Introduction to Organic Compounds
Organic chemistry is the study of carbon-containing compounds, and it touches nearly every area of science. Understanding the basics of organic compounds is essential for fields like biochemistry, medicine, environmental science, and engineering.
This guide covers the fundamentals: what makes carbon special, the major classes of organic compounds, how to name hydrocarbons using IUPAC rules, and how different types of hydrocarbons compare.

📚 Basics of Organic Chemistry
Organic chemistry is the study of carbon-containing compounds. But what makes carbon so special that it gets an entire branch of chemistry?
‼️ Carbon's Unique Bonding Characteristics
- Carbon has four valence electrons (recall: valence electrons are in an atom's outermost shell)
- C can form single (C-C), double (C=C), or triple (C≡C) bonds
- C can form long chains or ring structures due to its tetrahedral geometry
Those four valence electrons mean carbon can form four strong covalent bonds, making it incredibly versatile. It bonds with other carbon atoms to create long chains or rings, and it also bonds readily with hydrogen, oxygen, nitrogen, sulfur, and halogens. This versatility is why there are millions of known organic compounds.
Take a look below at polyyne, a molecule that demonstrates some of carbon's unique properties: forming both single and triple bonds while also possessing a ring-like structure.
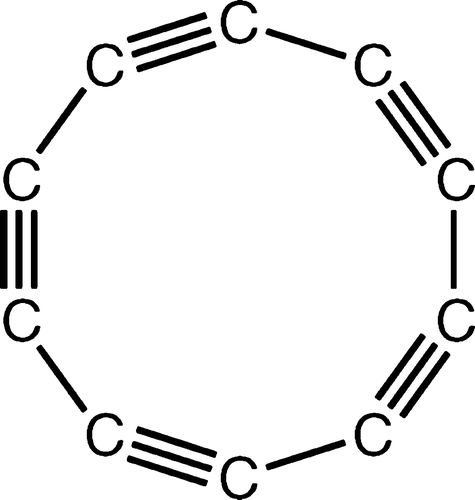
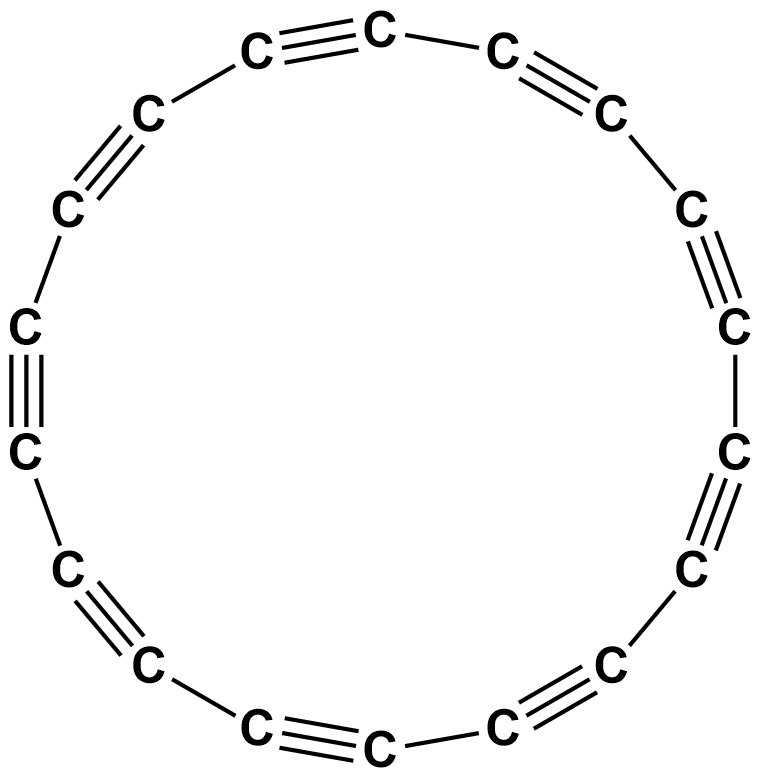
Polyyne molecule.
