Basic Organic Reactions and Mechanisms
Organic chemistry isn't just about naming molecules. The biomolecules you encounter in this unit (carbohydrates, proteins, and lipids) are the structural and functional foundation of living systems. Understanding how they're built, what they do, and how they're metabolized connects organic chemistry to real biological processes.
This guide covers the structure, function, and metabolism of each major biomolecule class.

Carbohydrates: Energy and Structure
Structure of Carbohydrates
Carbohydrates contain carbon, hydrogen, and oxygen, typically in a ratio close to . They range from simple sugars to long polymer chains.
- Monosaccharides are the simplest carbohydrates, consisting of a single sugar unit. Glucose () and fructose are common examples. These form ring structures in aqueous solution.
- Disaccharides consist of two monosaccharides joined by a glycosidic bond. Lactose (glucose + galactose) and sucrose (glucose + fructose) fall into this category.
- Polysaccharides are long chains of monosaccharide units. The prefix poly- means "many," so these are polymers of simple sugars. Cellulose provides structural support in plant cell walls, while glycogen serves as an energy reserve in animal tissues.
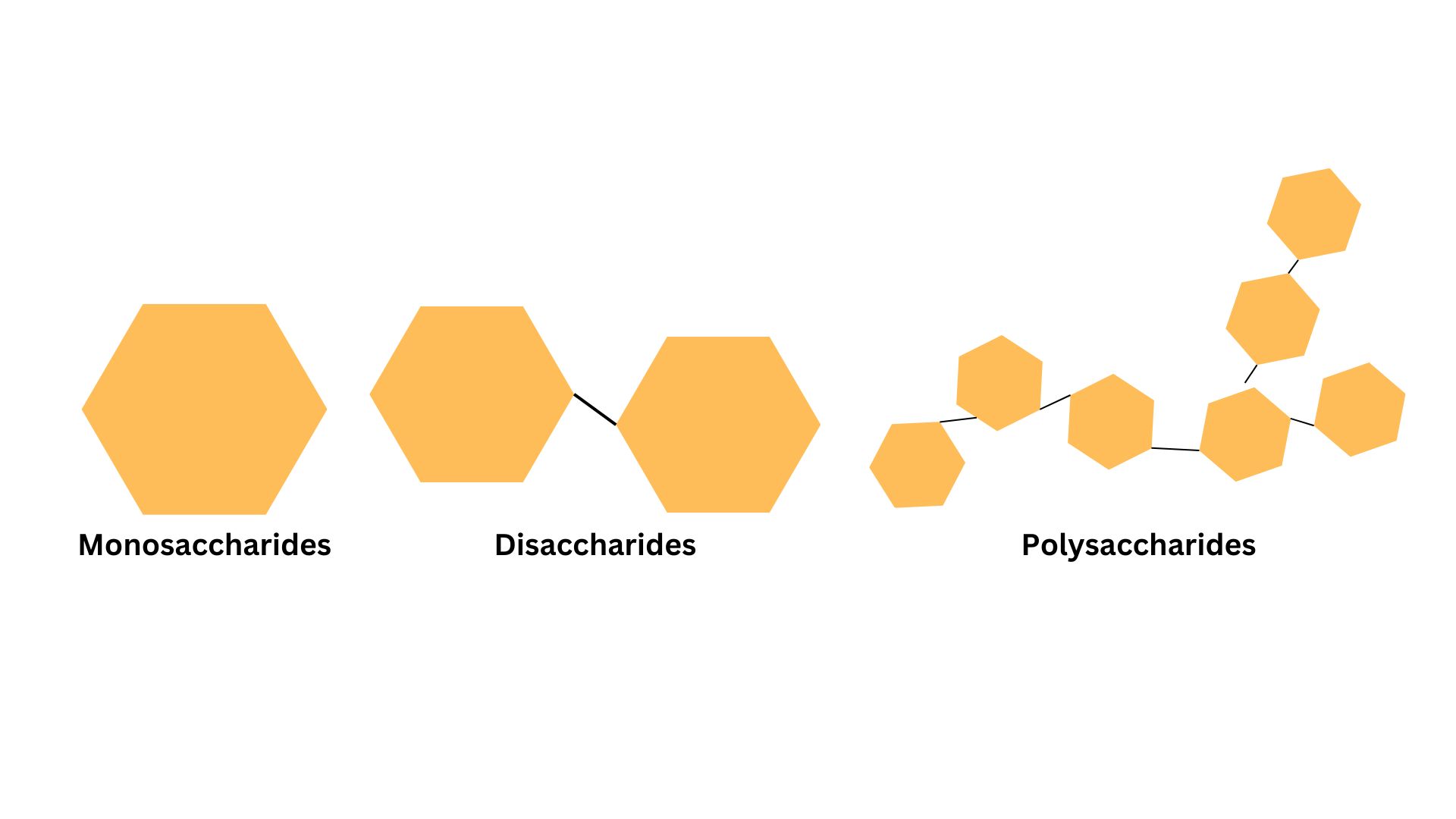
Image representing a monosaccharide, disaccharide, and polysaccharide.
