Colligative Properties
Colligative properties describe how dissolving a solute in a solvent changes the solution's physical behavior. The key idea: these properties depend only on how many solute particles are present, not on what those particles are. This concept connects solution chemistry to real-world applications like antifreeze, road salt, and medical treatments.

🎈 Intro to Colligative Properties
There are four main colligative properties:
- Boiling point elevation — the boiling point increases
- Freezing point depression — the freezing point decreases
- Vapor pressure lowering — the vapor pressure drops
- Osmotic pressure — pressure develops across a semipermeable membrane
All four share the same underlying principle: when solute particles are added to a solvent, they disrupt the solvent's normal behavior. The identity of the solute doesn't matter. What matters is the number of dissolved particles.
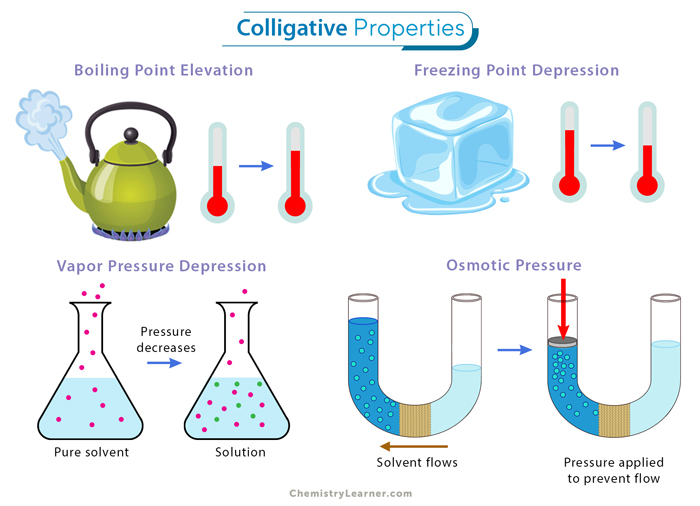
Image showing examples of colligative properties.
Image Courtesy of Chemistry Learner
Why are they important?
- They predict how a solution will behave differently from its pure solvent.
- They provide a method for determining the molar mass of an unknown solute.
- They explain biological processes like osmoregulation (how cells maintain water balance).
💧 Dependence on Solute Concentration
Colligative properties scale with the concentration of solute particles. The concentration unit used most often here is molality, not molarity.
Molality (m):
Molality measures moles of solute per kilogram of solvent. Unlike molarity (moles per liter of solution), molality doesn't change with temperature because mass doesn't expand or contract the way volume does. That makes it the preferred unit for colligative property calculations.
Note the denominator: it's kilograms of solvent, not liters. This is a common mistake on tests.
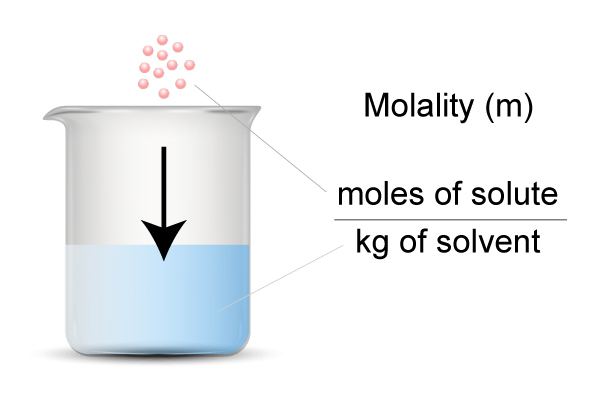
Image explaining how molality is calculated.
Image Courtesy of Surfguppy
🔥 Boiling Point Elevation
Adding a non-volatile solute to a solvent raises its boiling point. Why? Solute particles lower the solvent's vapor pressure (more on this in the Raoult's Law section), so the solution needs to reach a higher temperature before its vapor pressure equals atmospheric pressure and boiling occurs.
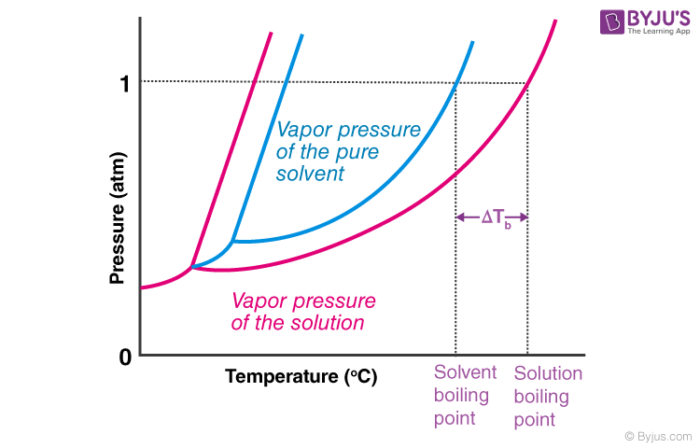
Image explaining boiling point elevation.
