Lewis Structures and Molecular Geometry
Lewis dot structures and molecular geometry give you the tools to predict how atoms bond and what shapes molecules take in three dimensions. These concepts connect directly to understanding why substances behave the way they do, from their polarity to their reactivity to their physical properties.

Lewis Dot Structures
Lewis dot structures are diagrams that show the valence electrons of atoms within a molecule. Each dot represents one valence electron, and lines drawn between two atoms represent shared (bonding) electron pairs. These structures help you see at a glance how electrons are distributed and where bonds form.
Valence Electrons
Valence electrons are the electrons in an atom's outermost energy level. They're the ones involved in bonding because they're the most accessible to other atoms.
In a Lewis dot structure, you place dots around an element's chemical symbol to represent its valence electrons. The number of valence electrons corresponds to the element's group number on the periodic table (for main-group elements). For example, carbon (Group 4A) has 4 valence electrons, and oxygen (Group 6A) has 6.
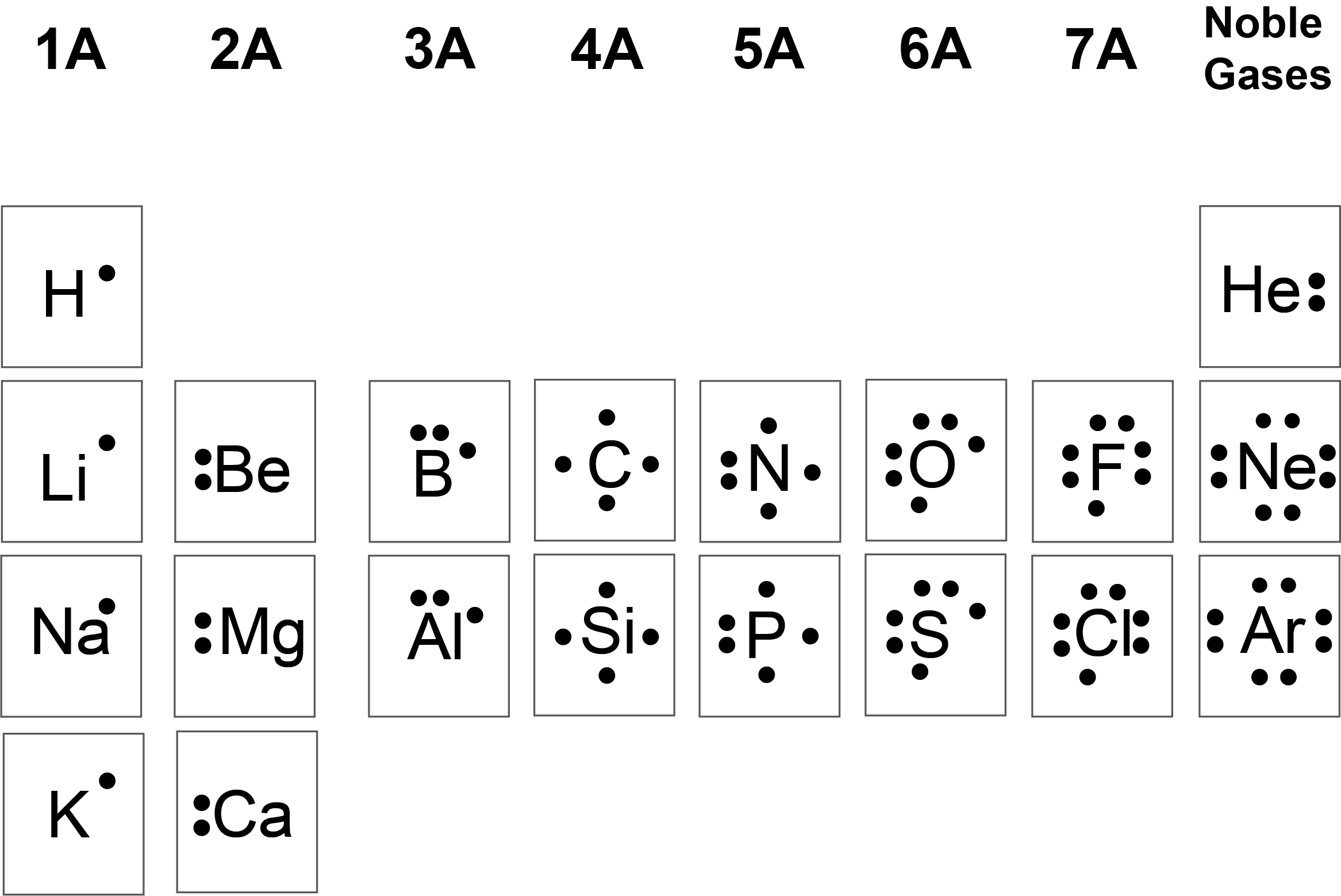
An image showing valence electrons as dots around elemental symbols.