Image Courtesy of Vibhi
Functions of Carbohydrates
- Energy Source: Glucose is the primary fuel for cellular respiration. Cells break it down to produce ATP.
- Energy Storage: Plants store glucose as starch; animals store it as glycogen. Both can be broken down when energy demand increases.
- Structural Role: Cellulose gives plant cell walls their rigidity. Chitin, a similar polysaccharide, provides structural support in fungal cell walls and arthropod exoskeletons.
Practice Question
- Identify whether lactose is a mono-, di-, or polysaccharide and explain its role in biological systems.
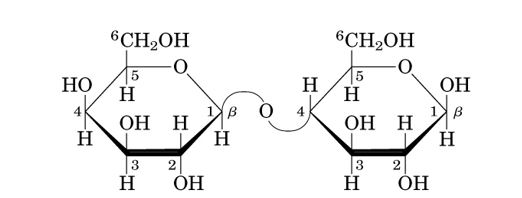
Image of a lactose molecule.
Image courtesy of Wikimedia Commons.
Explanation: Lactose is a disaccharide made of two sugar molecules (glucose and galactose) linked by a glycosidic bond. In biological systems, it's the primary carbohydrate in milk, providing energy for nursing infants. Enzymes in the body (specifically lactase) break lactose into its component monosaccharides for absorption in the digestive system.
Proteins: Structure and Function
Structure of Proteins
Proteins are polymers of amino acids linked by peptide bonds (a type of covalent bond formed through a condensation reaction between the amino group of one amino acid and the carboxyl group of another). Protein structure is organized into four levels:
- Primary Structure: The linear sequence of amino acids in the polypeptide chain. This sequence determines all higher levels of structure.
- Secondary Structure: Local folding patterns stabilized by hydrogen bonds between backbone atoms. The two main types are alpha-helices (coils) and beta-sheets (pleated folds).
- Tertiary Structure: The overall 3D shape of a single polypeptide, determined by interactions between amino acid side chains (R groups). These interactions include hydrogen bonds, ionic bonds, disulfide bridges, and hydrophobic interactions.
- Quaternary Structure: The arrangement of two or more polypeptide chains into a functional protein complex. Hemoglobin, for example, consists of four polypeptide subunits.
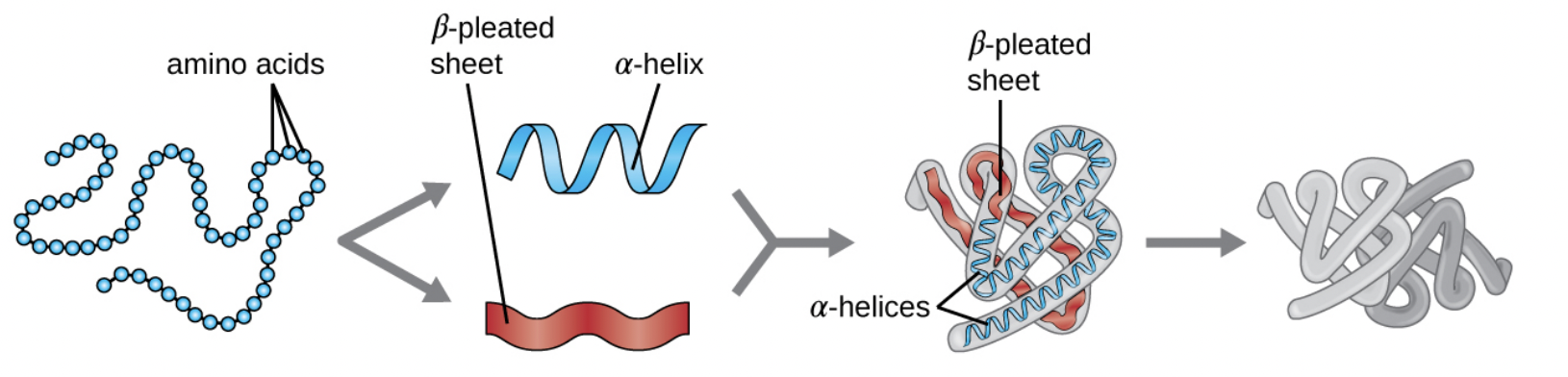
Image representing the four different protein structures.
Image Courtesy of Wikimedia Commons
Functions of Proteins
- Enzymes: Act as biological catalysts that speed up chemical reactions without being consumed. Each enzyme is specific to a particular reaction.
- Structural Components: Collagen provides tensile strength in connective tissues; keratin is the structural protein in hair, nails, and skin.
- Signaling Molecules: Some hormones (like insulin) are proteins that carry chemical messages between cells.
- Immune Defense: Antibodies are proteins that recognize and bind to specific foreign substances (antigens), helping the body fight infection.
Practice Question
-
Alpha-keratin, found in the outer layer of skin, hair, nails, feathers, and horns of vertebrates, has alpha-helices. What level of protein structure does alpha-keratin exhibit?