Image Courtesy of BYJU'S
Formula:
Where:
- = change in boiling point (always positive, since the boiling point goes up)
- = van't Hoff factor (number of particles the solute produces per formula unit)
- = ebullioscopic constant (a value specific to each solvent; for water, )
- = molality of the solution
Real-World Application:
Antifreeze (ethylene glycol) added to car radiators raises the boiling point of the coolant. This helps prevent the engine from overheating in summer. The same antifreeze also lowers the freezing point, protecting the engine in winter.

Antifreeze being put into a car.
Image Courtesy of Popular Mechanics
❄️ Freezing Point Depression
Adding solute particles to a solvent lowers its freezing point. The solute particles interfere with the solvent molecules' ability to arrange into an ordered crystal lattice (ice), so a lower temperature is needed to freeze.
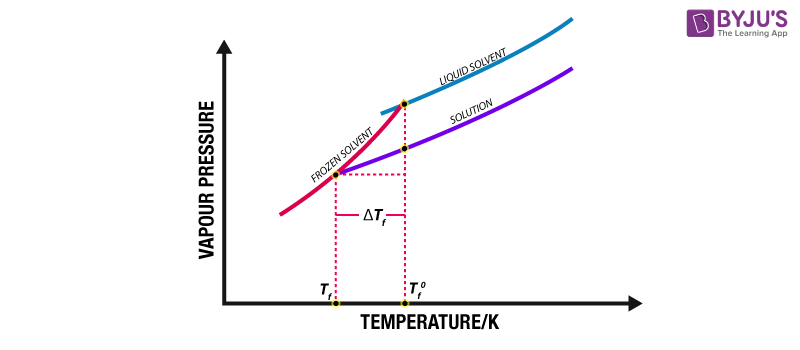
Image explaining freezing point depression.
Image Courtesy of BYJU'S
Formula:
Where:
- = change in freezing point (always positive as a calculated value; you subtract it from the normal freezing point)
- = cryoscopic constant (for water, )
- and have the same meanings as in boiling point elevation
Real-World Application:
Spreading salt () on icy roads lowers the freezing point of water. Since dissociates into and (), it's twice as effective per formula unit as a non-electrolyte like sugar.

Image showing how roads are salted.
Image Courtesy of Mental Floss
💦 Osmotic Pressure (π)
Osmosis is the movement of solvent molecules through a semipermeable membrane from a region of lower solute concentration to higher solute concentration. Osmotic pressure is the pressure required to stop that flow. This is critical in biology for maintaining fluid balance in cells.
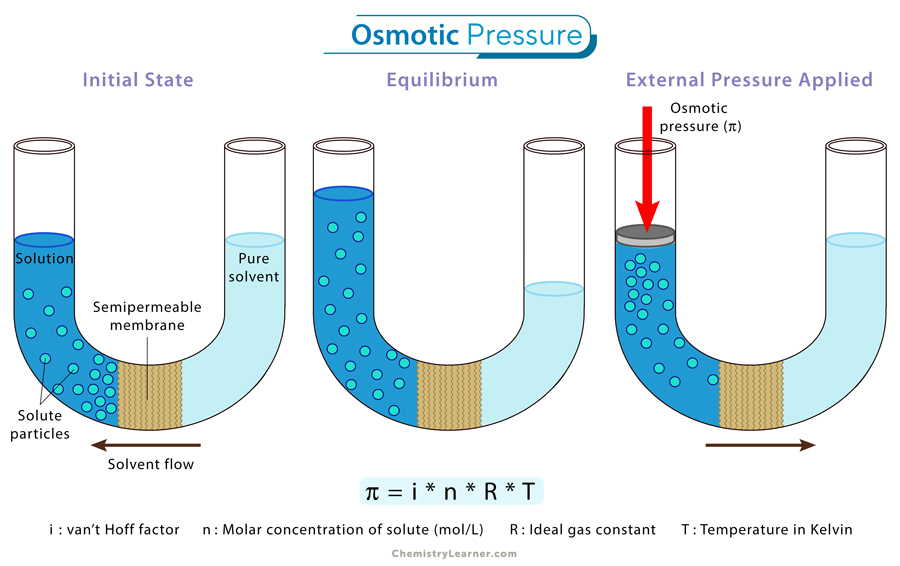
Image explaining osmotic pressure.