Image courtesy of eCampusOntario
Drawing Lewis Structures
Follow these steps to draw a Lewis structure:
- Count the total valence electrons. Add up the valence electrons from every atom in the molecule. For polyatomic ions, add one electron for each negative charge or subtract one for each positive charge.
- Identify the central atom. Pick the least electronegative atom as the center. Hydrogen is never the central atom (it can only form one bond). Carbon is almost always central when present.
- Draw the skeletal structure. Connect each outer atom to the central atom with a single bond (one line = 2 electrons).
- Distribute remaining electrons as lone pairs. Start with the outer atoms first, giving each an octet (except hydrogen, which only needs 2). Then place any leftover electrons on the central atom.
- Form multiple bonds if needed. If the central atom still lacks an octet after step 4, convert lone pairs from outer atoms into double or triple bonds until the central atom has eight electrons.
Resonance Structures
Some molecules can't be accurately represented by a single Lewis structure. When you can draw two or more valid structures that differ only in the placement of electrons (not atoms), those are called resonance structures. The actual molecule is a blend of all resonance forms. Ozone () and the nitrate ion () are classic examples.
Formal Charge
Formal charge helps you figure out which Lewis structure is the most stable when multiple arrangements are possible. Calculate it with:
The best Lewis structure is the one where formal charges are minimized (closest to zero) and any negative formal charges sit on the more electronegative atoms.
The Octet Rule
The octet rule states that atoms tend to bond in ways that give them eight valence electrons in their outer shell, achieving the same electron configuration as a noble gas. This arrangement is especially stable.
Hydrogen is the obvious small exception: it only needs 2 electrons to fill its outer shell (matching helium).
Exceptions to the Octet Rule
Not every molecule obeys the octet rule. There are three main categories of exceptions:
- Incomplete octets. Some elements are stable with fewer than eight electrons. Beryllium (Be) commonly has only 4 electrons in its compounds, and boron (B) is stable with 6. For example, in boron trifluoride (), boron has only 6 electrons around it.
- Expanded octets. Elements in Period 3 and beyond have access to empty d orbitals, which lets them accommodate more than 8 electrons. Phosphorus in (10 electrons) and sulfur in (12 electrons) are common examples. Only elements in Period 3+ can do this.
- Odd-electron molecules. If a molecule has an odd total number of valence electrons, it's impossible for every atom to have a complete octet. Nitric oxide (), with 11 valence electrons, is a classic example. These molecules are also called free radicals.
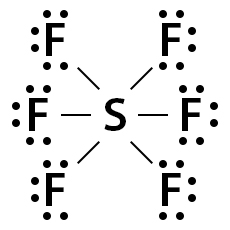
Sulfur hexafluoride Lewis dot structure.
Image Courtesy of Chemistry Learner
Molecular Geometry
The three-dimensional shape of a molecule affects its polarity, reactivity, and physical properties like melting and boiling points. To predict shape, you need VSEPR theory.
VSEPR Theory
Valence Shell Electron Pair Repulsion (VSEPR) theory says that electron groups (bonds and lone pairs) around a central atom repel each other and arrange themselves as far apart as possible. This repulsion determines the molecule's geometry.
