Properties of Acids and Bases
Acids and bases are two fundamental categories of chemical substances, and understanding their properties is essential for nearly every topic that follows in chemistry. This section covers how to identify acids and bases, the major definitions chemists use, the pH scale, and the difference between strong and weak varieties.

General Properties of Acids
Acids share several observable properties that help you identify them in the lab:
- Sour taste: Citric acid gives lemons their tartness, and phosphoric acid contributes to the sharp bite of cola. (Obviously, never taste chemicals in a lab setting.)
- Litmus test: Acids turn blue litmus paper red.
- Reaction with metals: Acids react with certain active metals (like zinc or magnesium) to produce hydrogen gas (). The bubbles you see when a metal dissolves in acid are escaping.
- Neutralization: When mixed with a base, an acid produces a salt and water.
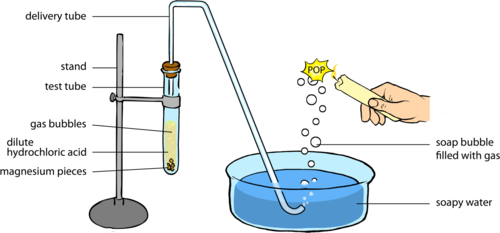
Image of a piece of metal reacting in acid, producing bubbles.
Image Courtesy of Siyavula
Brønsted-Lowry & Lewis Definitions of Acids
There are two major theoretical definitions you need to know:
- Brønsted-Lowry: An acid is a proton donor. It donates an ion to another substance. For example, when dissolves in water, it donates its to a water molecule.
- Lewis: An acid is an electron pair acceptor. This is a broader definition that includes substances without hydrogen, like , which can accept a lone pair from another molecule.
The Brønsted-Lowry definition covers most of what you'll encounter in this course, but the Lewis definition extends the concept to reactions that don't involve transfer at all.
General Properties of Bases
Bases have their own set of characteristic properties:
- Bitter taste and slippery feel: Soap feels slippery because it's basic. (Again, don't taste or touch unknown chemicals.)
- Litmus test: Bases turn red litmus paper blue.
- Neutralization: Bases react with acids to form water () and a salt, just like the reverse of the acid reaction above.
Quick demo to visualize neutralization: Mixing vinegar (acetic acid) with baking soda (sodium bicarbonate, a base) produces carbon dioxide gas, water, and sodium acetate. The fizzing you see is escaping.
Brønsted-Lowry & Lewis Definitions of Bases
- Brønsted-Lowry: A base is a proton acceptor. It receives an ion. When ammonia () dissolves in water, it accepts an from water to form .
- Lewis: A base is an electron pair donor. It provides a lone pair of electrons to form a bond. qualifies here too, since the nitrogen lone pair is what grabs the proton.
pH Scale
The pH scale quantifies how acidic or basic a solution is, running from 0 to 14:
- pH < 7 → acidic
- pH = 7 → neutral (pure water at 25°C)
- pH > 7 → basic (alkaline)
Each whole number change on the pH scale represents a tenfold change in concentration. A solution with pH 3 is ten times more acidic than one with pH 4, and one hundred times more acidic than pH 5. This logarithmic relationship is something students often overlook, but it's critical for understanding why small pH shifts can have big chemical effects.
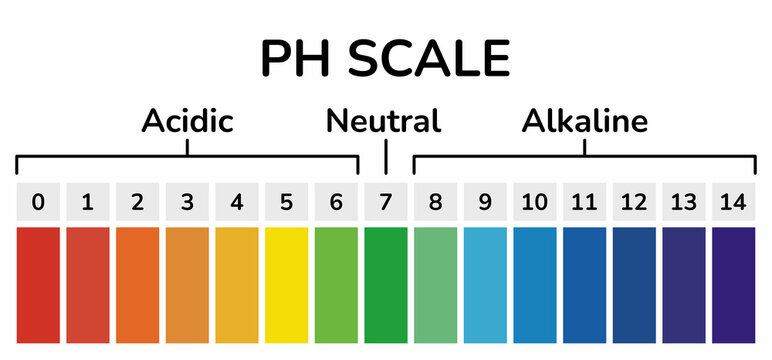
Image of a colorful pH scale showing the difference between acidic and basic.
Image Courtesy of Adobe Stock
Strong vs. Weak Acids/Bases
The distinction between strong and weak is about the degree of dissociation (or ionization) in water, not about concentration.
Strong Acids/Bases
Strong acids and bases dissociate completely in water. Every molecule breaks apart into ions.
- Strong acids you should memorize: , , , , , , and
- Strong bases are typically Group 1 and Group 2 metal hydroxides: , , ,
Because they fully dissociate, strong acids and bases produce the maximum number of ions in solution, which means they conduct electricity well and react rapidly.
Weak Acids/Bases
Weak acids and bases only partially dissociate in water. An equilibrium forms between the undissociated molecules and the ions.
- Acetic acid (, found in vinegar) is a common weak acid. In solution, only a small fraction of its molecules donate their .
- Ammonia () is a common weak base. It accepts a proton from water, but most molecules remain intact at any given moment.
