Electron Configurations
Electron configurations describe how electrons are arranged around an atom's nucleus. Since electrons drive chemical bonding and reactivity, knowing how they're distributed is essential for understanding why elements behave the way they do on the periodic table and in chemical reactions.

⚛️ Electron Configuration Principles
The Aufbau Principle
The Aufbau principle states that electrons fill the lowest-energy orbitals first before moving to higher-energy ones. This gives us a predictable filling order.
The filling order doesn't simply go 1, 2, 3 by shell number. Instead, it follows a diagonal pattern because some higher-shell sublevels (like 4s) are actually lower in energy than lower-shell sublevels (like 3d). The standard filling order is:
Each sublevel type holds a specific maximum number of electrons:
- s orbitals: 2 electrons
- p orbitals: 6 electrons
- d orbitals: 10 electrons
- f orbitals: 14 electrons
For example, oxygen has 8 electrons. Following the filling order: . The first 2 go into 1s, the next 2 into 2s, and the remaining 4 into 2p.
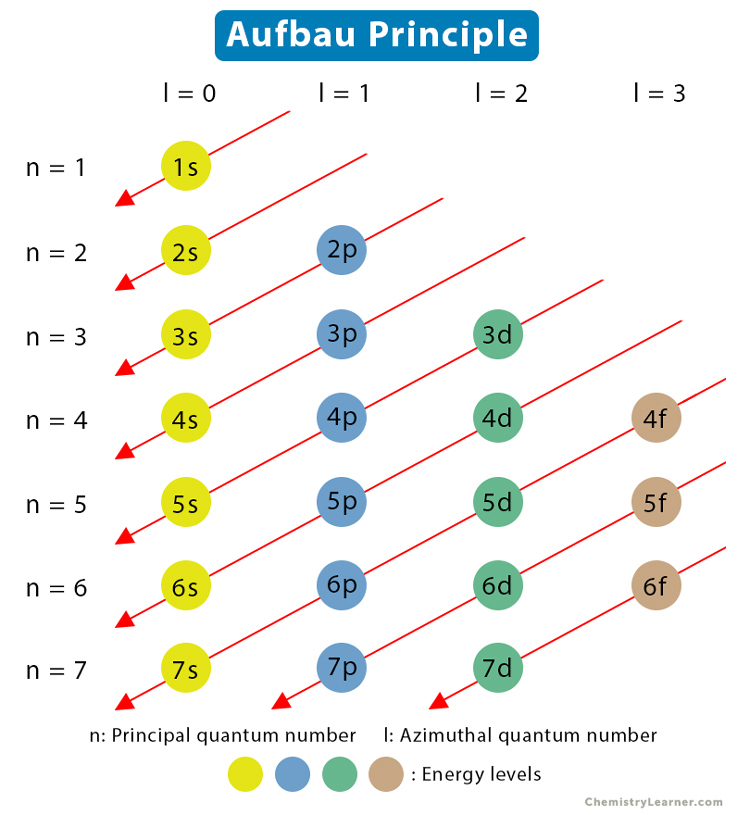
Diagram of the Aufbau Principle.
Image courtesy of Chemistry Learner
💡 Concept Check What is the electron configuration for chlorine, which has 17 electrons?
Solution:
Pauli Exclusion Principle
Each electron in an atom has a set of four quantum numbers that describe its state. The Pauli exclusion principle says no two electrons in an atom can have the same set of all four quantum numbers. The practical result: each orbital can hold a maximum of two electrons, and those two electrons must have opposite spins (one "up," one "down").
In orbital diagrams, we represent this with boxes (orbitals) and arrows (electrons). Two arrows in the same box always point in opposite directions.
