Molecular Polarity
Molecular polarity describes how charge is distributed unevenly across a molecule. This single property explains a huge range of chemical behavior, from why water dissolves salt to why oil and water separate. To predict and understand polarity, you need to connect three ideas: electronegativity, bond polarity, and molecular geometry.

⚛️ Understanding Molecular Polarity
What is Polarity?
A molecule is polar when electrons aren't shared equally among its atoms, creating regions of partial positive and partial negative charge. You can think of a polar molecule as having "poles," similar to a magnet: one end is slightly negative, the other slightly positive.
This unequal sharing happens because different atoms have different electronegativities. The more electronegative atom pulls bonding electrons closer to itself, gaining a partial negative charge (written as δ-), while the other atom becomes partially positive (δ+).
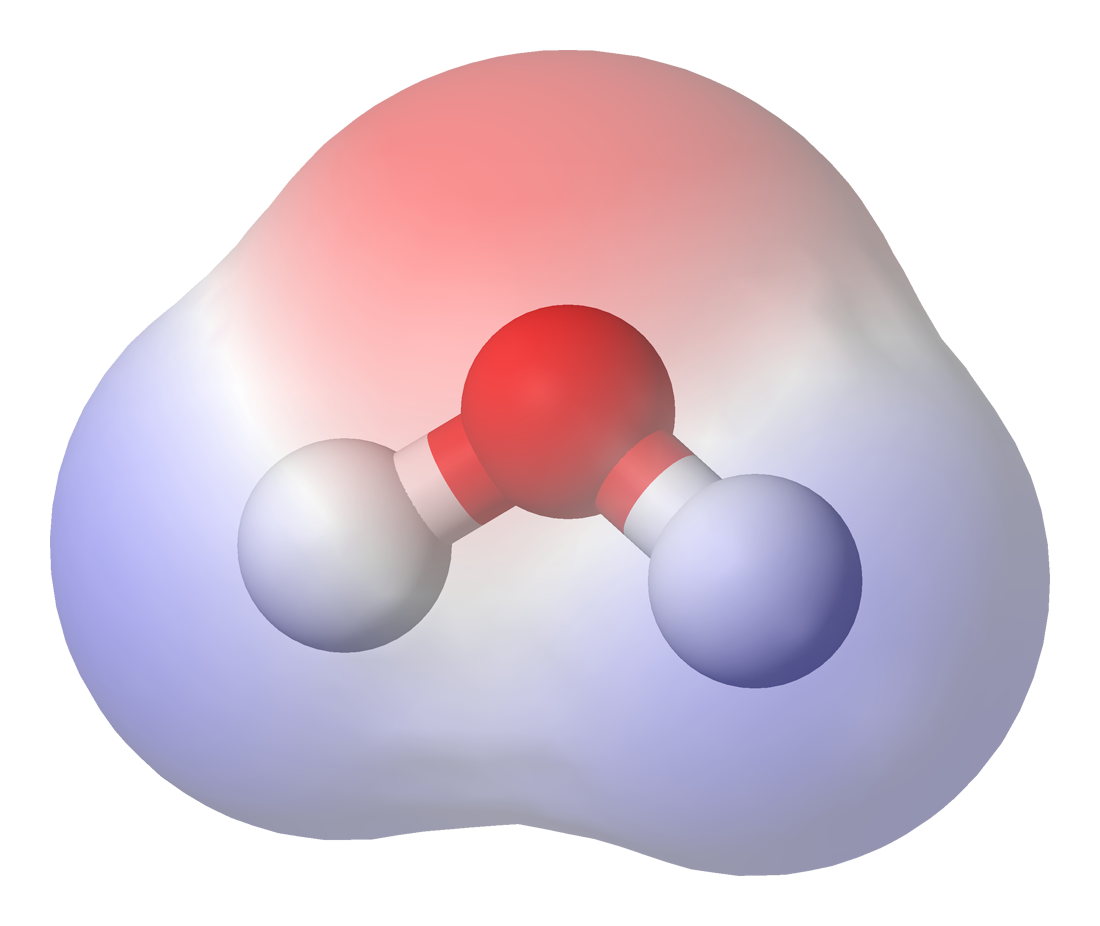
Chemical structure of water molecule.
