Understanding Matter
Matter is anything that has mass and takes up space. That definition sounds simple, but understanding how matter is classified, how it changes states, and how mixtures can be separated forms the foundation for nearly everything else in chemistry.

3️⃣ States of Matter
Matter exists in three primary states: solid, liquid, and gas. The differences between them come down to how their particles are arranged and how much energy those particles have.
🧊 Solids
Here are the key characteristics of a solid:
- Definite Shape and Volume: Solids hold a fixed shape and don't conform to their container. Their volume stays constant too.
- Closely Packed Particles: Particles in a solid are tightly packed in a regular, ordered arrangement.
- Vibrational Movement: These particles vibrate in place but don't move freely from one position to another.
These characteristics give solids two notable properties:
- High Density: The tight packing of particles means solids generally have higher densities than liquids or gases.
- Low Compressibility: Since the particles are already so close together, you can't squeeze a solid into a smaller volume.
Think about an ice cube. It has a fixed shape and a fixed volume. You can hold it in your hand, but you can't compress it or reshape it by squeezing.

Particle representation of a solid.
Image Courtesy of Live Science
💧 Liquids
Liquids behave differently from solids in a few important ways:
- Indefinite Shape, Definite Volume: A liquid takes the shape of whatever container it's in, but its volume stays constant.
- Less Closely Packed Particles than Solids: Particles in a liquid are still close together, but they aren't locked in place. They can slide past one another.
For density and compressibility:
- Moderately High Density: Liquids are less dense than solids but denser than gases. Density can shift slightly with temperature and pressure.
- Slightly Compressible: Because particles have a bit more space between them than in a solid, liquids are slightly compressible.
📣 Density rule exception: Liquid water is actually denser than ice. This is unusual behavior and is the reason ice floats. Most substances are denser in their solid form.
If you melt that ice cube from earlier, the resulting water takes the shape of its container, but the volume of water remains the same.
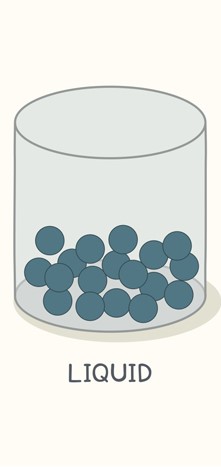
Particle representation of a liquid.
Image Courtesy of Live Science
⛽ Gases
Gases are the least restricted state of matter:
- Indefinite Shape and Volume: Gases expand to fill the entire volume of their container, adopting both its shape and volume.
- Widely Spaced Particles: Gas particles are spread far apart compared to solids and liquids.
- Rapid and Random Movement: Gas particles move quickly in all directions, which is why gases diffuse (spread out) to fill any available space.
Their two key properties:
- Low Density: The large distances between particles give gases much lower densities than solids or liquids.
- Highly Compressible: All that empty space between particles means gases can be compressed significantly.
Water vapor is hard to see, but you can observe gas behavior with something like a balloon. The air inside expands to fill the balloon completely, and you can squeeze the balloon to compress the gas into a smaller volume.

Particle representation of a gas.
Image Courtesy of Live Science
🔄 Phase Changes
Phase changes are transitions between states of matter. Every phase change involves either adding energy to or removing energy from a substance.
Here are the six phase changes you need to know:
- Melting: solid → liquid (requires energy)
- Freezing: liquid → solid (releases energy)
- Vaporization: liquid → gas (requires energy); includes both boiling and evaporation
- Condensation: gas → liquid (releases energy)
- Sublimation: solid → gas directly, skipping the liquid phase (requires energy)
- Deposition: gas → solid directly, skipping the liquid phase (releases energy)
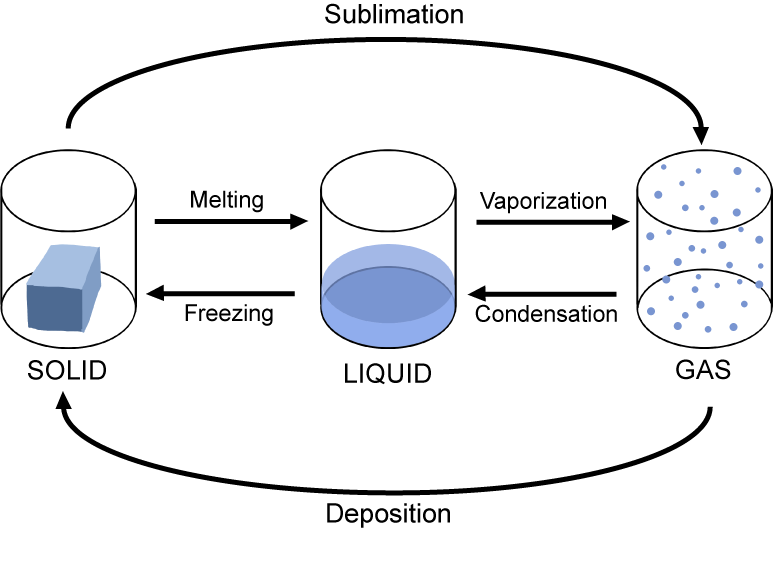
Three separate containers displaying the different phase changes between a solid, liquid, and gas.