Explanation: Secondary structure, because alpha-helices are a defining feature of secondary structure (local folding patterns stabilized by hydrogen bonds along the polypeptide backbone).
Lipids: Storage, Membranes, and Signaling
Structure of Lipids
Lipids are a diverse group of biomolecules united by one key property: they are hydrophobic (they don't dissolve well in water) because they consist largely of nonpolar hydrocarbon chains.
- Fats and Oils (Triglycerides): Composed of one glycerol molecule bonded to three fatty acid chains via ester linkages. Saturated fatty acids have no carbon-carbon double bonds and pack tightly (solid at room temperature). Unsaturated fatty acids contain one or more double bonds, creating kinks that prevent tight packing (liquid at room temperature).
- Phospholipids: Similar to triglycerides, but one fatty acid is replaced by a phosphate group. This gives them a hydrophilic "head" and two hydrophobic "tails," making them amphipathic.
- Steroids: Built on a framework of four fused carbon rings with various functional groups attached. Cholesterol is the most well-known example.
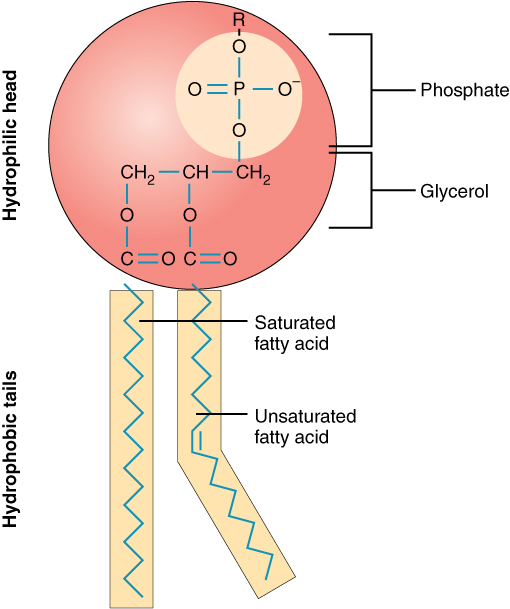
Image representing a phospholipid.
Image Courtesy of Wikimedia Commons
The hydrophobic tail region consists of long fatty acid chains with mostly nonpolar bonds. Because these chains cannot form hydrogen bonds with water, they are repelled by aqueous environments.
Functions of Lipids
- Energy Storage: Fats store about 9 kcal per gram, more than double the energy density of carbohydrates or proteins (~4 kcal/g each). However, the body preferentially burns carbohydrates first because they're faster to break down, then lipids, and finally proteins.
- Membrane Structure: Phospholipids self-assemble into bilayers that form the basis of all cell membranes, creating a selective barrier between the cell and its environment.
- Signaling Molecules: Steroid hormones (like testosterone and estrogen) act as chemical messengers. Because they're lipid-soluble, they can pass through cell membranes and bind to intracellular receptors.
Practice Question
-
In water, phospholipids spontaneously form lipid bilayer membranes. Based on their structure, demonstrated in the photo above, explain why this might occur.
Explanation: The hydrophilic phosphate heads are attracted to water and orient toward the aqueous environment. The hydrophobic fatty acid tails are repelled by water and orient inward, away from it. This causes phospholipids to self-assemble into bilayers where the hydrophobic tails are sandwiched between two layers of hydrophilic heads, minimizing contact between the nonpolar tails and water.
Metabolism of Biomolecules
Each class of biomolecule has its own metabolic pathways for synthesis and breakdown. Here's a summary of the key processes.
Carbohydrate Metabolism
- Glycolysis breaks glucose (a 6-carbon molecule) into two molecules of pyruvate (3 carbons each), generating a small amount of ATP.
- Gluconeogenesis is the reverse process: synthesizing glucose from non-carbohydrate sources (like amino acids or glycerol) when blood sugar is low.
- Glycogenesis is the synthesis of glycogen from glucose for storage. Glycogenolysis is the breakdown of glycogen back into glucose when energy is needed. (Notice the suffixes: -genesis = creation, -lysis = breakdown.)
Protein Metabolism
- Translation converts the genetic information encoded in mRNA into a polypeptide chain, which then folds into a functional protein.
- Proteolysis breaks proteins down into individual amino acids. These can be recycled to build new proteins or converted into energy.
Lipid Metabolism
- Beta-oxidation breaks fatty acid chains into two-carbon units (acetyl-CoA), releasing energy. The glycerol backbone can enter glycolysis separately.
- Triglyceride synthesis (lipogenesis) stores excess energy as fat within adipose cells when caloric intake exceeds demand.
Final Tips
- Relate structure to function for every biomolecule. Why does a phospholipid form a bilayer? Because of its amphipathic structure. That kind of reasoning is what exam questions test.
- Know the monomers and polymers: amino acids build proteins, monosaccharides build polysaccharides, fatty acids and glycerol build triglycerides.
- Pay attention to the types of bonds: glycosidic bonds in carbohydrates, peptide bonds in proteins, ester linkages in lipids. These bonds form through condensation (dehydration) reactions and break through hydrolysis.