KMT and Gas Laws
This section covers the Kinetic Molecular Theory and the foundational gas laws that describe how gases respond to changes in pressure, volume, and temperature. These laws show up constantly in chemistry, so understanding both the math and the reasoning behind them is essential.

Kinetic Molecular Theory (KMT)
Gas laws are rooted in the Kinetic Molecular Theory (KMT), which describes gases as large numbers of tiny particles in constant, random motion. KMT gives us a framework to predict and explain how gases respond to changes in pressure, volume, and temperature.
The KMT is based on five fundamental assumptions:
- Gas particles are in constant, random, straight-line motion.
- Particles are separated by great distances relative to their size, so a gas is mostly empty space.
- Collisions between particles are elastic, meaning no kinetic energy is lost during a collision. The total kinetic energy of the system stays constant.
- There are no attractive or repulsive forces between gas particles.
- The average kinetic energy of gas particles is directly proportional to the gas's temperature (in Kelvin).
When a gas perfectly follows all five assumptions, we call it an ideal gas. No real gas is truly ideal, but many gases behave close to ideally under normal conditions. Real gases deviate from ideal behavior most noticeably at high pressures (particles get forced close together, so intermolecular forces matter) and low temperatures (particles slow down enough for those forces to have an effect).
Fundamental Gas Laws
These laws describe how pressure, volume, and temperature relate to each other for a fixed amount of gas.
Boyle's Law
Boyle's Law describes the relationship between the pressure and volume of a gas when temperature is held constant.
📍
- and are the initial pressure and volume.
- and are the new pressure and volume after change.
Pressure and volume are inversely proportional. As volume decreases, pressure increases, and vice versa, as long as temperature stays the same.
Think about breathing. When your diaphragm contracts and your chest cavity expands, the volume inside your lungs increases. That increase in volume causes a drop in pressure, and air rushes in from the higher-pressure atmosphere outside.
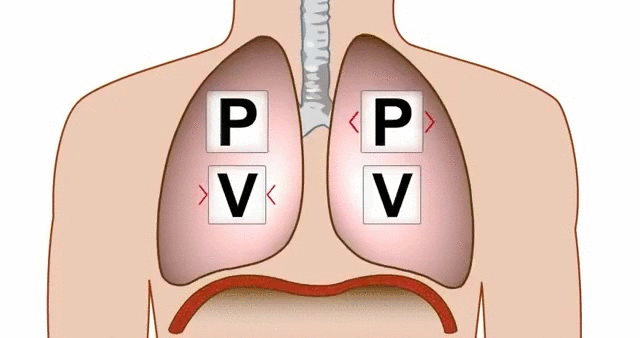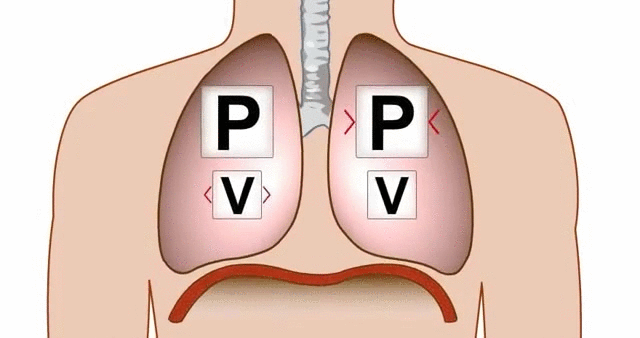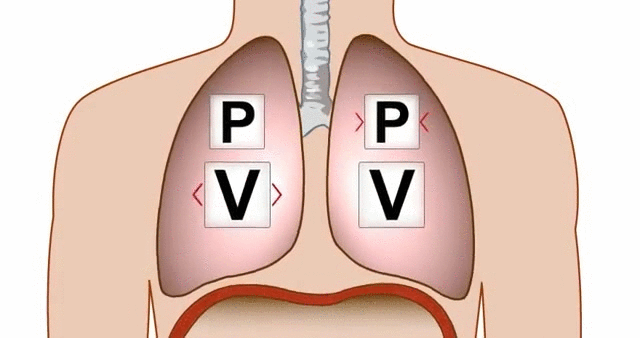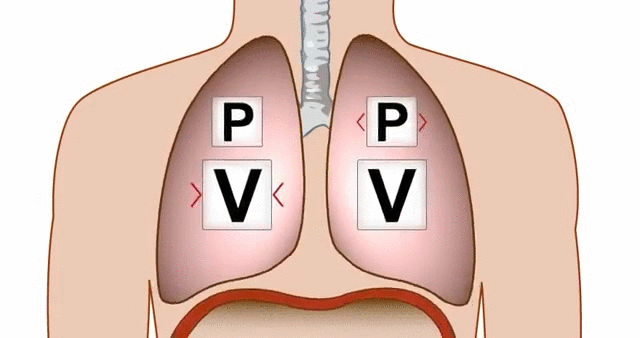
GIF of the lungs expanding and contracting, demonstrating the relationship between pressure and volume.