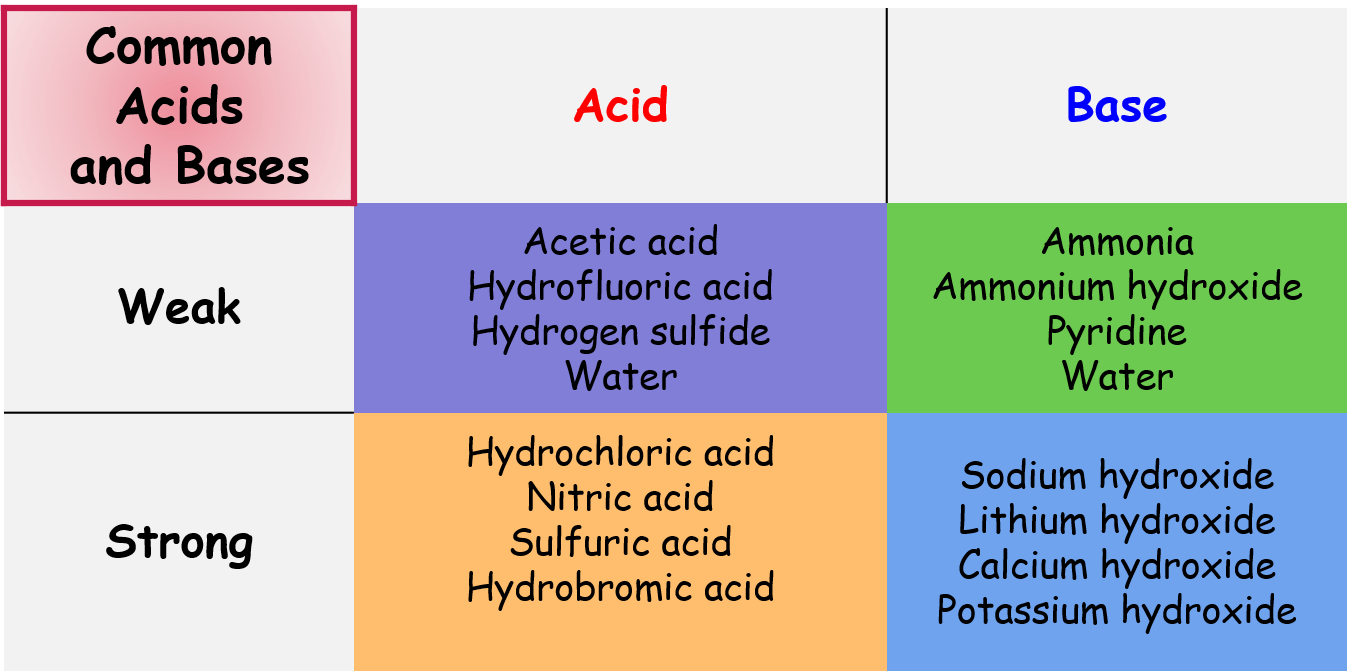
Image showing common strong and weak acids and bases.
Image Courtesy of Expii
Distinguishing Between Strength and Concentration
This is a common point of confusion: strength and concentration are not the same thing.
- Strength refers to what percentage of molecules dissociate into ions.
- Concentration refers to how many moles of solute are dissolved per liter of solution.
You can have a dilute strong acid (few molecules total, but all dissociated) or a concentrated weak acid (many molecules total, but only some dissociated).
Practice Question: What would be more corrosive: a strong acid at low concentration or a weak acid at high concentration?
Answer: It depends on the specific concentrations and the acid's dissociation constant, so there's no universal answer. A very concentrated weak acid can actually produce more ions in solution than a very dilute strong acid, making it more corrosive in that scenario. The key takeaway is that you need to consider both strength and concentration together.
Environmental & Biological Implications
Acids and bases matter well beyond the lab:
- Acid rain forms when sulfur dioxide () and nitrogen oxides () react with atmospheric water. The resulting sulfuric and nitric acids lower the pH of rain to around 4.2-4.4 (normal rain is about 5.6), which damages ecosystems by acidifying lakes and leaching nutrients from soil.
- Blood pH in humans is tightly regulated at about 7.35-7.45. Even small deviations outside this range can be life-threatening, which is why the body uses buffer systems (like the carbonic acid-bicarbonate buffer) to maintain stability.
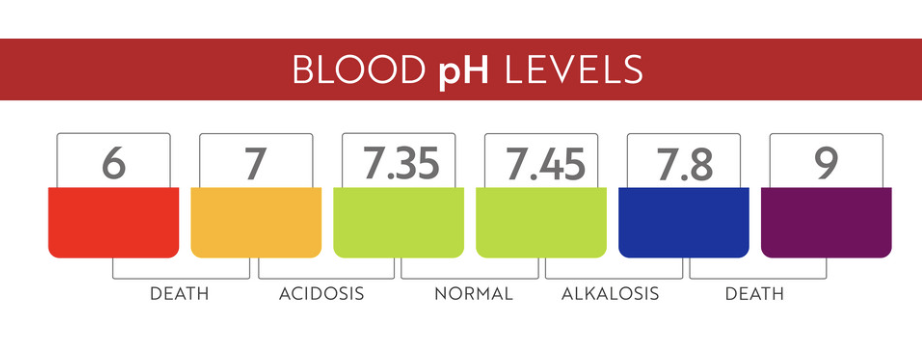
Image showing the regulation of blood pH.
Image Courtesy of VectorStock
Real-Life Application: Antacids like or work by neutralizing excess in your stomach, raising the pH and relieving heartburn. This is a straightforward neutralization reaction happening inside your body.