Image Courtesy of Wikipedia
Water is the classic example. Oxygen has a higher electronegativity than hydrogen (you don't need to memorize exact values, but know the trend). Because oxygen pulls the shared electrons more strongly, it carries a δ- charge, and each hydrogen carries a δ+ charge.
Dipole Moments
A dipole moment is the quantitative measure of polarity in a bond or molecule. It's represented as an arrow pointing from the positive end toward the negative end, and it has both direction and magnitude.
The magnitude of a dipole moment is measured in Debye units (D). Two factors increase a dipole moment:
- A larger electronegativity difference between the bonded atoms
- A greater distance between the partial charges (longer bond length)
📍 The dipole moment arrow (→) has a + sign at its tail (the positive pole) and points toward the negative pole. In water, each O–H bond has its own dipole moment arrow pointing toward oxygen.
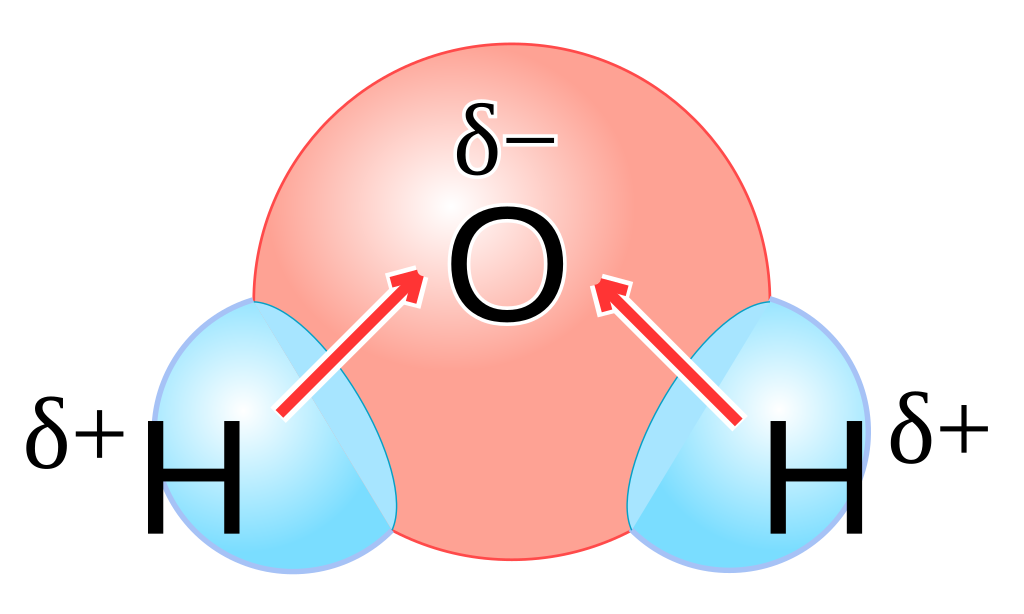
Dipole moments in a water molecule.
Image Courtesy of Wikimedia Commons
Polar vs Nonpolar Molecules
Molecules are classified as polar or nonpolar based on whether their overall electron distribution is uneven or even. This is one of the most tested concepts in this unit.
-
Nonpolar molecule: Electrons are shared equally (or the dipole moments cancel out due to symmetry). Atoms either have similar electronegativities, or the molecular geometry is symmetrical enough that individual bond dipoles point in opposite directions and cancel.
- Diatomic elements like and are nonpolar because identical atoms have the same electronegativity.
- has polar bonds, but its linear shape causes the two bond dipoles to cancel perfectly, making the overall molecule nonpolar.