GIF Courtesy of Alchaeus via Imgur.
✏️ Boyle's Law Practice Question
A balloon has a volume of 2.0 liters at a pressure of 1.0 atm. If the balloon is compressed to a volume of 1.5 liters, what will be the new pressure inside the balloon, assuming the temperature remains constant?
Identify your variables: , , , and is unknown.
Volume went down, so pressure went up. That's consistent with the inverse relationship.
🧠 Boyle's Law Conceptual Question
A sealed syringe contains air and is plunged into a container of water at room temperature. As the plunger is pushed in, reducing the volume of air by half, what happens to the pressure inside the syringe? Explain your answer using Boyle's Law.
The volume is halved, and you need to figure out what happens to pressure. Since pressure and volume are inversely proportional, cutting the volume in half means the pressure must double. You can see this algebraically by setting up Boyle's Law:
For both sides to remain equal, must be . When volume is halved, pressure doubles.
Charles' Law
Charles' Law describes the direct relationship between the volume and temperature of a gas at constant pressure. When temperature goes up, volume goes up. When temperature goes down, volume goes down.
📍
- and represent volume.
- and represent temperature (must be in Kelvin).
</>Code- $$0 \degree C + 273 = 273 \ K$$
Why does this happen? Connect it back to KMT assumption #5: as temperature increases, particles gain kinetic energy and move faster. Faster-moving particles spread farther apart, so the gas occupies more volume (assuming the container can expand).
Hot air balloons are a classic example. Heating the air inside the balloon increases its volume, making the air inside less dense than the cooler air outside, and the balloon rises.
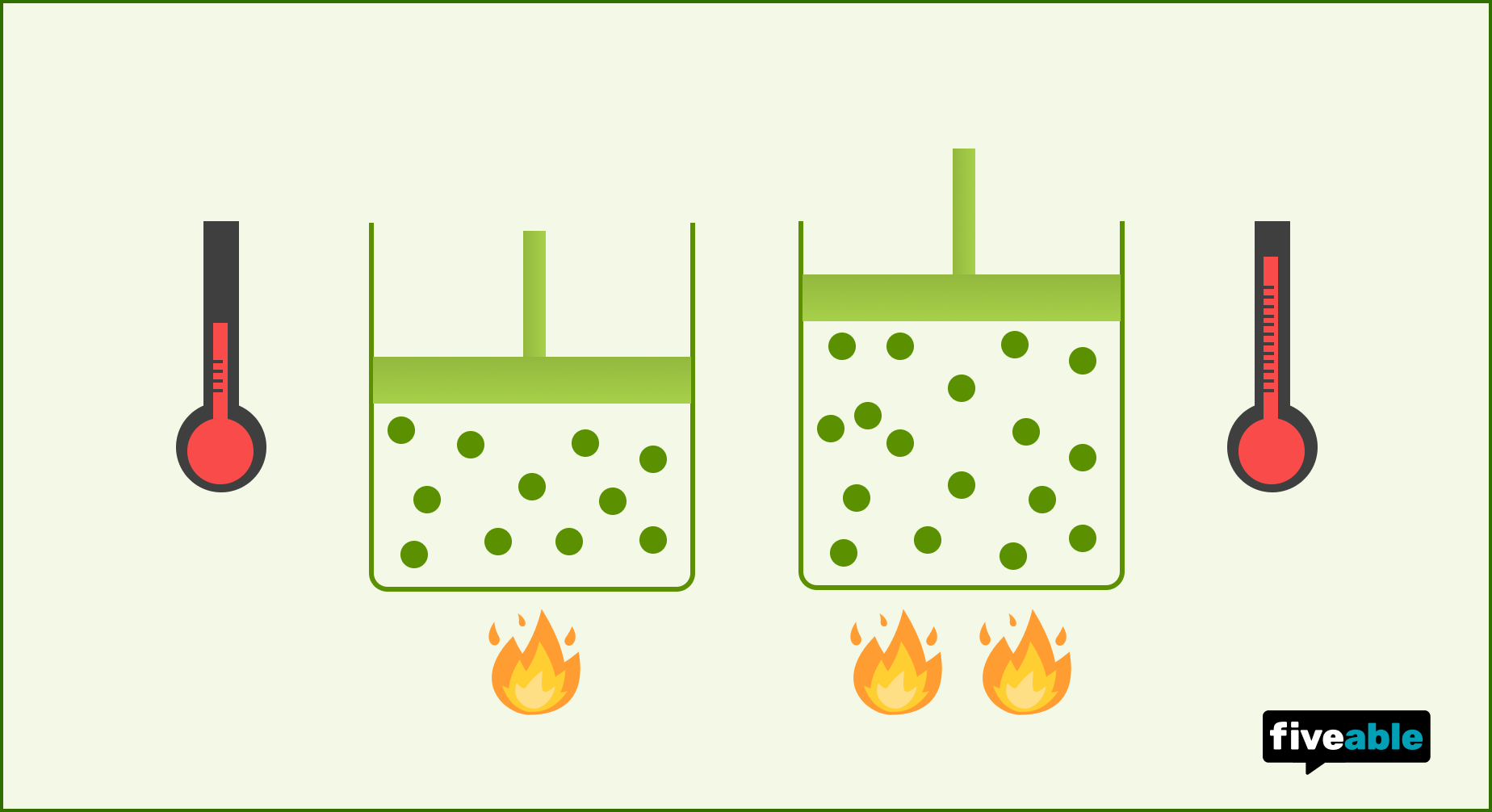
Image displaying the relationship between volume of a gas and temperature of a gas.