Image courtesy of American Chemical Society
We define organic compounds as compounds containing carbon atoms bonded to other carbon atoms or to other elements. There are an enormous number of organic compounds, but they can be organized into several key types based on their structure.
Key Types of Organic Compounds
Don't worry too much about the functional groups (e.g., hydroxyl, carbonyl) for now. Those will be covered in the next study guide. For now, just focus on recognizing each class.
- Hydrocarbons contain only carbon and hydrogen atoms. These are the simplest organic compounds and serve as the backbone for many others.
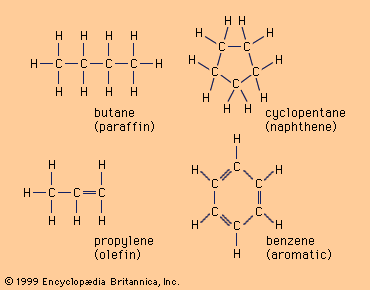
Examples of hydrocarbons.
Image courtesy of Britannica
- Alcohols have one or more hydroxyl (-OH) groups attached to a carbon chain. Ethanol (the alcohol in beverages) is a common example.
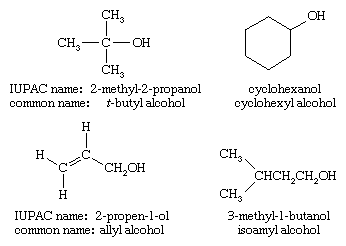