Image Courtesy of Shmoop
Why does melting require energy? Think about holding an ice cube. The heat from your hand transfers to the ice, giving its molecules enough energy to break free from their fixed positions and flow as a liquid. That's what "requires energy" means: the substance must absorb energy for the change to happen.
Freezing works the opposite way. When you put water in a freezer, the cold environment pulls heat energy away from the water molecules. As they lose energy, they slow down and lock into a fixed, structured arrangement, forming solid ice. That's what "releases energy" means: the substance gives off energy to its surroundings during the change.
A helpful pattern to remember: transitions toward more particle freedom (solid → liquid → gas) require energy, while transitions toward less freedom (gas → liquid → solid) release energy.
🖇️ Physical vs Chemical Properties
Every substance has physical and chemical properties. You'll explore these in much greater depth when you study bonding later, but here's what you need to know now.
📄 Physical Properties
Physical properties can be observed or measured without changing the substance's chemical identity. The substance remains the same thing before and after you observe the property.
- Color
- Odor
- Density
- Melting point
- Boiling point
For example, measuring the mass of a sample of iron or boiling water to observe it turn to steam are both physical observations. The iron is still iron, and the water is still .
🔥 Chemical Properties
Chemical properties describe a substance's ability to undergo changes that transform it into different substances. You can only observe a chemical property when a chemical change actually occurs.
- Reactivity with other chemicals (e.g., iron rusting when exposed to oxygen)
- Flammability (e.g., paper burning)
- Oxidation state
Combining vinegar with baking soda is a classic example: the bubbling you see is gas being produced, which is a completely new substance. That's a chemical change.
🤔 Categorizing Matter by Composition
Beyond classifying matter by state (solid, liquid, gas), chemists also classify it by composition: what is it made of at the atomic level?
Pure Substances
A pure substance has a uniform and definite composition. It contains only one type of particle throughout. Pure substances fall into two categories:
- 🪙 Elements: A pure substance made of only one kind of atom. Every element appears on the periodic table. Carbon (C), gold (Au), and oxygen (O) are all elements. You cannot break an element down into simpler substances by any chemical means.
- 🌊 Compounds: A pure substance made of two or more elements chemically bonded in a fixed ratio. Water is always , meaning two hydrogen atoms for every one oxygen atom. Carbon dioxide is always . Because compounds involve chemical bonds, they can only be broken down into their elements through chemical reactions, not physical methods.
Mixtures
A mixture contains two or more substances that are physically combined but not chemically bonded. Each substance in the mixture keeps its own chemical identity and properties.
Mixtures are categorized by how uniformly their components are distributed:
- 💨 Homogeneous mixtures: These have a uniform composition throughout. The components are evenly distributed and can't be visually distinguished from one another.
- Atmospheric air is a homogeneous mixture of gases like and . You can't look at air and tell where the nitrogen ends and the oxygen begins.
- 🥗 Heterogeneous mixtures: These do not have a uniform composition. The components are unevenly distributed and can often be physically identified and separated.
- A salad is a heterogeneous mixture. You can see the lettuce, tomatoes, and croutons as distinct pieces and pick them apart.
