Chemical Bonds 101
Chemical bonds are the forces that hold atoms together in molecules and compounds. The type of bond that forms depends on how atoms interact with each other's electrons. This unit covers the three main bond types: ionic, covalent, and metallic.

Ionic, Covalent, and Metallic Bonds
Ionic Bonds
Ionic bonds form through the transfer of electrons from a metal to a nonmetal. Take as a classic example: sodium gives up one electron to chlorine. This transfer creates positively charged ions (cations) and negatively charged ions (anions). The bond itself is the electrostatic attraction between these oppositely charged ions.
Key Features:
- Formed between metals (which lose electrons) and nonmetals (which gain electrons).
- Results in ionic compounds with distinct positive and negative ions.
- Creates crystalline solids with high melting and boiling points due to strong electrostatic forces throughout the lattice.
- Soluble in polar solvents like water, because the polar molecules can pull the ions apart.
- Conduct electricity when dissolved or molten, since the ions are free to move and carry charge. In solid form, the ions are locked in place and cannot conduct.
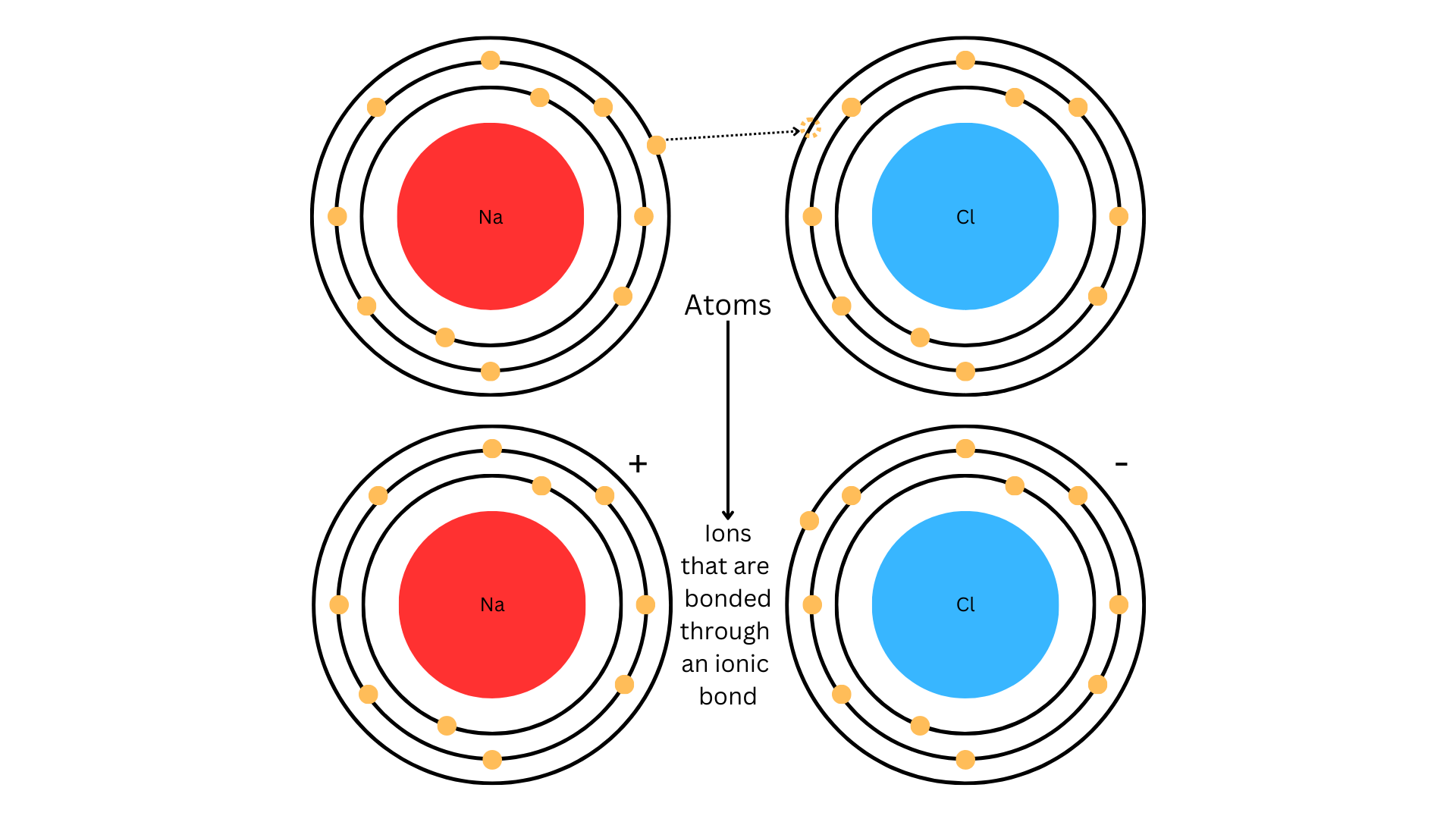
A diagram showing a sodium atom transferring an electron to a chlorine atom, resulting in Na+ and Cl- ions bonded together to form NaCl.