Examples of alcohols.
Image courtesy of Alamy
- Ketones have a carbonyl group (C=O) located within the carbon chain (not at the end).
- Aldehydes have a carbonyl group at the end of the carbon chain.
- Carboxylic acids have a carboxyl group (-COOH). Acetic acid (vinegar) is a familiar example.
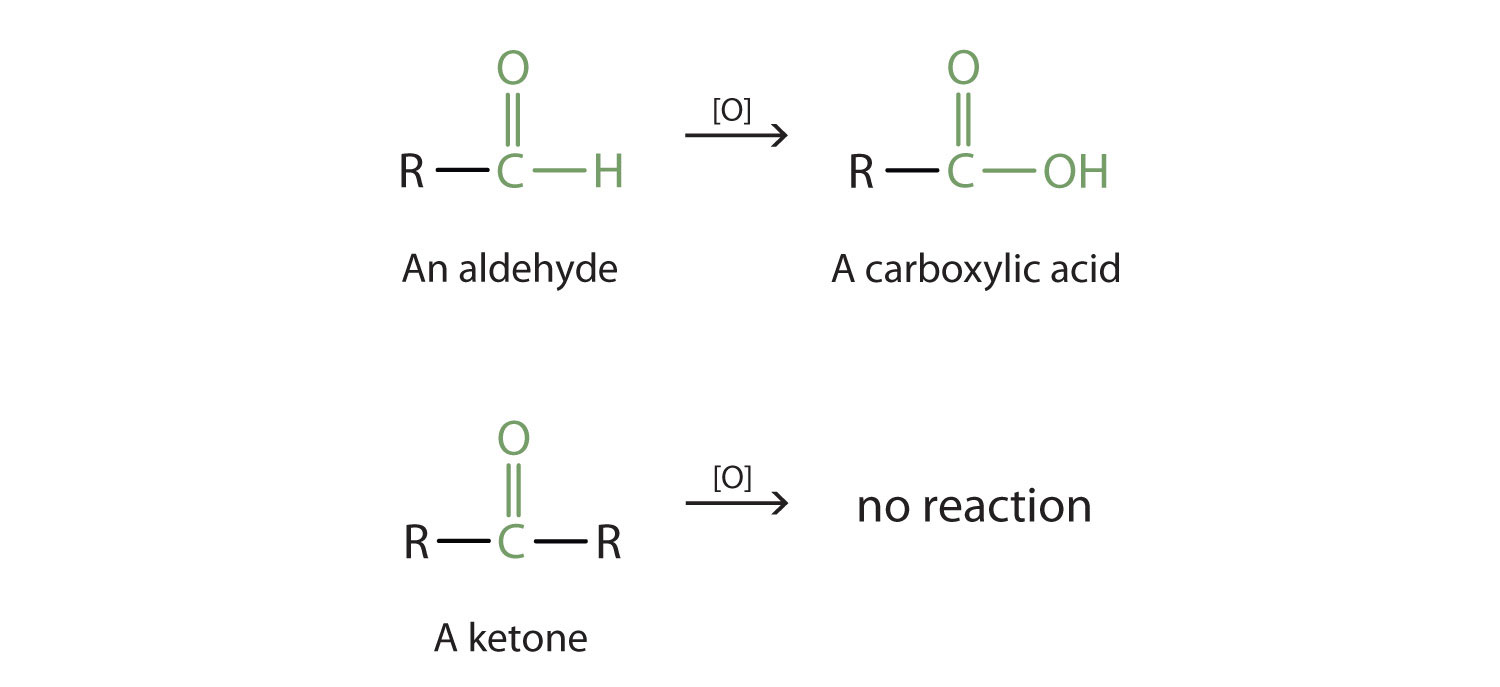
Examples of an aldehyde, ketone, and carboxylic acid.
Image courtesy of Ontario Open Library
Pay attention to where the C=O (or -COOH) sits in each of these three types. That's the key difference between them.
- Amines feature an amino group (-). Notice the recurring -NH- pattern in the structures below.
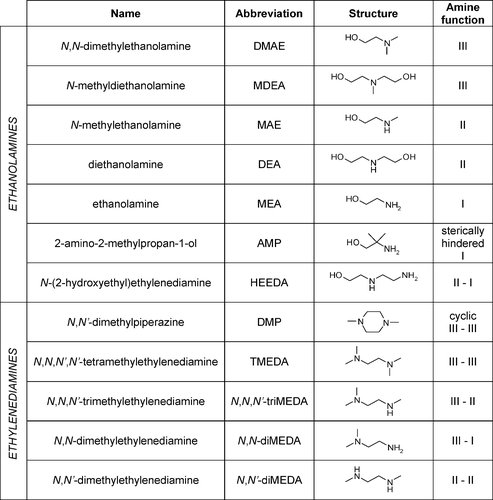
Examples of amines.
Image courtesy of American Chemical Society
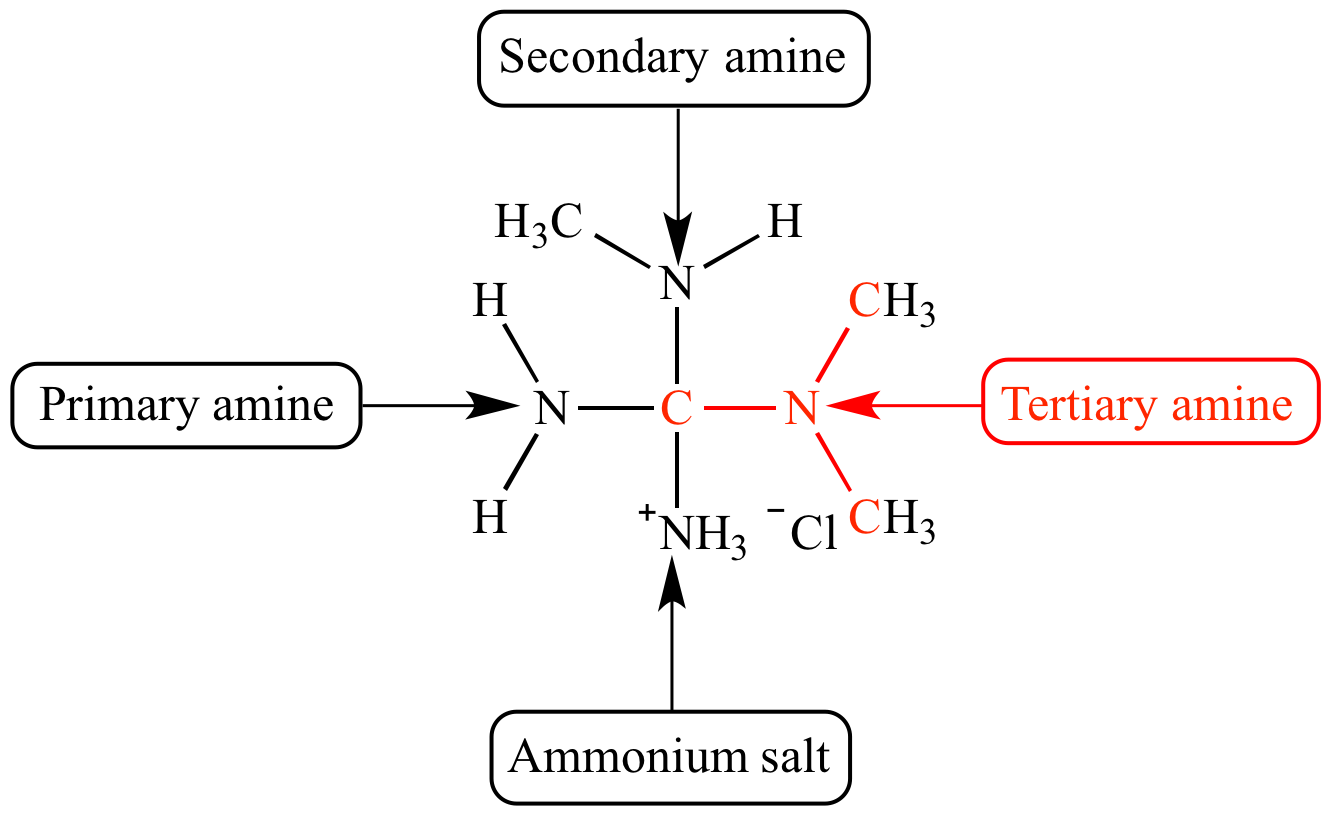
Examples of amines.
Image courtesy of American Chemical Society
Functional Groups
Functional groups are specific groups of atoms within molecules that determine the characteristics and chemical reactivity of those molecules. Each type of organic compound listed above is defined by its functional group. You'll explore these in more detail in the next guide.
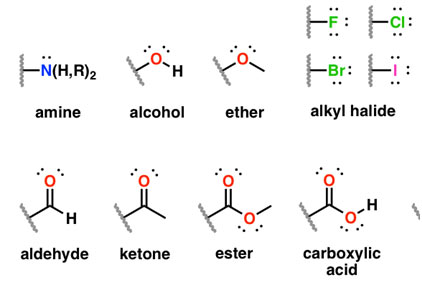
Image displaying different types of functional groups.
Image courtesy of GuyHowto
🧪 Structure & Nomenclature of Hydrocarbons
Structural Formulas vs. Molecular Formulas
These two types of formulas tell you different things about a molecule:
- The molecular formula tells you which atoms are present and how many of each. For example, tells you there are 2 carbon atoms and 6 hydrogen atoms.
- The structural formula shows how those atoms are connected to each other (e.g., a ball-and-stick or line model). This matters because molecules with the same molecular formula can have different structures (more on that in the isomers section).
