Calorimetry: Measuring Heat Changes in Chemical Reactions
Calorimetry is the experimental technique for measuring heat absorbed or released during chemical and physical changes. In thermochemistry, it's how you connect theoretical energy calculations to real, measurable temperature data. This section covers the core principles, the two main types of calorimeters, the key equation , and how to use calorimetry data to find enthalpy changes.

Principles of Calorimetry
Calorimetry works because of a simple physical fact: when two objects at different temperatures come into contact, heat flows from the hotter object to the cooler one until both reach the same temperature. That final shared temperature is called thermal equilibrium.
The entire method rests on the Law of Conservation of Energy: energy is never created or destroyed, only transferred or converted. In a calorimeter, all the heat lost by one substance must be gained by another. You can write this as:
This relationship is what makes calorimetry calculations possible. If you can measure the temperature change of the surroundings (usually water), you can figure out how much heat the reaction released or absorbed.
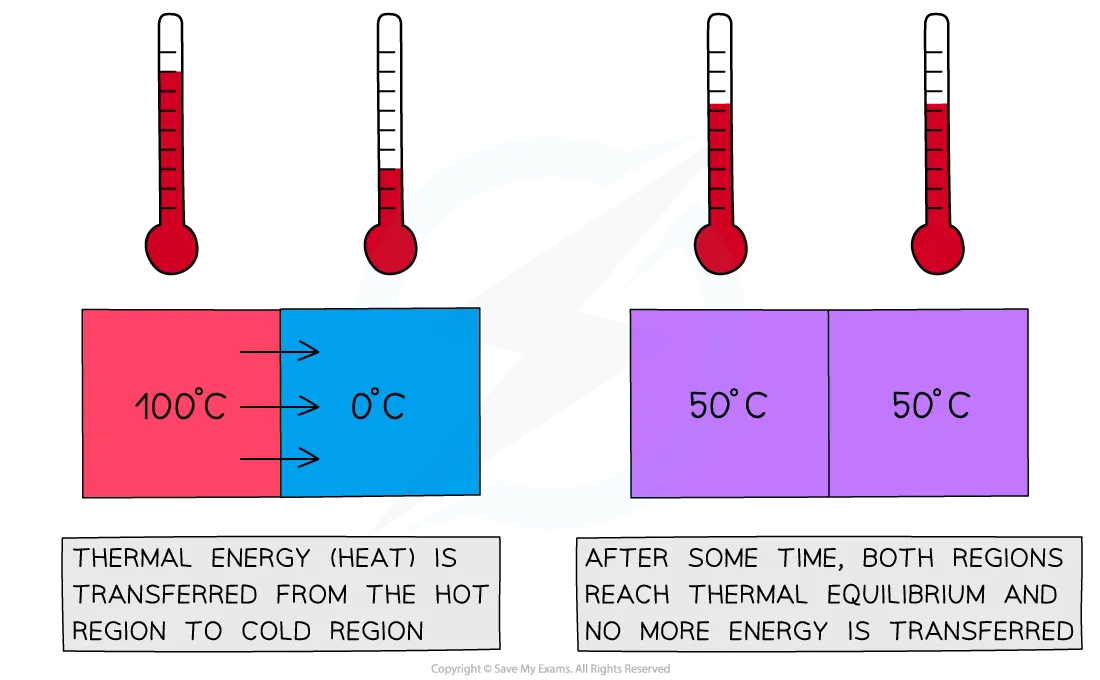
Image showing the relationship between thermal energy and equilibrium.
Image Courtesy of Save My Exams
Types of Calorimeters
Coffee Cup Calorimeter
This is the calorimeter you'll most likely use in lab. It's made from two nested styrofoam cups with a lid and a thermometer inserted through the top. Styrofoam is a good insulator, so minimal heat escapes to the room.
Because the cups are open to the atmosphere, reactions occur at constant pressure. That means the heat measured () directly equals the enthalpy change of the reaction. This setup works well for dissolving salts, neutralization reactions, and other aqueous-phase processes.
Limitations: Some heat always leaks through the styrofoam, so coffee cup calorimeters aren't perfectly accurate. For honors-level work, you should recognize this as a source of experimental error.
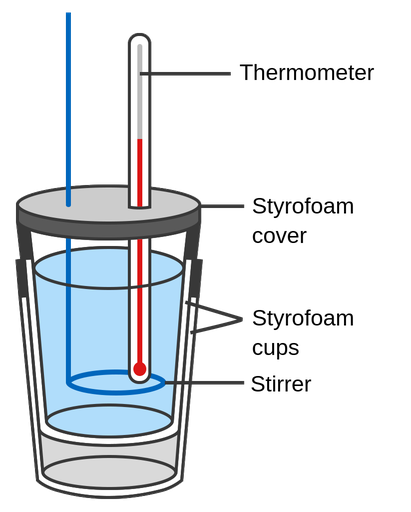
Image of a coffee cup calorimeter.