Image Courtesy of Chemistry Learner
Formula:
Where:
- = osmotic pressure (in atm or Pa)
- = van't Hoff factor
- = molarity of the solution
- = gas constant ( or , depending on your pressure units)
- = temperature in Kelvin
Note that osmotic pressure uses molarity (), not molality. This is the one colligative property formula where that's the case.
Application:
Dialysis machines for kidney failure patients use osmotic pressure principles. The machine's membrane allows waste molecules to pass through while keeping blood cells and proteins in the bloodstream.
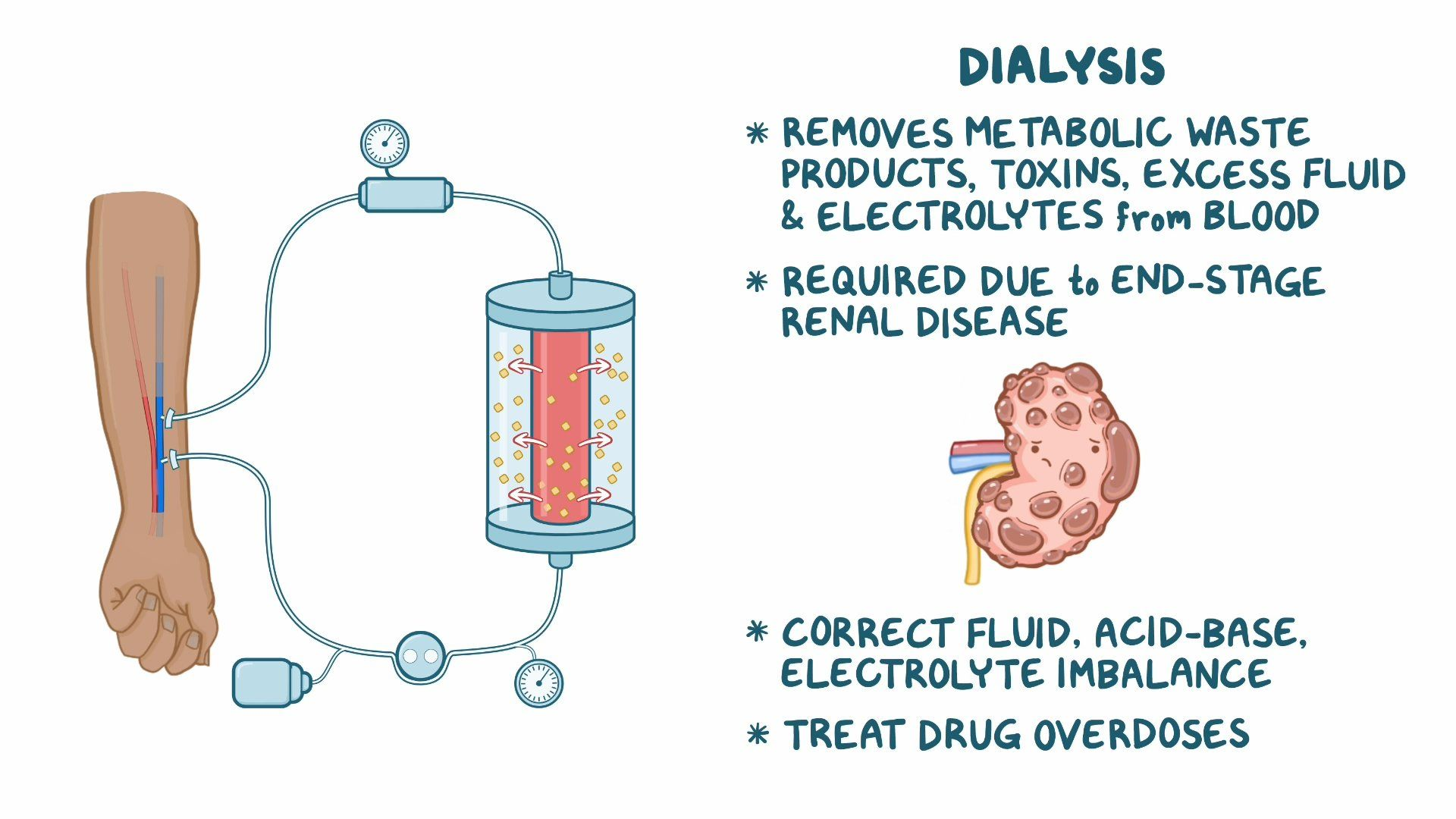
Image showing how dialysis works.