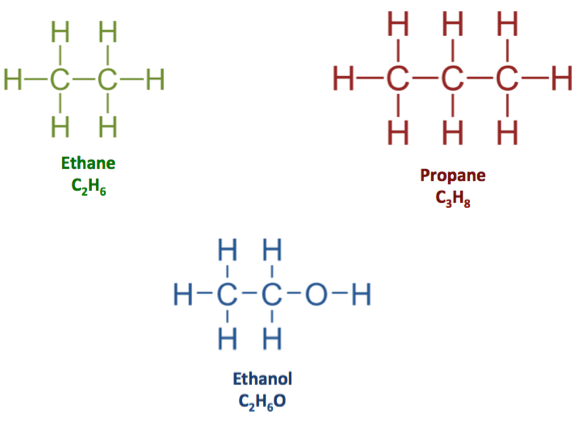
Structural formulas of ethane, propane, and ethanol.
Image courtesy of Inspired Pencil
IUPAC Nomenclature
Nomenclature just means a naming system. The International Union of Pure and Applied Chemistry (IUPAC) provides the standard rules for naming organic compounds. Here's the process:
‼️ Naming Organic Compounds (IUPAC Rules)
- Identify the longest continuous chain of carbon atoms. This determines the base name (meth- = 1, eth- = 2, prop- = 3, but- = 4, pent- = 5, hex- = 6).
- Number the chain starting from the end nearest to any substituent (branch).
- Name each substituent and list them as prefixes in alphabetical order.
- Combine the substituent names with their position numbers, then add the base name.
Here's an example:
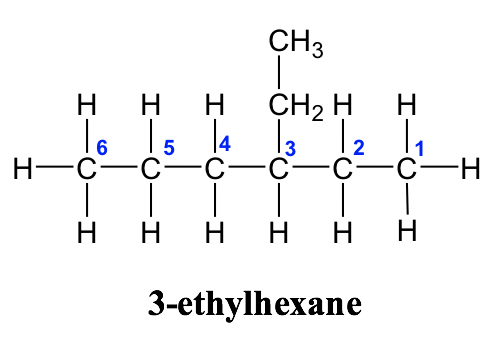
3-ethylhexane molecule.
Image courtesy of Kwantlen Polytechnic University
Walking through the 4-step process:
-
The longest continuous chain has six carbons (6 = hex- prefix). Only single bonds are present, so it's an alkane (-ane suffix).
-
See the blue numbers in the image: numbering from right to left puts the branch at a lower position (carbon 3) than numbering left to right would, so we start from the right.
-
The substituent on the 3rd carbon contains two carbon atoms (), which corresponds to an ethyl group (eth- = 2 carbons).
-
Putting it together: 3-ethylhexane.
Isomerism in Hydrocarbons
One of the things that makes organic chemistry so rich is the existence of isomers: molecules that share the same molecular formula but have different structures. There are two main types you should know:
Structural isomers have the same molecular formula but different connectivity between atoms. The atoms are bonded in a different order.
Structural isomers example
Image courtesy of ThoughtCo
Another structural isomer example
Image courtesy of Shinken
Geometric isomers (cis-trans isomers) have the same connectivity but differ in spatial arrangement around a double bond. Because double bonds restrict rotation, groups can be locked on the same side (cis) or opposite sides (trans) of the bond.
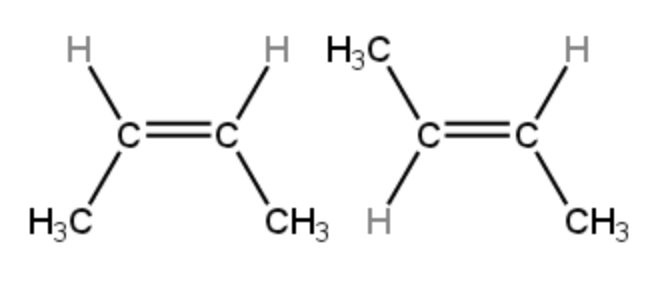
Geometric isomers example
Image courtesy of ChemTalk
🔍 Alkanes, Alkenes, Alkynes & Aromatic Hydrocarbons
Hydrocarbons are classified by the type of carbon-carbon bonds they contain. These bonding differences affect the molecule's geometry, reactivity, and physical properties.