Image Courtesy of Labster
Bomb Calorimeter
A bomb calorimeter is a sealed, rigid steel container submerged in a known mass of water. The reaction takes place inside the "bomb" at constant volume. Because the container is sealed and heavily insulated, very little heat escapes, making measurements much more precise.
Bomb calorimeters are used for combustion reactions, such as determining the energy content of fuels or food. Since volume is constant (not pressure), the heat measured here technically corresponds to the internal energy change (), though for most reactions the difference from is small.
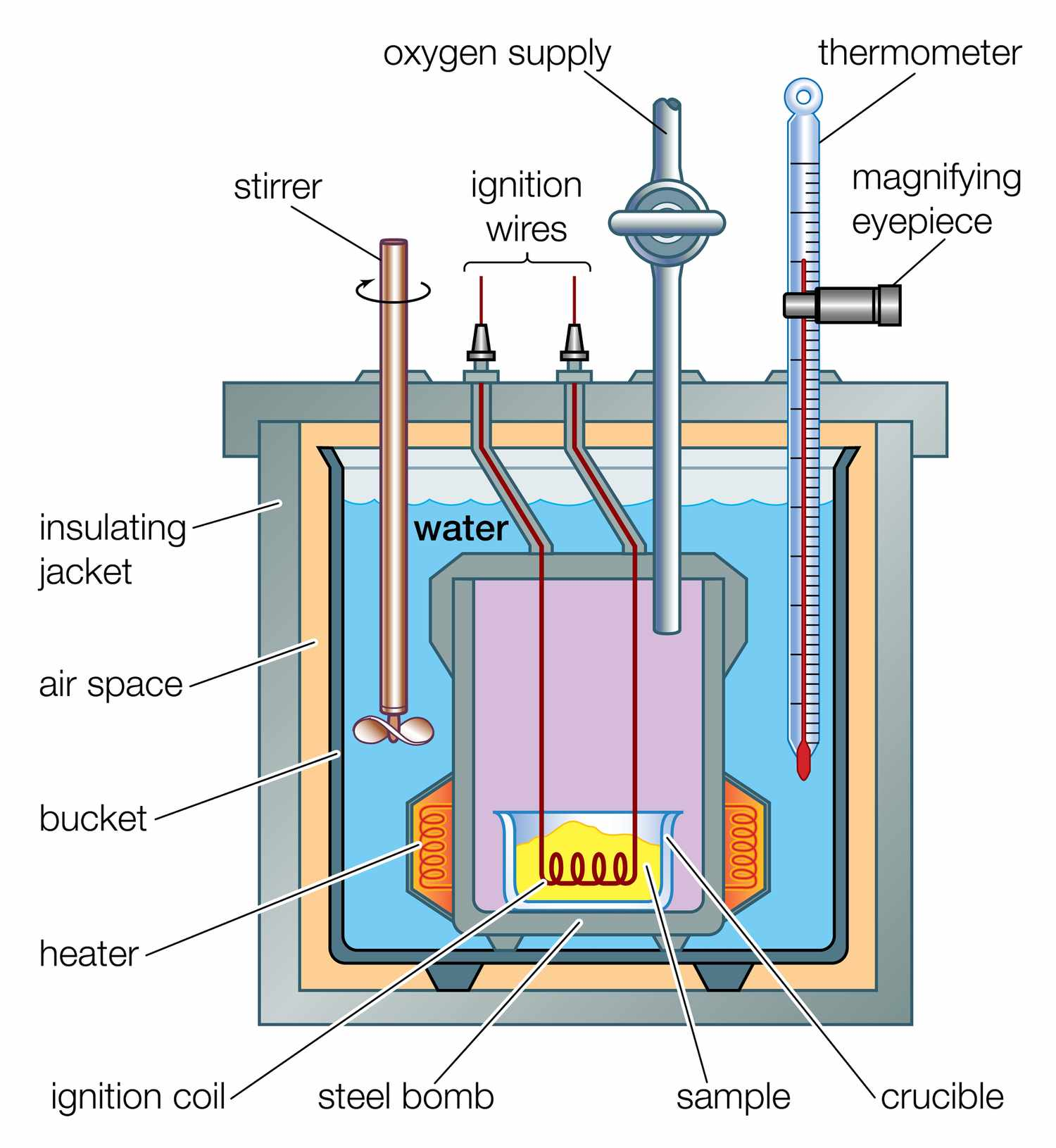
Image of a bomb calorimeter.
Image Courtesy of ThoughtCo
Calculating Heat Changes
The central equation in calorimetry connects heat transfer to mass, specific heat, and temperature change.
Specific Heat Capacity vs. Molar Heat Capacity
- Specific heat capacity (): The amount of heat required to raise the temperature of 1 gram of a substance by 1°C. Units: J/g°C. For water, , which is unusually high compared to most substances. This is why water is used as the surrounding fluid in most calorimeters.
