Periodic Table Trends
The periodic table isn't just organized by atomic number. Its structure reveals predictable patterns in how elements behave, from how big their atoms are to how strongly they attract electrons. These trends come down to two competing factors: nuclear charge (how many protons pull on electrons) and electron shielding (how inner electrons block that pull from reaching outer electrons).
This guide covers atomic radius, ionization energy, electronegativity, ionic size, and how you can use these trends to predict reactivity and bonding.

Trends Across Periods and Groups
Atomic Radius
Across a period (left to right): Atomic radius decreases. Each element adds a proton to the nucleus and an electron to the same energy level. Since electrons in the same shell don't shield each other very well, the growing nuclear charge pulls all the electrons in tighter. For example, sodium (Na) has a larger atomic radius than chlorine (Cl), even though they're in the same period.
Down a group (top to bottom): Atomic radius increases. Each row adds a new electron shell, placing valence electrons farther from the nucleus. The inner shells also shield the valence electrons from the full nuclear charge. That's why cesium (Cs) is much larger than lithium (Li).
Ionization Energy
Ionization energy is the energy required to remove the outermost electron from a gaseous atom.
Across a period: Ionization energy increases. The stronger nuclear charge holds electrons more tightly, so it takes more energy to pull one away. Neon requires far more energy to ionize than sodium.
Down a group: Ionization energy decreases. Valence electrons sit farther from the nucleus and are shielded by more inner shells, so they're easier to remove. Francium has a much lower first ionization energy than lithium.
Electronegativity
Electronegativity measures how strongly an atom attracts electrons in a chemical bond (using the Pauling scale).
Across a period: Electronegativity increases. Higher nuclear charge means atoms pull harder on shared electrons. Fluorine (3.98) is the most electronegative element on the table.
Down a group: Electronegativity decreases. Greater distance and more shielding weaken the nucleus's pull on bonding electrons. Iodine is less electronegative than fluorine for this reason.
The unifying idea: All three trends trace back to the same two factors. Across a period, increasing nuclear charge dominates (electrons are in the same shell, so shielding barely changes). Down a group, increasing distance and shielding dominate (even though nuclear charge also grows, it can't compensate).
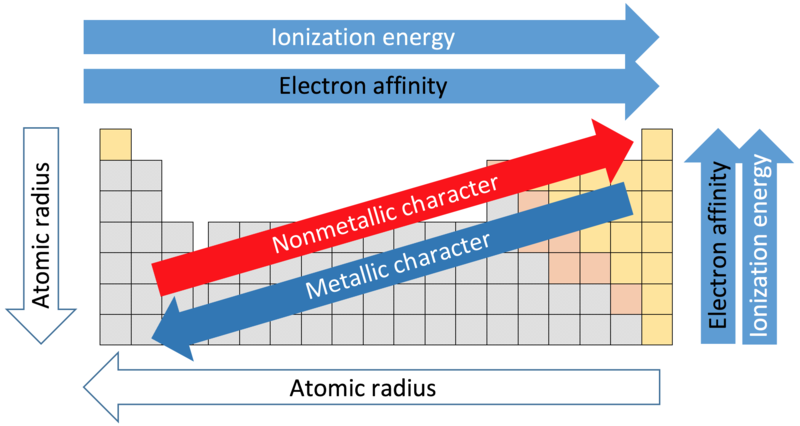
An image showing the overall trends across periods and groups of the periodic table.
Image courtesy of LibreTexts Chemistry
Understanding Periodic Trends
Atomic vs. Ionic Size
When atoms gain or lose electrons to form ions, their size changes:
- Cations (positive ions) are smaller than their parent atom. Losing electrons removes an outer shell (or at least reduces electron-electron repulsion), and the same nuclear charge now pulls fewer electrons in tighter. For example, is significantly smaller than Na because it loses its entire third shell.
- Anions (negative ions) are larger than their parent atom. Extra electrons increase electron-electron repulsion, causing the electron cloud to expand. is larger than Cl because the 18 electrons repel each other more than the original 17 did.
A quick mnemonic: "cations" has a "t" that looks like a plus sign (positive). "anions" starts like "angry" or "negative."
Valence Electrons
Valence electrons are the electrons in the outermost energy level. They determine how an element bonds and reacts.
- For main-group elements (Groups 1-2 and 13-18), the number of valence electrons equals the ones digit of the group number. Group 1 has 1 valence electron; Group 17 has 7.
- Elements in the same group share the same number of valence electrons, which is why they show similar chemical behavior. All alkali metals (Group 1) readily lose one electron; all halogens (Group 17) readily gain one.
Predicting Properties Using Trends
Reactivity Trends
Reactivity depends on whether you're looking at metals or nonmetals, and the reasoning is different for each:
- Metals become more reactive going down a group. Their ionization energy drops, so they lose valence electrons more easily. Potassium reacts explosively with water, while lithium reacts gently.
- Nonmetals become more reactive going up a group. Their electronegativity increases and atomic radius shrinks, so they gain electrons more effectively. Fluorine is the most reactive nonmetal; iodine is comparatively mild.