- Alkanes have only single C-C bonds
- Alkenes have at least one double C=C bond
- Alkynes have at least one triple C≡C bond
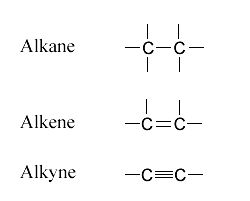
Alkane, alkene, alkyne structures
Image courtesy of Innunco
Alkanes
- Saturated hydrocarbons, meaning every carbon is bonded to the maximum number of hydrogen atoms (only single bonds).
- Nonpolar molecules with relatively low reactivity.
- Key reactions: combustion (exothermic reaction with , producing and ) and substitution reactions (where an H atom is replaced by another atom or group, such as a halogen).
Alkenes
- Unsaturated hydrocarbons containing at least one C=C double bond. "Unsaturated" means they have fewer hydrogen atoms than the maximum possible.
- Exhibit cis-trans isomerism because rotation is restricted around the double bond.
- Undergo addition reactions such as hydrogenation (adding across the double bond) or hydration (adding ).
Alkynes
- Unsaturated hydrocarbons containing at least one C≡C triple bond, which gives them a linear geometry at the triple bond.
- More reactive than alkenes due to the concentration of electron density in the triple bond. They undergo similar addition reactions but may require different catalysts or conditions.
Aromatic Hydrocarbons
Beyond classifying by bond type, there's a special category: aromatic compounds. These are ring structures stabilized by delocalized electrons shared across the ring. Think of the electrons as being spread evenly around the ring rather than locked between two specific atoms.