Image Credit to Fiveable.
✏️ Charles' Law Practice Question
A yellow balloon inflated in a room at has a volume of . The balloon is then heated to a temperature of . What is the new volume if the pressure remains constant?
First, convert both temperatures to Kelvin:
Write out Charles' Law and plug in values:
Rearrange and solve:
Temperature increased, and volume increased along with it. That confirms the direct relationship.
Gay-Lussac's Law
Gay-Lussac's Law describes the direct relationship between pressure and temperature when volume is held constant. Temperature up, pressure up. Temperature down, pressure down.
📍
- and represent pressure.
- and represent temperature (in Kelvin).
Here's the reasoning:
- Volume is constant, so the gas is in a rigid container that can't expand or contract.
- As temperature rises, particles move faster (KMT assumption #5) and collide with the container walls more frequently and with greater force.
- More forceful, more frequent collisions = higher pressure.
If temperature decreases, particles slow down, hit the walls less often and with less force, and pressure drops.
A practical example: aerosol cans (like hair spray) carry warnings not to expose them to heat. The can is rigid, so if temperature rises, pressure builds with no way to relieve it. Enough heat and the can could burst. Compare that to a balloon, where the flexible walls allow volume to change, preventing dangerous pressure buildup. That difference is exactly why Gay-Lussac's Law specifies constant volume.
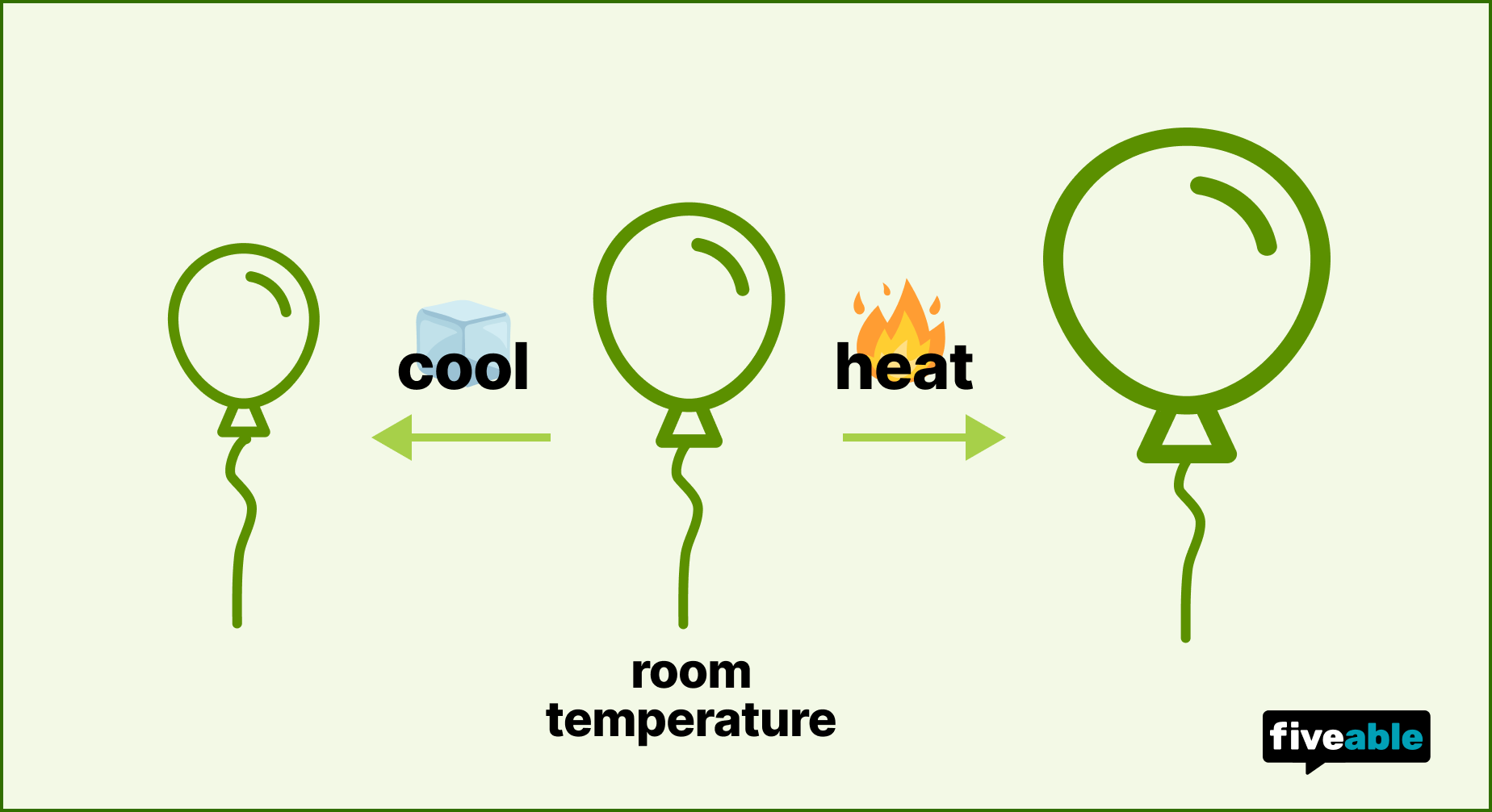
Image showing the volume-temperature relationship for gases.
Image Credit to Fiveable.
✏️ Gay-Lussac's Law Practice Question
The pressure in a car tire is at . After a trip, the tire pressure rises to . Assuming the volume is constant, what is the temperature of the air in the tire in ?
Write out Gay-Lussac's Law:
Convert the given temperature to Kelvin:
Assign values: , , , and solve for .
The question asks for Celsius, so convert back:
Combined Gas Law
The Combined Gas Law merges Boyle's, Charles's, and Gay-Lussac's laws into one equation. You can actually derive any of the three individual laws from it by holding one variable constant. It applies when the amount of gas (number of moles) stays the same but pressure, volume, and temperature all change.