To use VSEPR:
- Draw the Lewis structure of the molecule.
- Count the total number of electron domains (also called electron groups) around the central atom. A single bond, double bond, triple bond, or lone pair each count as one domain.
- Determine the electron-domain geometry based on the total number of domains.
- Determine the molecular geometry based on only the positions of the atoms (lone pairs are "invisible" in the molecular shape, but they still push bonding pairs around).
The distinction between electron-domain geometry and molecular geometry matters. For example, water () has 4 electron domains (2 bonding pairs + 2 lone pairs), giving it a tetrahedral electron-domain geometry but a bent molecular geometry.
Molecular Geometry Types
Key geometries based on electron domains around the central atom:
| Electron Domains | Bonding Pairs | Lone Pairs | Molecular Geometry | Bond Angle |
|---|---|---|---|---|
| 2 | 2 | 0 | Linear | 180° |
| 3 | 3 | 0 | Trigonal Planar | 120° |
| 3 | 2 | 1 | Bent | ~117° |
| 4 | 4 | 0 | Tetrahedral | ~109.5° |
| 4 | 3 | 1 | Trigonal Pyramidal | ~107° |
| 4 | 2 | 2 | Bent | ~104.5° |
| 5 | 5 | 0 | Trigonal Bipyramidal | 90° & 120° |
| 6 | 6 | 0 | Octahedral | 90° |