-
Polar molecule: Electrons are shared unequally and the molecular geometry doesn't allow the dipole moments to cancel. The molecule has a net dipole moment.
- is polar because its bent shape prevents the two O–H bond dipoles from canceling.
This distinction between bond polarity and molecular polarity is critical. A molecule can contain polar bonds and still be nonpolar overall if its geometry is symmetrical.
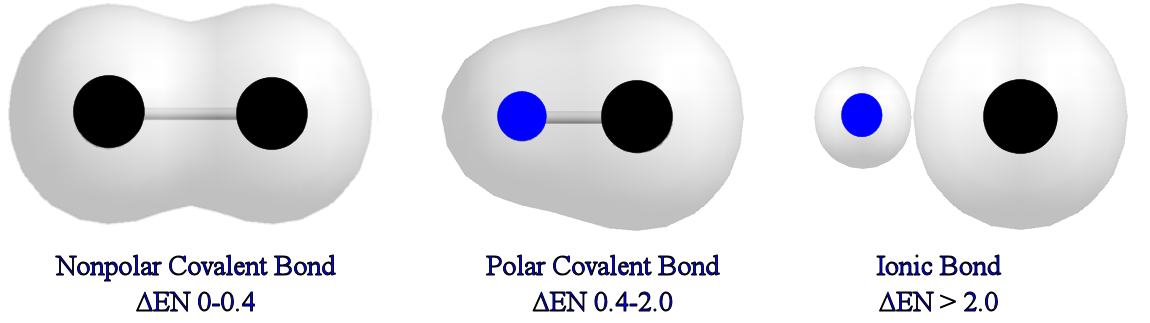
Diagram comparing polar and nonpolar molecules with electron clouds.
Image Courtesy of Angelo State
🔍 Determining Molecular Polarity
Predicting whether a molecule is polar or nonpolar comes down to two steps: check the bond polarities, then check the shape.
Step 1: Evaluate Electronegativity Differences
Look at each bond in the molecule. The greater the electronegativity difference between two bonded atoms, the more polar that bond is.
- If the bonded atoms are identical (like in ), the bond is nonpolar.
- If there's a small to moderate difference (like C–H), the bond is slightly polar.
- If there's a large difference (like O–H or N–H), the bond is significantly polar.
Step 2: Consider Molecular Geometry
Even if a molecule has polar bonds, the overall molecule might still be nonpolar if its shape is symmetrical enough for the dipole moments to cancel. This is where VSEPR theory comes in: it predicts molecular shapes based on electron pair repulsion.
📍 Common Shapes to Know:
- Linear: 180° bond angle, straight line
- Bent: Bond angle less than 180°, angled shape
- Trigonal planar: Three bonds in a flat plane, 120° apart
- Tetrahedral: Four bonds arranged in 3D, 109.5° apart
To review VSEPR theory in detail, check out the previous guide.
The key question is: does the molecule's shape allow the bond dipoles to cancel?
- Symmetrical shapes (linear with identical outer atoms, trigonal planar with identical outer atoms, tetrahedral with identical outer atoms) → dipoles cancel → nonpolar
- Asymmetrical shapes (bent, trigonal pyramidal, or any shape with lone pairs on the central atom or different outer atoms) → dipoles don't cancel → polar
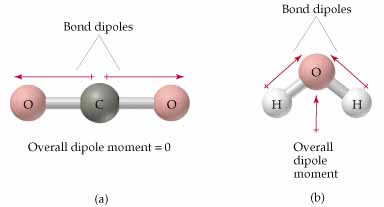
Linear versus bent molecule and their corresponding polarity.
Image Courtesy of Kent Chemistry
In this image, molecule (a) is . It's linear, so the two C=O bond dipoles point in exactly opposite directions and cancel. The molecule is nonpolar overall, even though each C=O bond is polar.
Molecule (b) is . Its bent shape means the two O–H bond dipoles point in roughly the same direction (both toward oxygen). They don't cancel, so water is polar.