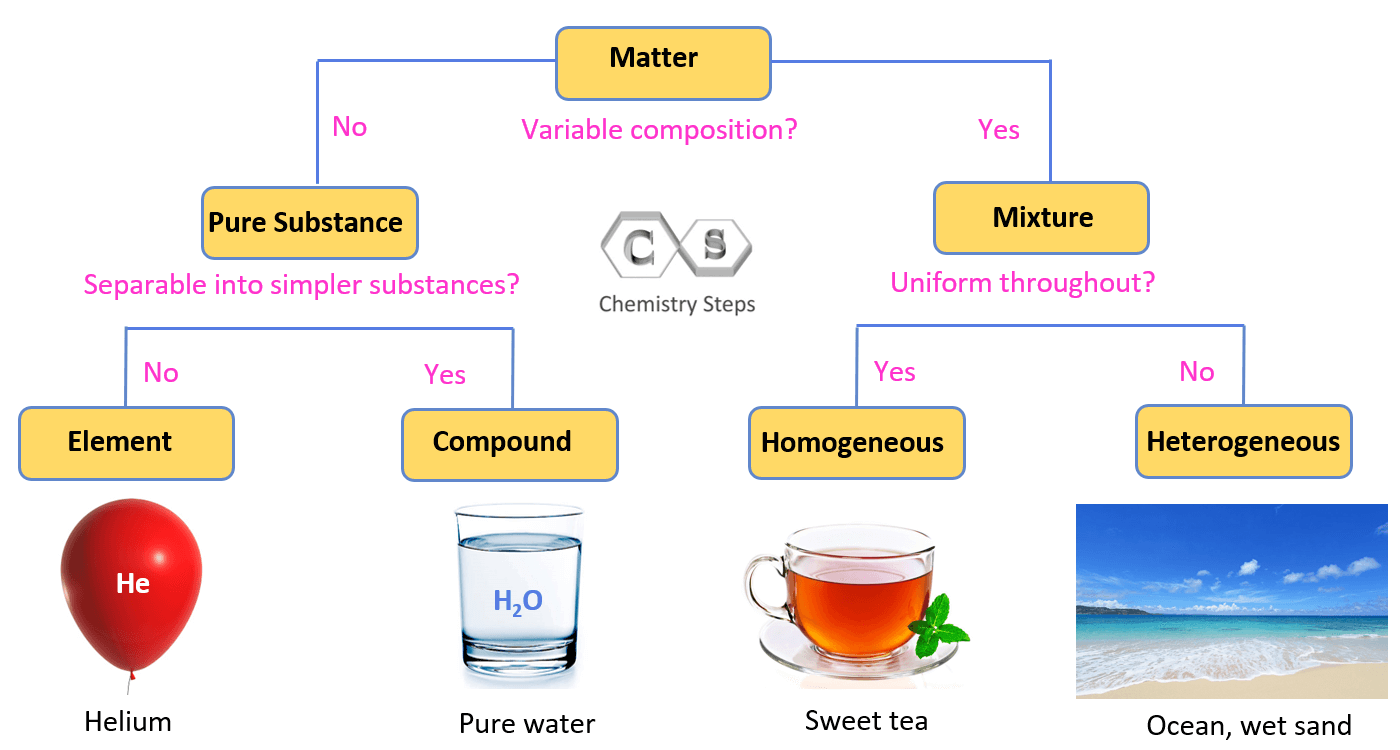
Image displaying how to categorize matter by composition.
Image Courtesy of General Chemistry Steps
🗂️ Purity & Separation Methods
Chemists frequently need to separate mixtures into their individual components. The method you choose depends on the physical properties of the substances in the mixture.
The three most common separation techniques are:
- Filtration — separates solids from liquids based on particle size
- Distillation — separates liquids based on differences in boiling points
- Chromatography — separates components based on how they move through a medium
Filtration
Filtration uses a porous barrier (like filter paper) to separate solid particles from a liquid. The liquid passes through the tiny pores, but the solid particles are too large to fit through and get trapped.
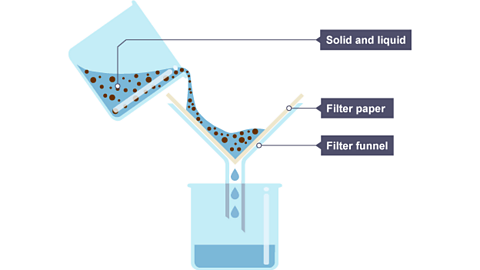
One beaker filled with solid and liquid mixture, and that is filtered through a filter paper and filter funnel into another beaker that is collecting water.
Image Courtesy of BBC.
A sand-and-water mixture is a straightforward example. Pour it through filter paper, and the sand gets caught while the water flows through.
Distillation
Distillation separates a liquid mixture by exploiting differences in boiling points. Here's how it works:
- Heat the mixture until the component with the lowest boiling point vaporizes first.
- The vapor travels into a condenser, where it cools and turns back into a liquid.
- Collect that condensed liquid separately.