Image Courtesy of Osmosis
🔢 Van't Hoff Factor and Ionization
The van't Hoff factor () represents the number of particles one formula unit of solute produces when it dissolves. This factor is what connects colligative properties to electrolytes.
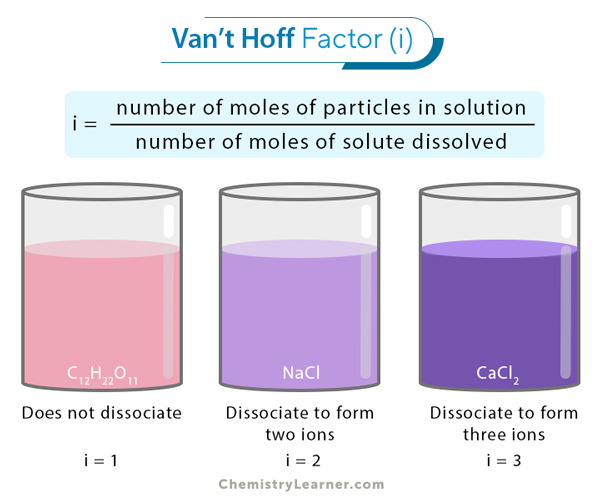
Image showing the van't Hoff factor and how it works.
Image Courtesy of Chemistry Learner
Here's how to determine :
- Non-electrolytes (like glucose or sucrose) don't dissociate. They stay as whole molecules, so .
- Strong electrolytes fully dissociate into ions. Count the total ions per formula unit.
- Weak electrolytes only partially dissociate, so falls between 1 and the theoretical maximum.
Example:
Sodium sulfate dissolving in water:
One formula unit produces 2 sodium ions and 1 sulfate ion = 3 particles total. So .
A few common values to know:
| Solute | Dissociation | Theoretical |
|---|---|---|
| Glucose () | None | 1 |
| 2 | ||
| 3 | ||
| 3 |
In practice, measured values for strong electrolytes are often slightly less than the theoretical values due to ion pairing in solution. For honors-level problems, use the theoretical value unless told otherwise.
📉 Raoult's Law and Its Applications
Raoult's Law explains vapor pressure lowering quantitatively. When you add a non-volatile solute to a solvent, some solvent molecules at the surface get replaced by solute particles. Fewer solvent molecules can escape into the gas phase, so the vapor pressure drops.
Where:
- = vapor pressure of the solution
- = mole fraction of the solvent (always less than 1 when solute is present)
- = vapor pressure of the pure solvent
Since , the solution's vapor pressure is always lower than the pure solvent's. The more solute you add, the lower gets, and the more the vapor pressure drops.
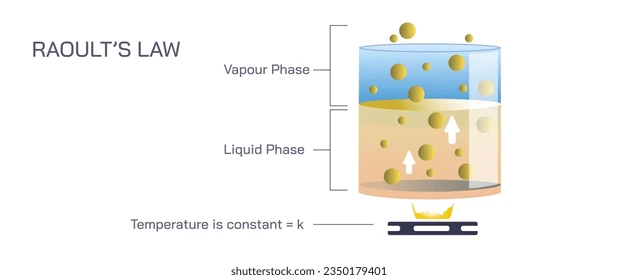
Image explaining Raoult's law.
Image Courtesy of Shutterstock
This vapor pressure lowering is actually the root cause of both boiling point elevation and freezing point depression. Lower vapor pressure means you need a higher temperature to boil and a lower temperature to freeze.
⚛️ Colligative Properties of Electrolytes
Electrolytes dissociate into ions when dissolved, which means they produce more particles per formula unit than non-electrolytes. More particles = bigger colligative effects.
- Strong electrolytes (like , , ) fully dissociate. A 1 m solution of effectively behaves like a 2 m solution of particles because each formula unit gives 2 ions.
- Weak electrolytes (like acetic acid, ) only partially dissociate. Their value is greater than 1 but less than the theoretical maximum.
- Non-electrolytes (like sugar or ethanol) don't dissociate at all. .