💧 Polarity: Effects on Physical Properties
Polarity directly affects how substances behave, particularly their boiling points and solubility.
Boiling Points & Melting Points
Polar molecules generally have higher boiling and melting points than nonpolar molecules of similar size. This is because polar molecules experience stronger intermolecular forces (the attractions between molecules), which require more energy to overcome. The next study guide covers intermolecular forces in detail, but for now, remember the trend: greater polarity → stronger attractions → higher boiling/melting points.
Solubility
The guiding principle here is "like dissolves like."
- Polar solvents dissolve polar solutes. Water (polar) dissolves table salt and sugar well.
- Nonpolar solvents dissolve nonpolar solutes. Oil (nonpolar) dissolves wax and grease easily.
- Polar and nonpolar substances don't mix well with each other. That's why oil and water separate.
📍 Soap molecules have a dual structure: one end is hydrophilic (attracted to water, polar) and the other end is hydrophobic (attracted to oils/grease, nonpolar). This lets soap bridge the gap between water and oily grime, which is why it's effective at cleaning.
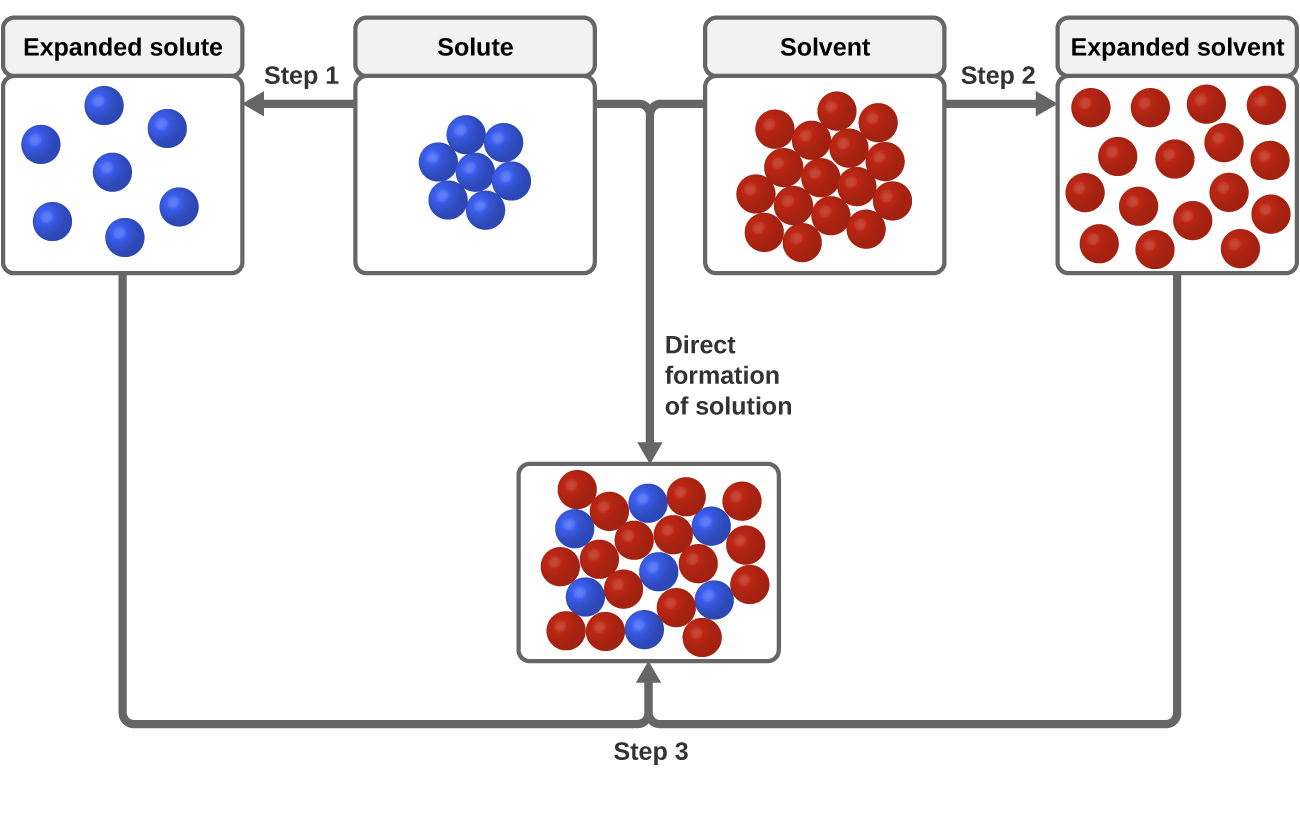
Image depicting dissolution process for both polar and nonpolar substances.