- Molar heat capacity: The amount of heat required to raise the temperature of 1 mole of a substance by 1°C. Units: J/mol°C.
You'll use specific heat capacity far more often in calorimetry problems. Molar heat capacity shows up more when comparing substances on a per-particle basis.
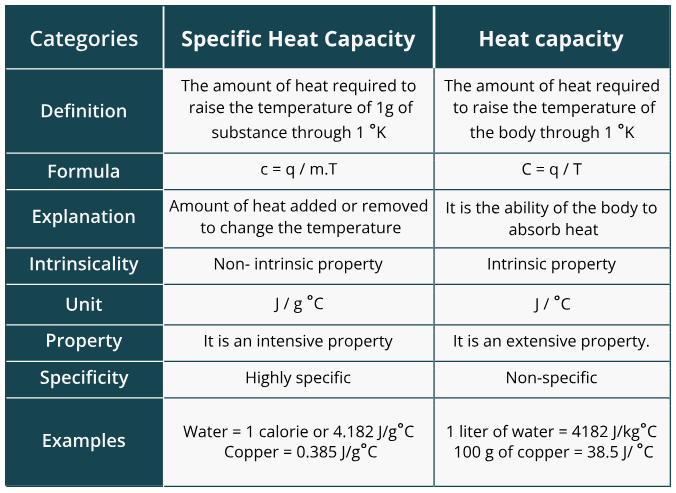
Image describing the differences between specific heat and heat capacities.
Image Courtesy of PSIBERG
The Equation:
This equation calculates the heat () absorbed or released by a substance when its temperature changes.
- = heat absorbed or released (Joules)
- = mass of the substance (grams)
- = specific heat capacity (J/g°C)
- = change in temperature = (°C)
The sign of tells you the direction of heat flow:
- Positive : the substance absorbed heat (temperature went up)
- Negative : the substance released heat (temperature went down)
Practice Problem
Problem: A 150 g iron block () cools from 200°C to 100°C. How much heat was transferred?
Step-by-step solution:
-
Identify your variables: , , ,
-
Calculate :
-
Plug into the equation:
The negative sign confirms the iron block released 6750 J of heat as it cooled. If this block were placed in water inside a calorimeter, the water would gain +6750 J (conservation of energy).
Common mistake: Forgetting to subtract in the right order for . Always do final minus initial. If you flip it, your sign will be wrong, and you'll misidentify whether heat was absorbed or released.
Determining Enthalpy Changes
Calorimetry data lets you calculate the enthalpy change () of a reaction, which tells you the heat flow at constant pressure per amount of substance reacted.
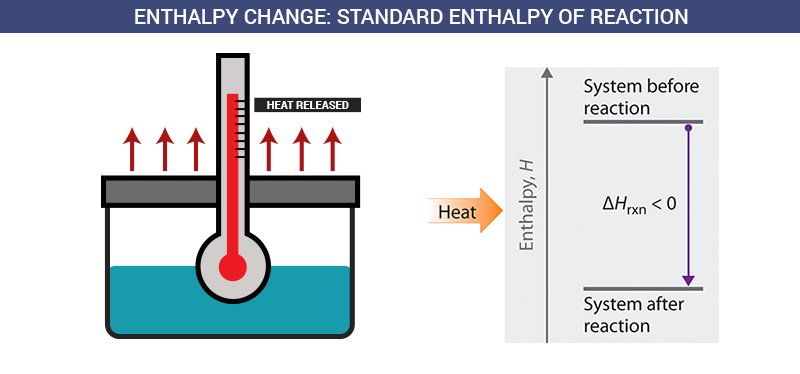
Image showing the standard enthalpy of reaction.
Image Courtesy of Careers Today
Enthalpy (H) vs. Heat (q)
- Enthalpy (): A thermodynamic property representing the total heat content of a system at constant pressure. You can't measure directly; you measure changes in it.
- Heat (): The energy actually transferred between a system and its surroundings due to a temperature difference. This is what the calorimeter measures.
At constant pressure (like in a coffee cup calorimeter), for the process occurring.
Enthalpy Change ()
- Exothermic reactions release heat to the surroundings:
- Endothermic reactions absorb heat from the surroundings:
To find per mole of reactant from calorimetry data:
- Use to calculate the heat gained or lost by the water (or solution).
- Apply conservation of energy: (the reaction's heat is equal and opposite to what the water gained).
- Divide by the moles of the limiting reactant:
The sign of should make physical sense. If the water temperature increased, the reaction was exothermic, so should be negative.
Conclusion
Calorimetry connects measurable temperature changes to the energy changes driving chemical reactions. The core workflow is straightforward: measure a temperature change, apply , and use conservation of energy to find the heat of reaction. From there, dividing by moles gives you . Practice setting up problems carefully, watch your signs, and keep track of what's gaining vs. losing heat.