Image Courtesy of Vibhi
Practice Question
Which of these compounds would most likely form an ionic bond? A) B) C) D)
Answer: C. Magnesium is a metal and chlorine is a nonmetal, so electron transfer occurs.
Covalent Bonds
Covalent bonds form when two nonmetal atoms share one or more pairs of electrons. Neither atom has a low enough electronegativity to simply give up electrons, so sharing is the way both atoms can achieve a stable electron configuration.
Key Features:
- Nonpolar covalent bonds involve equal sharing of electrons. This happens when the atoms have the same or very similar electronegativities, like in or .
- Polar covalent bonds involve unequal sharing. The more electronegative atom pulls the shared electrons closer, creating partial charges: a slight negative charge () on that atom and a slight positive charge () on the other. Water () is a good example, where oxygen pulls electron density away from hydrogen.
- Covalent compounds generally have lower melting and boiling points compared to ionic compounds, because the intermolecular forces between covalent molecules are weaker than the electrostatic forces in an ionic lattice.
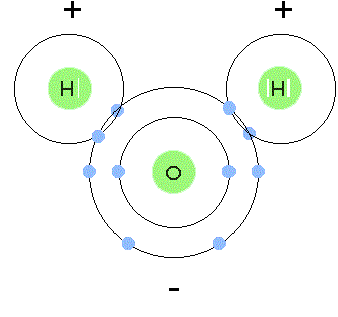
A diagram illustrating the covalent bond between O and H.
Image Courtesy to Socratic
Practice Question
Which of the following best describes a covalent bond?
a) The transfer of electrons between atoms.
b) The sharing of electrons between atoms.
c) The attraction between oppositely charged ions.
d) The interaction of metal atoms with metallic properties.
Answer: B
Metallic Bonds
Metallic bonds occur within pure metals and alloys. Instead of transferring or sharing electrons with specific neighbors, metal atoms release their valence electrons into a shared "sea" of delocalized electrons that flows around positively charged metal nuclei arranged in a lattice.
Key Features:
- The delocalized electrons move freely through the lattice, which is why metals conduct electricity and heat so well.
- Malleability (ability to be hammered into sheets) and ductility (ability to be drawn into wires) also come from this bonding model. When force is applied, the layers of metal cations can slide past each other without breaking the bond, since the electron sea simply shifts with them.
- Metals generally have high melting points, though there's a wide range (mercury is liquid at room temperature, while tungsten melts at over 3400°C).
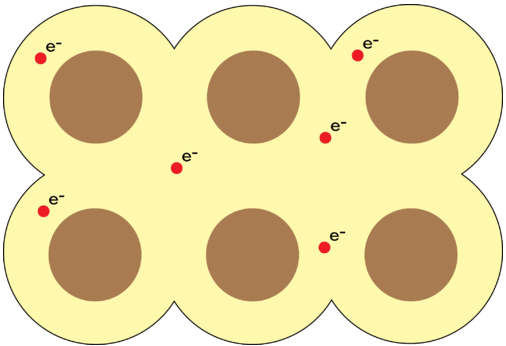
A diagram showing positively charged ions surrounded by a "sea" of delocalized electrons that can flow freely throughout the metal lattice.
Image Courtesy of LibreTexts
Practice Question
What property is most characteristic of metals due to metallic bonding?
A) Low melting point B) Poor conductivity C) High malleability D) Insolubility in water
Answer: C
Electronegativity and Bond Type
The electronegativity difference () between two bonded atoms is the main tool for predicting what type of bond will form. Electronegativity measures how strongly an atom attracts shared electrons.
Here's the logic:
- Large difference (): One atom pulls so much harder that it essentially strips the electron away. This is an ionic bond, like and where .
- Moderate difference (): The atoms share electrons, but unequally. This is a polar covalent bond, like in where oxygen's higher electronegativity creates partial charges.
- Small or no difference (): The atoms share electrons roughly equally. This is a nonpolar covalent bond, like in , , or bonds.
Quick Reference:
Keep in mind that these cutoffs are approximate guidelines, not hard rules. Some sources use slightly different thresholds, so focus on understanding the trend: as increases, bonding shifts from nonpolar covalent to polar covalent to ionic.