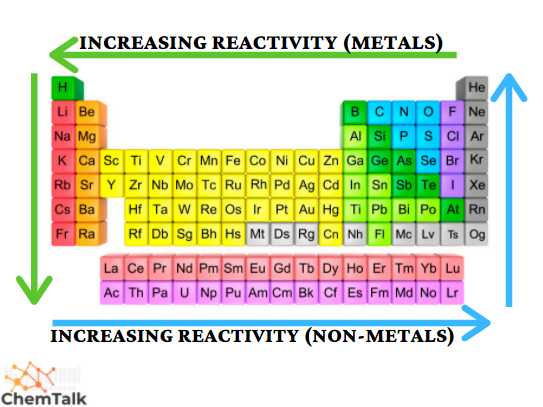
Periodic table with green colored arrows going right to left & going down showing the increasing reactivity for metal elements, and blue arrows going left to right & going up showing the increasing reactivity for non-metal elements.
Image courtesy of Chemistry Talk
Bonding Characteristics
Electronegativity differences between two atoms predict what type of bond they'll form:
- Large difference (generally > 1.7) → Ionic bond (e.g., NaCl: Na has 0.93, Cl has 3.16)
- Small or no difference → Covalent bond (e.g., : both atoms are oxygen)
- Metals bonding with metals → Metallic bond (delocalized electrons shared across a "sea")
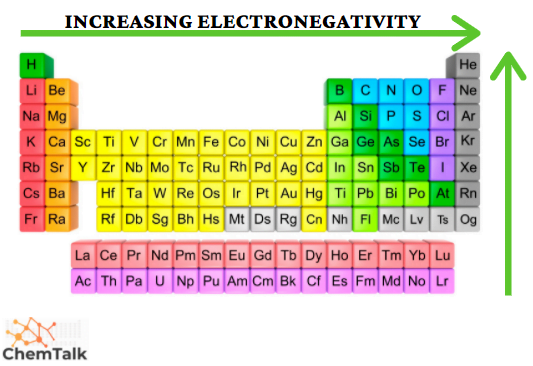
Periodic table with one arrow going left to right and another arrow going up, displaying the increase of electronegativity trend.
Image courtesy of Chemistry Talk
Physical Properties
Periodic trends also connect to physical properties:
- Elements with low ionization energies (metals on the left side) tend to have higher melting and boiling points because metallic bonding is strong. Transition metals in particular can have very high melting points (tungsten melts at 3422°C).
- Smaller atomic radii can lead to higher densities because atoms pack more tightly in the solid state. Osmium, one of the densest elements, sits among the smaller transition metals.
Anomalies and Exceptions
Not every element follows the general trends perfectly:
- Noble gases have exceptionally high ionization energies because their valence shells are completely full, making them very stable. This is why helium has a higher ionization energy than hydrogen despite being to its right.
- Lanthanide contraction: The lanthanides (elements 57-71) have poor shielding from their 4f electrons. This causes elements after the lanthanides (like hafnium) to be unexpectedly small, with atomic radii similar to the elements directly above them in the periodic table.
- There are also small dips in ionization energy within a period. For instance, oxygen has a slightly lower ionization energy than nitrogen because nitrogen's half-filled 2p subshell is extra stable.
Practice Questions
- Explain why fluorine has a higher electronegativity than iodine.
- Predict whether potassium or calcium will react more vigorously with water, based on their positions in the periodic table.
- Why does helium have a higher first ionization energy than hydrogen?
- Is an ion larger or smaller than a neutral oxygen atom? Justify your answer.
- Discuss why silicon is chosen over phosphorus for making semiconductors.
Answers
-
Fluorine has only 2 electron shells compared to iodine's 5. Its valence electrons are much closer to the nucleus with less shielding, so fluorine's nucleus attracts bonding electrons far more strongly. This gives fluorine a higher electronegativity (3.98 vs. 2.66).
-
Potassium is more reactive. Both are in Period 4, but potassium (Group 1) has a lower ionization energy than calcium (Group 2). It loses its single valence electron more easily, making it react more vigorously. Calcium needs to lose two electrons and holds them more tightly due to its higher nuclear charge.
-
Helium has two protons pulling on two electrons that occupy a full 1s shell. That full-shell stability, combined with a higher nuclear charge than hydrogen's single proton, makes it much harder to remove an electron from helium. Helium's first ionization energy (2372 kJ/mol) is nearly double hydrogen's (1312 kJ/mol).
-
is larger than neutral oxygen. Oxygen normally has 8 electrons, but has 10. The two extra electrons increase electron-electron repulsion while the nuclear charge (8 protons) stays the same, so the electron cloud expands outward.
-
Silicon is a metalloid with four valence electrons, giving it intermediate electrical conductivity that can be precisely controlled through doping (adding small amounts of other elements). Phosphorus has five valence electrons and behaves more like a nonmetal, lacking the tunable conductivity that semiconductor applications require. Interestingly, phosphorus is actually used as a dopant added to silicon to modify its conductivity.