Ideal Gas Law and Beyond
The previous section covered Kinetic Molecular Theory (KMT) and several foundational gas laws. Here's a quick recap of those laws before we build on them:
- Boyle's Law:
- Charles' Law:
- Gay-Lussac's Law:
- Combined Gas Law:
- Avogadro's Law:
For detailed explanations and worked examples of these individual laws, check the previous study guide.

The Ideal Gas Law (PV = nRT)
The Ideal Gas Law ties together all four gas variables into a single equation, so you don't need to juggle five separate laws. It relates pressure (P), volume (V), number of moles (n), and temperature (T) through the ideal gas constant (R).
The Ideal Gas Law is: , where
- P = pressure (in atm)
- V = volume (in L)
- n = number of moles
- R = ideal gas constant =
- T = temperature (in K)
What makes this equation so useful is that it's really just Boyle's, Charles', and Avogadro's Laws combined. If you hold and constant, you get Boyle's Law. Hold and constant, and Charles' Law falls out. One equation covers all the cases.
When do gases behave ideally? Gases best follow this equation at high temperatures and low pressures. Under those conditions, molecules are spread far apart and moving fast, so intermolecular attractions are negligible and the molecules' own volume is tiny compared to the container. As you increase pressure or decrease temperature, real gases start deviating from ideal predictions.
No real gas is perfectly ideal, but the Ideal Gas Law is an excellent model for predicting gas behavior under typical conditions.
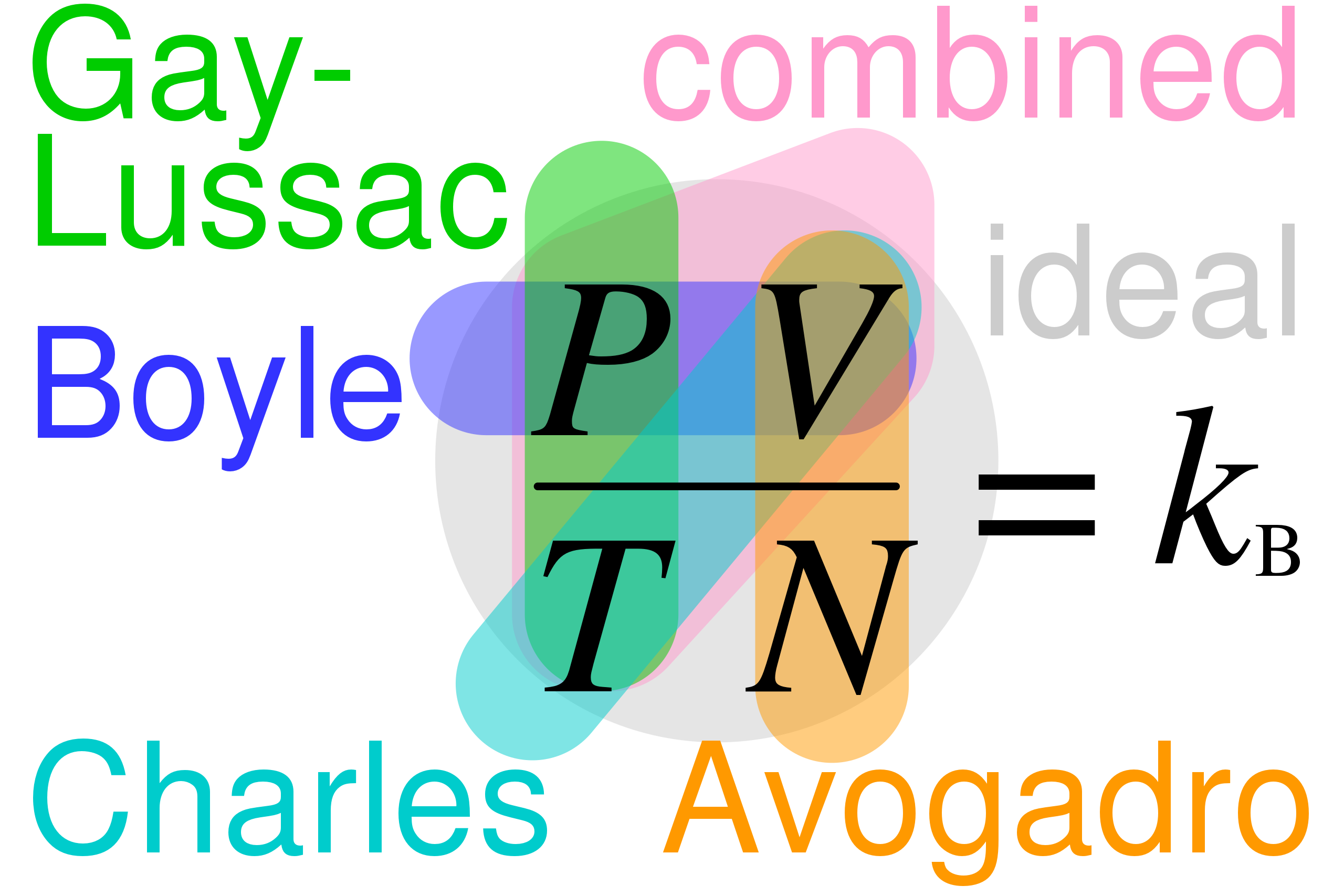
Image courtesy of Wikimedia Commons
Practice Questions
Practice Question 1: A sample of neon has a volume of 12 L at sea level (1 atm) and room temperature (25°C). Calculate the number of moles of neon.
Identify your knowns and unknown:
- Convert temperature to Kelvin:
- Rearrange for :
- Plug in values:
Practice Question 2: A gas sample contains 0.5 moles at a pressure of 2.50 atm and a temperature of 300 K. Calculate the volume.
- , , ,
- Rearrange for :
- Plug in:
Practice Question 3: A gas sample contains 2.5 moles at a volume of 10.0 L and a temperature of 400 K. Calculate the pressure.
- , , ,
- Rearrange for :
- Plug in:
Practice Question 4: A gas sample contains 0.25 moles at a pressure of 2.00 atm and a volume of 5.00 L. Calculate the temperature.
- , , ,
- Rearrange for :
- Plug in:
Notice the pattern across all four problems: identify your known variables, rearrange to isolate the unknown, make sure your units are consistent (temperature must be in Kelvin), and plug in. That's the whole process every time.
Applying the Ideal Gas Law in Our World
The Ideal Gas Law shows up in more places than you'd expect:
- Environmental Science: Calculating how air density changes with altitude, which matters for understanding weather patterns and pollution dispersion.
- Vehicular Engineering: Airbags rely on precise gas calculations so they inflate to the right volume in milliseconds during a crash. The chemical reaction inside the airbag produces a known number of moles of gas, and determines the final volume.
Practice Question 5: What is the density of dry air at STP if dry air has an average molar mass of 29 g/mol?
At STP: ,
- Find the volume of 1 mole of gas at STP using :
This result (22.4 L/mol at STP) is worth remembering. It's called the molar volume of an ideal gas at STP.
- Use with the molar mass and molar volume:
Real Gases and Deviations from Ideality
This content is optional and is included for deeper understanding.
Real gases deviate from ideal behavior when the assumptions of KMT start to break down:
- At high pressures (or low volumes), gas molecules are packed closely together. Their own physical volume becomes significant compared to the container, and intermolecular forces (attractions and repulsions) start to matter.
- At low temperatures, molecules move more slowly and spend more time near each other, making attractive forces more influential. This can even lead to condensation into a liquid.
Van der Waals Equation
To account for these deviations, the van der Waals equation modifies the Ideal Gas Law with two correction terms:
- The term corrects for intermolecular attractions (which reduce the measured pressure below what an ideal gas would exert).
- The term corrects for the finite volume of the gas molecules themselves (which reduces the available space).
- The constants and are specific to each gas.
Wrapping Up the Ideal Gas Law
Gases behave most ideally at high temperatures and low pressures. Under those conditions, lets you predict any one of the four variables (P, V, n, T) if you know the other three. Always convert temperature to Kelvin and keep your units consistent with your value of R.
For Honors Chemistry, the Ideal Gas Law is your go-to tool for gas problems. Master the rearrangement for each variable, and you'll be set.