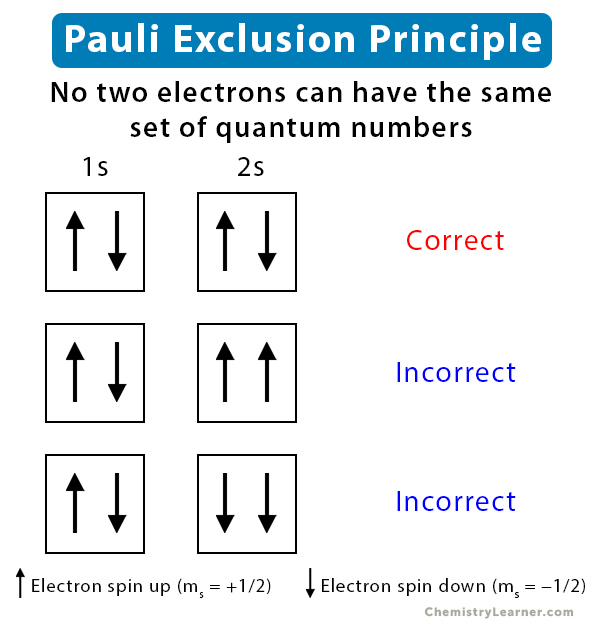
Visual representation of the Pauli Exclusion Principle.
Image courtesy of Chemistry Learner
Hund's Rule
When you have multiple orbitals at the same energy level (like the three 2p orbitals), Hund's rule says: fill each orbital with one electron first, all with the same spin, before pairing any of them up. Think of it like passengers on a bus choosing empty seats before sitting next to someone.
This matters because electrons repel each other. Spreading out into separate orbitals minimizes that repulsion and produces a lower-energy, more stable arrangement.
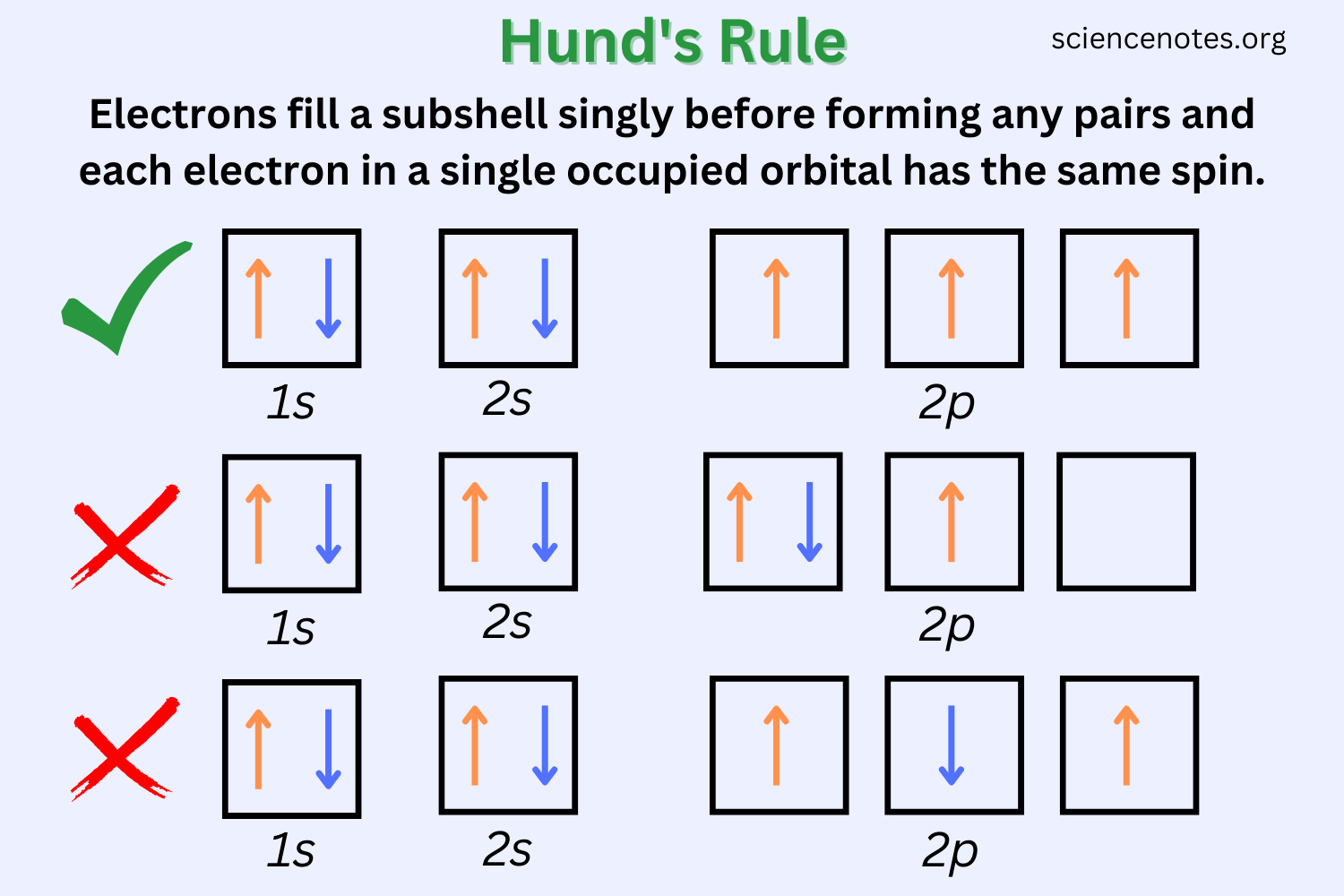
Image showing Hund's Rule.
Image Courtesy of Science Notes
✏️ Applying It
🔢 Writing Electron Configurations
Here's the process for writing an electron configuration:
- Determine the number of electrons (equal to the atomic number for a neutral atom).
- Follow the Aufbau filling order, placing electrons into the lowest-energy sublevels first.
- Fill each sublevel to its maximum (s = 2, p = 6, d = 10, f = 14) before moving to the next.
- Write the configuration using the notation: principal energy level, sublevel letter, superscript for electron count (e.g., ).
Tip: The periodic table itself maps directly onto the filling order. Each row (period) corresponds to a new principal energy level, and the blocks of the table correspond to sublevel types: the first two columns are the s-block, the right six columns are the p-block, the middle ten are the d-block, and the bottom two rows are the f-block.
Practice Question: Write the electron configuration for neon (Ne, 10 electrons).
Answer:
💫 Orbital Diagrams
Orbital diagrams show how electrons are distributed across individual orbitals, including their spin directions. They give you more detail than a written configuration because you can see which orbitals are paired and which aren't.
To draw an orbital diagram:
- Write the Aufbau electron configuration first.
- Draw boxes for each orbital in the configuration. Since each orbital holds at most 2 electrons: s sublevels get 1 box, p sublevels get 3 boxes, d sublevels get 5 boxes, and f sublevels get 7 boxes.
- Fill the boxes with arrows representing electrons. Follow Hund's rule (one electron in each equal-energy orbital before pairing) and the Pauli exclusion principle (paired electrons get opposite spins, shown as up and down arrows).
Practice Question: Draw an orbital diagram for boron (B, 5 electrons).
Boron's configuration is . The 1s and 2s boxes each get a pair of opposite-spin arrows. The 2p sublevel has 3 boxes, and only one gets a single arrow.