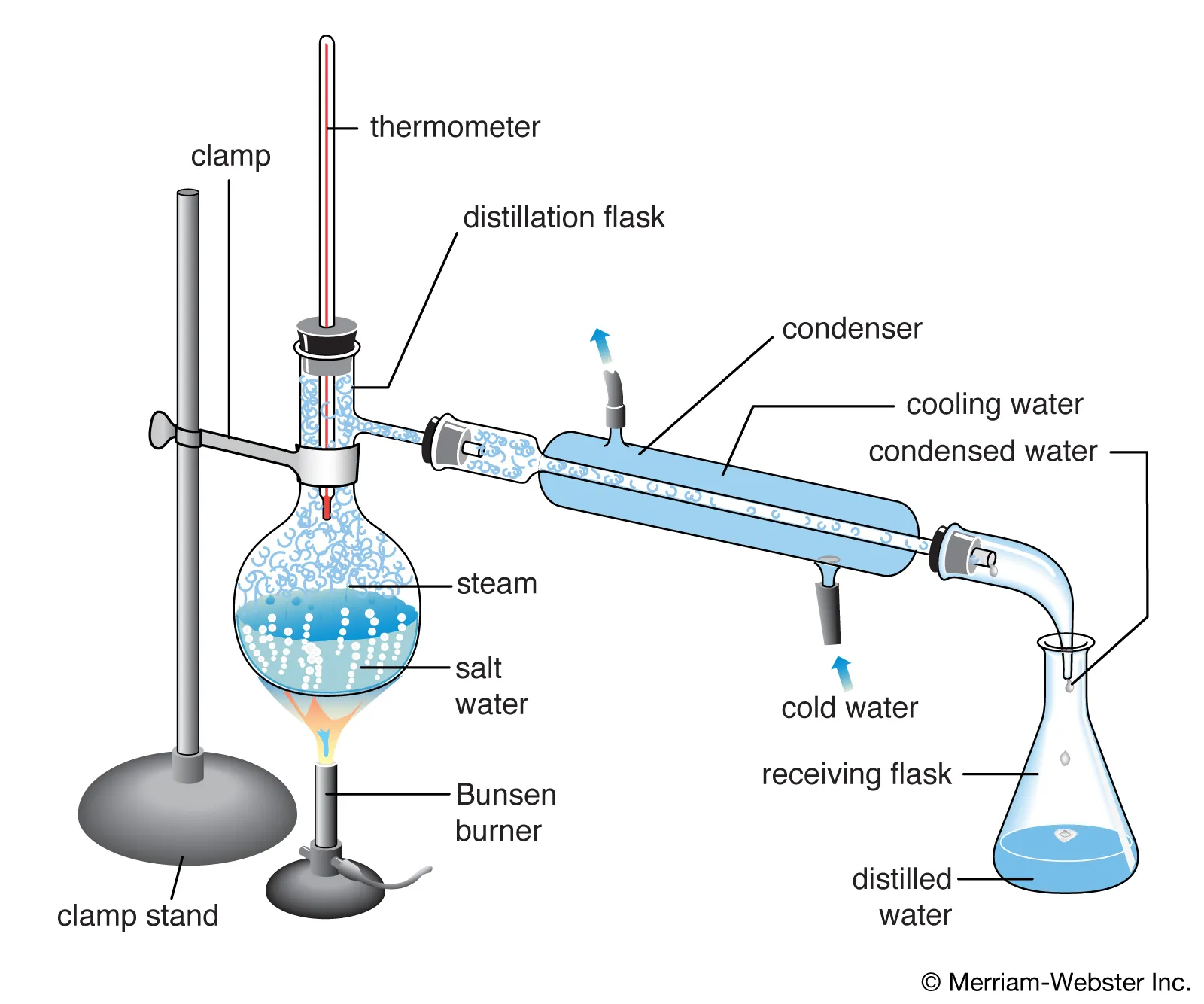
Image displaying how distillation works.
Image Courtesy of Britannica.
For a water-and-ethanol mixture, ethanol boils at about 78°C while water boils at 100°C. Heating the mixture causes the ethanol to vaporize first, so the collected vapor is enriched in ethanol.
Chromatography
Chromatography separates components in a mixture based on how they interact with two phases: a stationary phase (which stays in place) and a mobile phase (which moves through or across the stationary phase). Different components travel at different speeds, causing them to separate.
Common types include:
- Paper Chromatography: Paper serves as the stationary phase; a solvent moves up the paper as the mobile phase.
- Thin-Layer Chromatography (TLC): A thin coating of material (like silica gel) on a plate acts as the stationary phase.
- Gas Chromatography (GC): Separates volatile substances using a gas as the mobile phase.
- Liquid Chromatography (LC): Uses a liquid mobile phase passing through a column containing the stationary phase.
For this class, you'll most likely only need to understand paper or thin-layer chromatography. Check with your instructor on which types they expect you to know.
⭐ Closing
You now have a solid framework for understanding matter: its states, how it changes between them, and how to classify and separate it. These concepts are the building blocks for everything else in chemistry, so make sure you're comfortable with the vocabulary and the reasoning behind each classification before moving on.