To see how this works: if temperature is constant (), the temperatures cancel and you're left with Boyle's Law. If pressure is constant, you get Charles' Law. If volume is constant, you get Gay-Lussac's Law.
✏️ Combined Gas Law Practice Question
A weather balloon is inflated to a volume of at a pressure of and a temperature of . The balloon rises to an altitude where the pressure is and the temperature is . Assuming the balloon can freely expand, what is its new volume?
Write out the Combined Gas Law:
Convert temperatures to Kelvin:
Plug in all known values:
Rearrange and solve for :
The balloon's volume more than doubled, which makes sense: pressure dropped significantly (from 850 to 325 mmHg), and even though temperature also dropped, the pressure decrease had a much larger effect.
Steps for Solving Gas Law Problems
Here's a reliable process for any gas law calculation:
-
List what you're given. Write down every value with its units.
-
Assign variables. Identify which values are initial conditions (, , ) and which are final conditions (, , ). Identify your unknown.
- If any temperatures are in Celsius, convert to Kelvin now.
-
Choose the right equation. Figure out which variable is constant (or if none are, use the Combined Gas Law).
-
Rearrange to isolate the unknown. Get your unknown variable alone on one side before plugging in numbers.
-
Substitute and solve. Plug in values with units, cancel units, and calculate.
-
Check your answer. Does the direction of change make sense? (e.g., if you decreased volume, did pressure increase?) Report with correct significant figures and units.
Avogadro's Law
Avogadro's Law states that equal volumes of gases at the same temperature and pressure contain equal numbers of molecules. Put another way, volume is directly proportional to the number of moles of gas, as long as temperature and pressure stay constant.
📍
- and represent volume.
- and represent the number of moles of gas.
If you add more gas to a flexible container (at constant and ), the volume increases proportionally. Double the moles, double the volume. This relationship is a key building block for the Ideal Gas Law, which combines all of these relationships into a single equation.