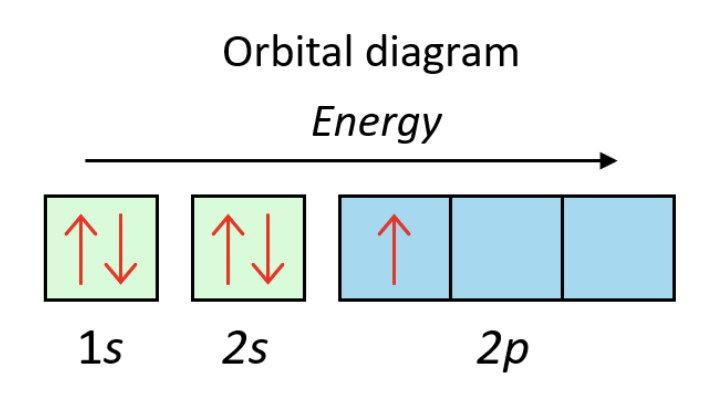
Orbital diagram for boron.
Image courtesy of Chemistry Steps
⛔ Exceptions
Some elements don't follow the expected Aufbau filling order because half-filled and fully filled d sublevels are unusually stable. The two most common exceptions you need to know:
- Chromium (Cr): Expected , actual . One electron shifts from 4s to 3d to achieve a half-filled d sublevel.
- Copper (Cu): Expected , actual . One electron shifts from 4s to 3d to achieve a fully filled d sublevel.
This pattern shows up in other transition metals too, but Cr and Cu are the ones most commonly tested.
🍎 Applications
🐚 The Shell Model & Periodicity
The periodic table is arranged by increasing atomic number, but its structure also reflects electron shell filling patterns, which is why elements in the same column share similar properties.
- Period Trends: Moving across a period (left to right), each element adds one more proton and one more electron. Electrons fill within the same principal energy level across a period.
- Group Similarities: Elements in the same group have the same valence electron configuration (same number and type of outer electrons), which is why they behave similarly in reactions. For example, all Group 1 alkali metals have a single valence electron.
🔌 Valence Electrons & Chemical Reactivity
Valence electrons are the electrons in the outermost energy level. They're the ones directly involved in forming chemical bonds, so they determine how an element reacts.
You can quickly find the number of valence electrons for main-group elements from their group number: Group 1 has 1, Group 2 has 2, Group 13 has 3, and so on up to Group 18 with 8 (except helium, which has 2).
🧪 Influence of Electron Configuration on Chemical Behavior
How electrons are arranged drives several key behaviors:
- Ion formation: Atoms lose or gain electrons to achieve a stable, full outer shell. Sodium () loses its one valence electron to become , while chlorine () gains one to become . Both end up with noble gas configurations.
- Electronegativity: This measures how strongly an atom attracts shared electrons in a bond. Noble gases already have full outer shells, so they generally don't form bonds or exhibit electronegativity.
- Metallicity: Metals have few valence electrons and tend to lose them. Nonmetals have many valence electrons and tend to gain or share them.
🖇️ Bond Types & Electron Configuration
- Ionic Bonds: Formed between metals and nonmetals through the transfer of electrons (e.g., ).
- Covalent Bonds: Formed by the sharing of electron pairs between atoms, typically between nonmetals (e.g., ).
- Metallic Bonds: Metal atoms share a "sea" of delocalized valence electrons, which explains why metals conduct electricity and are malleable.
🌐 Real-World Implications
Electron configurations have direct practical applications:
- Magnetic properties: Atoms with unpaired electrons are paramagnetic (attracted to magnets). Atoms with all electrons paired are diamagnetic (weakly repelled by magnets).
- Semiconductors: Elements like silicon have electronic structures that allow controlled electron movement, making them the basis of computer chips and solar cells.
- Drug design and materials science: Understanding how atoms bond (which comes from their electron configurations) is critical for designing new molecules with specific properties.
📌 Tips and Tricks
- Always fill orbitals starting from the lowest energy level up. Use the diagonal rule or the periodic table's block structure to remember the order.
- The mnemonic "Smart People Don't Fret" helps you remember the sublevel types in order: s, p, d, f.
- Watch for Cr and Cu as the most tested Aufbau exceptions. The pattern is that half-filled () and fully filled () d sublevels are extra stable.
- For main-group elements, the group number tells you the valence electron count. Use this as a quick check on your configurations.