meaning it gained electrons).
Notice that S and O don't change oxidation states at all. The sulfate ion () is just a spectator here.
c) Oxidizing agent and reducing agent:
- Oxidizing Agent: (from ). It gets reduced by accepting electrons from zinc.
- Reducing Agent: . It gets oxidized by donating electrons to .
Remember: the oxidizing agent is the one that gets reduced, and the reducing agent is the one that gets oxidized. It feels counterintuitive at first, but it clicks once you think about it from the perspective of what each species causes to happen to the other.