Less common geometries that arise when lone pairs are present on 5- or 6-domain central atoms include seesaw, T-shaped, square pyramidal, and square planar.
Notice that lone pairs compress bond angles slightly below the ideal values because lone pairs repel more strongly than bonding pairs.
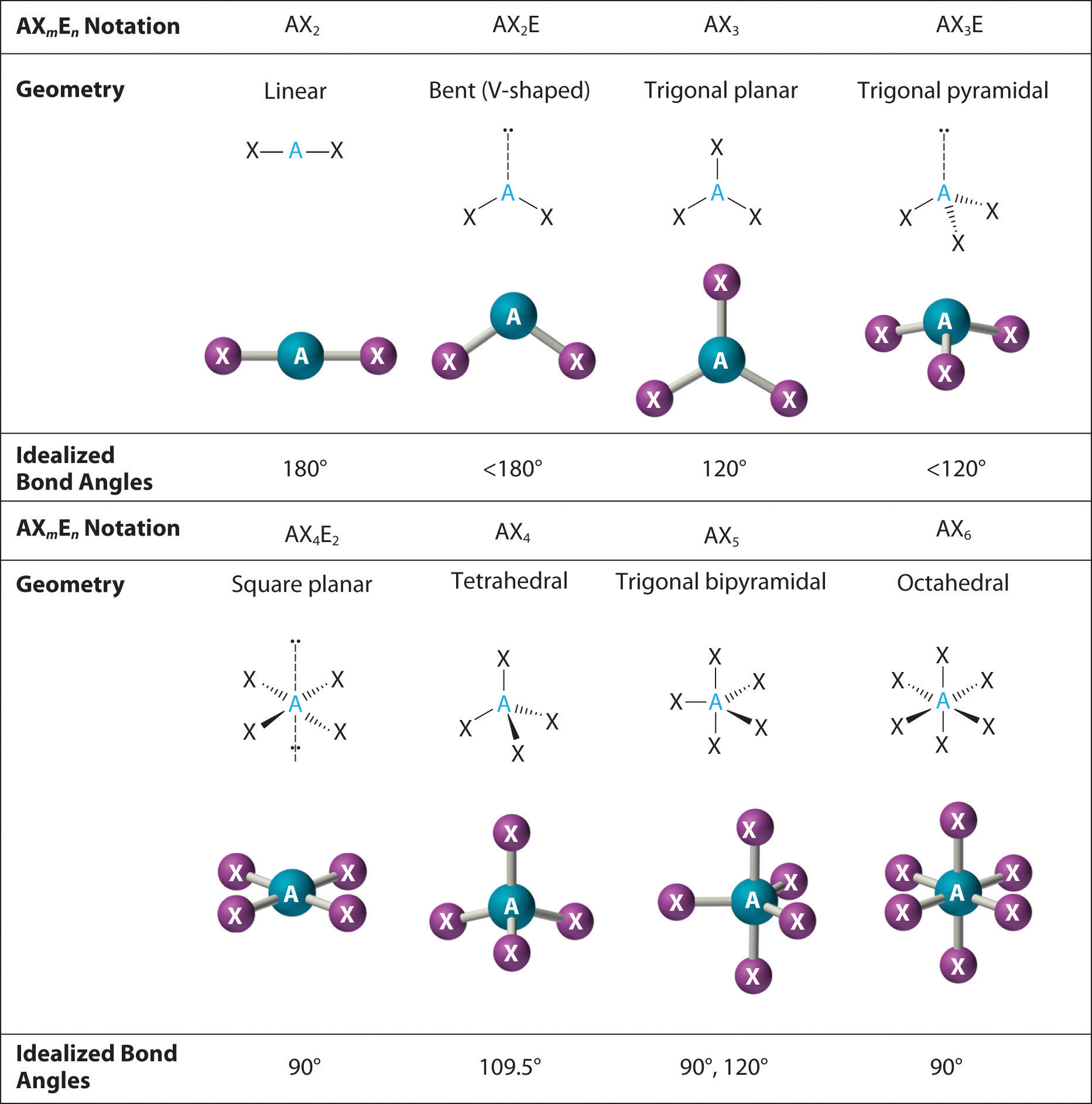
Diagrams showing examples of each geometry type with specified bond angles.
Image Courtesy of 2012Books
Relationship Between Molecular Shape and Properties
Molecular shape directly determines whether a molecule is polar or nonpolar. A molecule is polar when its bond dipoles don't cancel out due to asymmetric geometry. For example, is linear and nonpolar (the two dipoles cancel), while is bent and polar (the two dipoles don't cancel).
Polarity then affects intermolecular forces: polar molecules experience dipole-dipole interactions, while nonpolar molecules rely on weaker London dispersion forces. These intermolecular forces determine macroscopic properties like boiling point, solubility, and state of matter at room temperature. Molecular shape also plays a role in biological function, since enzymes and proteins depend on precise 3D geometry to interact with other molecules.
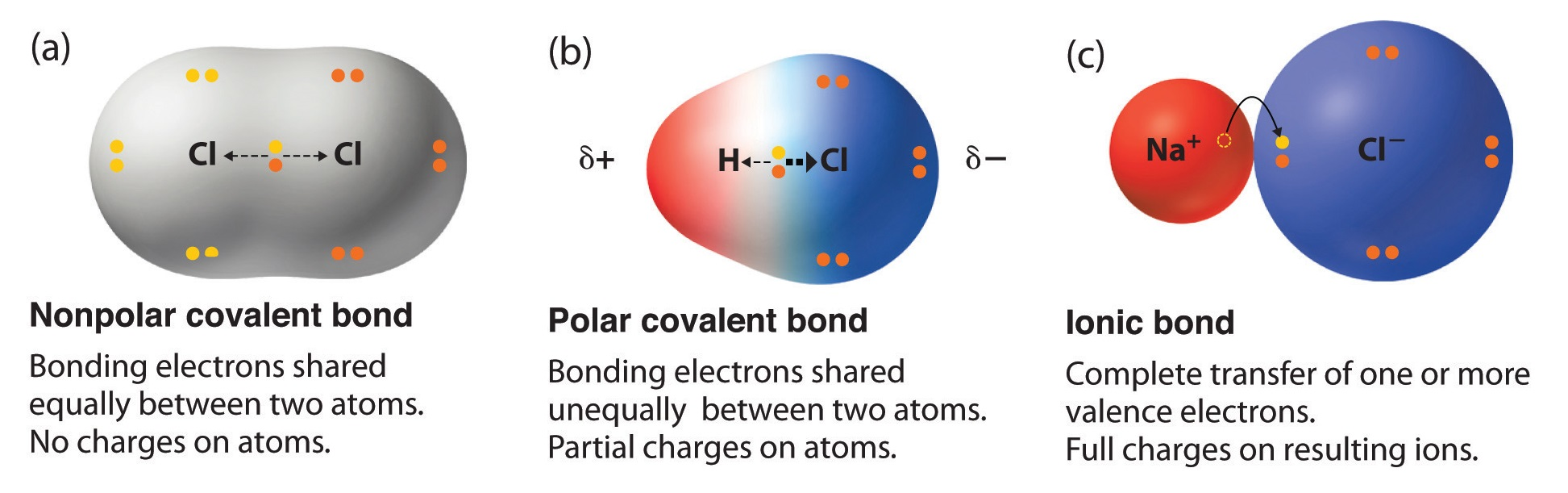
Visual comparison of polar versus nonpolar molecules.