This is exactly why the van't Hoff factor appears in every colligative property formula. It accounts for the actual number of particles in solution.
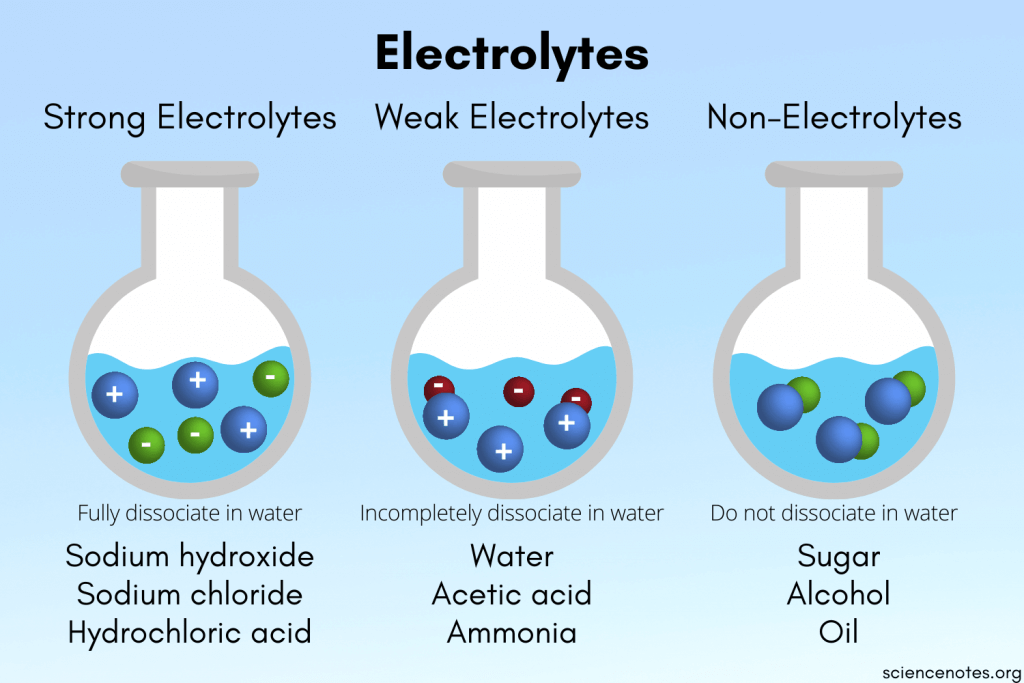
Image describing strong, weak, and non-electrolytes.
Image Courtesy of ScienceNotes.org
🛠️ Practical Application
Colligative properties show up in several real-world technologies:
- Seawater desalination uses reverse osmosis, where pressure greater than the osmotic pressure is applied to force water backward through a membrane, separating it from dissolved salts.
- Freeze-drying (lyophilization) in pharmaceuticals relies on understanding how solutes affect freezing points to preserve medications and biological samples.
- De-icing of roads and aircraft depends on freezing point depression.
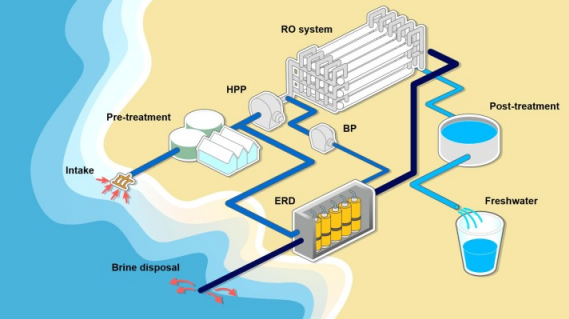
Image showing the seawater desalination process.
Image Courtesy of Waterman Engineers Australia
🚨 Practice Questions
- How does adding a non-volatile solute to a solvent affect its freezing point?
Answer: It lowers the freezing point. Solute particles disrupt the formation of the solvent's crystal lattice, requiring a lower temperature to freeze.
- How does a strong electrolyte differ from a weak electrolyte in terms of colligative properties?
Answer: A strong electrolyte fully dissociates in solution, producing more particles per formula unit and causing larger changes in colligative properties. A weak electrolyte only partially dissociates, so its effect is smaller.
- How does vapor pressure relate to colligative properties?
Answer: Adding a solute lowers the solution's vapor pressure (Raoult's Law). This vapor pressure lowering is the fundamental cause of both boiling point elevation and freezing point depression.