Image Courtesy of Chemistry LibreTexts
🚀 Additional Effects of Molecular Polarity
Polarity influences chemistry well beyond boiling points and solubility.
Chemical Reactivity & Interaction:
- Polarity affects the type of bonding that forms. Atoms with very large electronegativity differences tend to form ionic bonds (electron transfer), while atoms with moderate differences form polar covalent bonds (unequal sharing). The degree of polarity in a bond influences how that molecule reacts with other substances.
Biological Systems:
- Cell membranes are built from molecules with both polar and nonpolar regions. The nonpolar interior of the membrane acts as a barrier, allowing the cell to control which polar and nonpolar molecules pass through. Protein folding also depends on polarity: polar amino acids orient toward water, while nonpolar ones tuck inward.
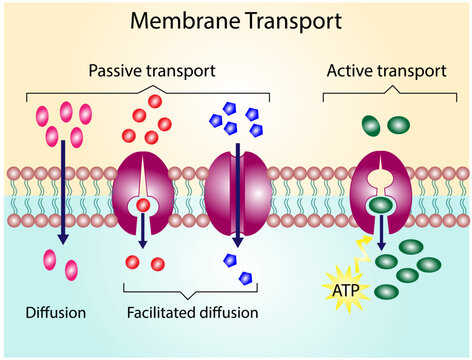
Diagram showing membrane transport.
Image Courtesy of Adobe Stock
Separation Techniques:
- Chromatography exploits polarity differences to separate mixtures. In a typical setup, polar molecules interact more strongly with a polar stationary phase and move slowly, while nonpolar molecules travel faster. By choosing solvents and surfaces of different polarities, scientists can isolate specific compounds from a mixture.
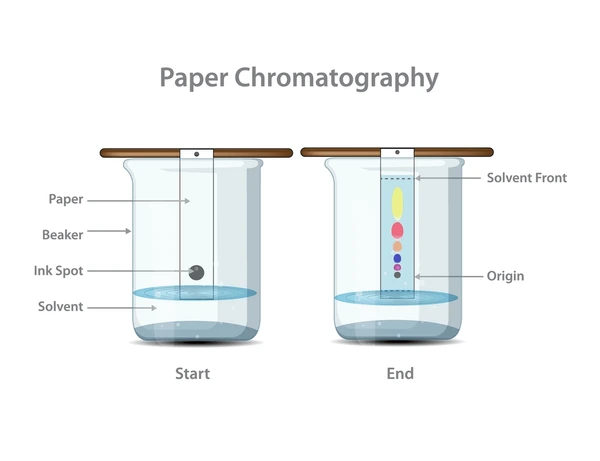
Image of how chromatography works.
Image Courtesy of Shutterstock
🌟 Wrapping Up
The core skill for this topic is connecting three ideas: electronegativity differences create polar bonds, molecular geometry determines whether those bond dipoles cancel, and the resulting molecular polarity drives physical properties like boiling point and solubility.
A good test of your understanding: Why does soap clean greasy stains? Soap's nonpolar end attaches to the grease, while its polar end is pulled into the surrounding water. This lets water carry the grease away, a direct application of "like dissolves like" and the dual polar/nonpolar nature of soap molecules.