Image Courtesy of LibreTexts
Advanced Applications
These topics build on Lewis structures and VSEPR:
- Hybridization Theory: The central atom's electron domains correspond to hybrid orbitals (, , , , ), which explain the geometry from an orbital perspective. Two domains = , three = , four = , and so on.
- Molecular Orbital Theory: An alternative bonding model that describes electrons as occupying orbitals spread over the entire molecule rather than localized between two atoms.
- Resonance: Drawing and evaluating resonance structures using formal charge to determine relative stability.
Guided Practice Questions
1. Draw the complete Lewis structure for methanol ().
Step 1: Count total valence electrons.
- Carbon: 4
- Hydrogen (×4 total): 4 × 1 = 4
- Oxygen: 6
- Total: 14 valence electrons
Step 2: Identify the central atom. Carbon is the central atom (least electronegative, excluding hydrogen). Oxygen connects to carbon and one hydrogen.
Step 3: Draw the skeletal structure and connect atoms with single bonds. Place carbon in the center, bonded to three hydrogens and one oxygen. The oxygen is also bonded to one hydrogen. That's 5 single bonds = 10 electrons used.
</>CodeH | H - C - O - H | H
Step 4: Distribute remaining electrons. 14 total − 10 used in bonds = 4 electrons remaining. Place them as 2 lone pairs on oxygen.
Step 5: Check octets and formal charges.
- Carbon has 4 bonds = 8 electrons (octet satisfied).
- Oxygen has 2 bonds + 2 lone pairs = 8 electrons (octet satisfied).
- Each hydrogen has 1 bond = 2 electrons (duet satisfied).
- All formal charges are zero. The structure is complete.
</>CodeH | H - C - O - H | ‥ H
(The two dots on oxygen represent its two lone pairs.)
2. Explain why does not follow the octet rule. Discuss the implications for its structure and properties.
Phosphorus is in Period 3, so it has access to empty 3d orbitals. In , phosphorus forms 5 bonds (one to each fluorine), giving it 10 electrons around it. This is an expanded octet.
Geometry: With 5 bonding pairs and 0 lone pairs, VSEPR predicts a trigonal bipyramidal shape. Three fluorines sit in the equatorial plane (120° apart), and two fluorines occupy axial positions (90° from the equatorial plane).
</>CodeF F \ / F - P - F | F
Polarity: Despite having five polar bonds, the trigonal bipyramidal geometry is symmetric. The bond dipoles cancel out, making a nonpolar molecule overall.
Hybridization: Phosphorus undergoes hybridization to form five equivalent bonding orbitals that accommodate the expanded octet.
Reactivity: The expanded octet allows phosphorus to act as a Lewis acid, accepting electron pairs from other species. This makes reactive toward electron-rich molecules.
Wrapping Up
Lewis structures give you a way to map out electron distribution in molecules, and VSEPR theory lets you predict their 3D shapes from those structures. Together, they connect atomic-level bonding to the macroscopic properties you can observe. Practice drawing structures for a variety of molecules, and always double-check your electron counts, octets, and formal charges.