Benzene () is the classic aromatic compound: a six-membered carbon ring with delocalized electrons. You'll see it drawn two ways: with alternating single/double bonds, or with a circle inside a hexagon (both represent the same thing).
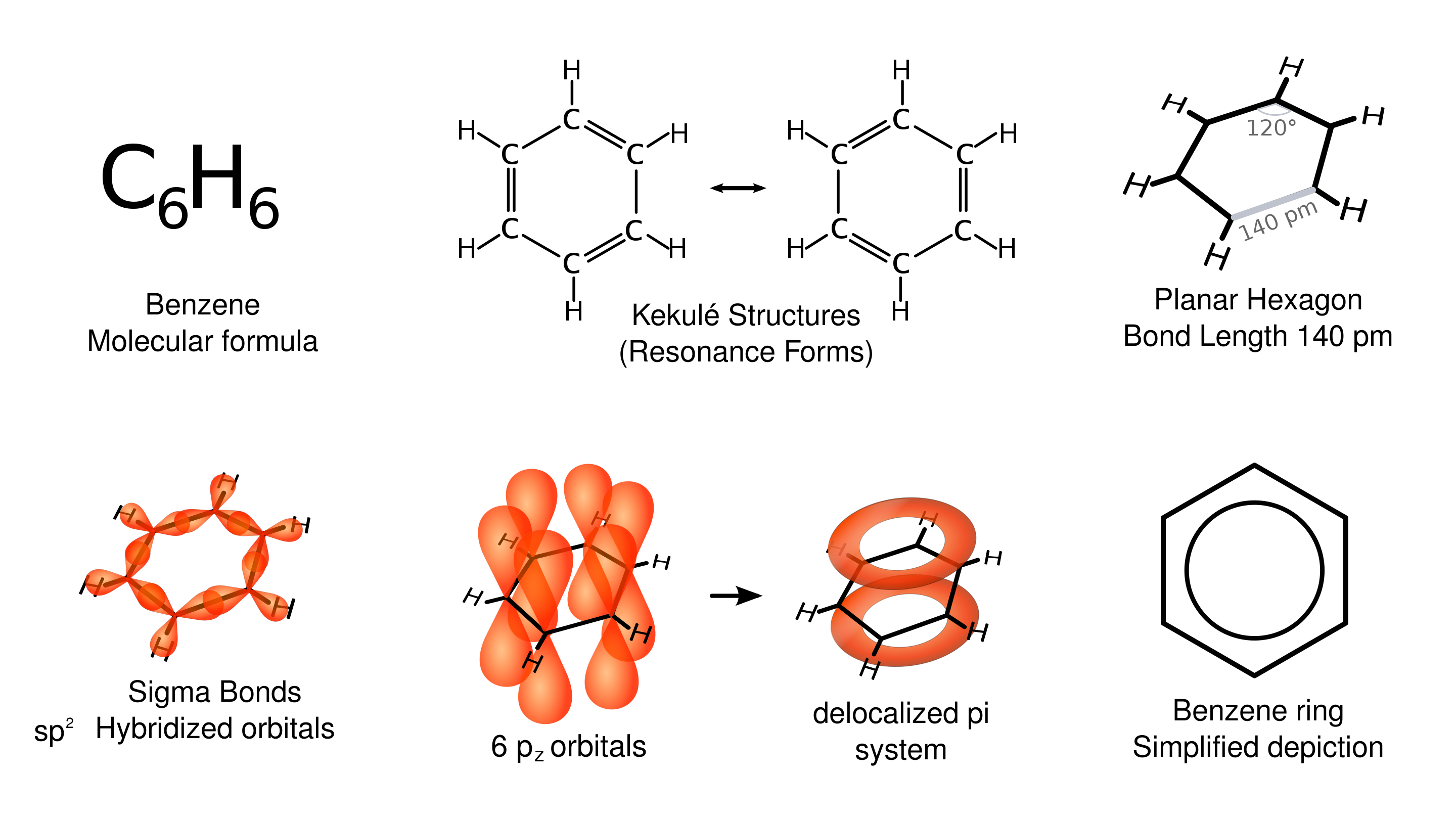
Overview of benzene, a common aromatic compound
Image courtesy of Wikimedia Commons
🌍 Real-world Applications & Implications
Organic compounds, especially hydrocarbons, serve as building blocks for materials and processes across many industries.
- Fuels: Hydrocarbon fuels like gasoline and natural gas release large amounts of energy when combusted. Gasoline powers vehicles, and natural gas heats homes. The combustion of methane, for example:
- Environmental Impact: Burning fossil fuels emits , a greenhouse gas that contributes to global warming. Incomplete combustion also produces pollutants like carbon monoxide and particulates that contribute to smog and poor air quality.
- Petrochemical Industry: Hydrocarbons aren't just burned for energy. Hydrocarbon derivatives are used to manufacture plastics, synthetic fibers, pharmaceuticals, and countless other products.
✏️ Practice Questions
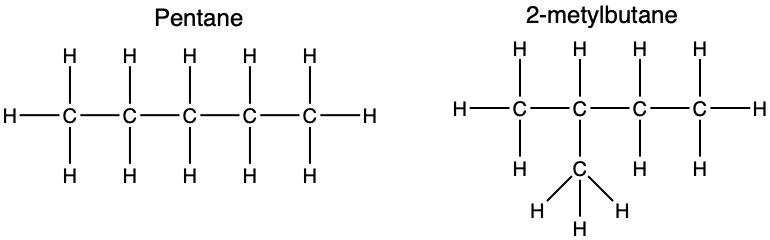
(1) Draw structural formulas for all possible isomers of pentane.
The prefix "pent-" tells you there are five carbons (think pentagon = 5 sides). Since pentane is an alkane, use the formula : that gives hydrogens, so the molecular formula is .
Your job is to find all the different ways to arrange 5 carbons in a chain with single bonds only. Try drawing them yourself, then check against the image below. There are three isomers: n-pentane (straight chain), isopentane (one branch), and neopentane (two branches on the central carbon).
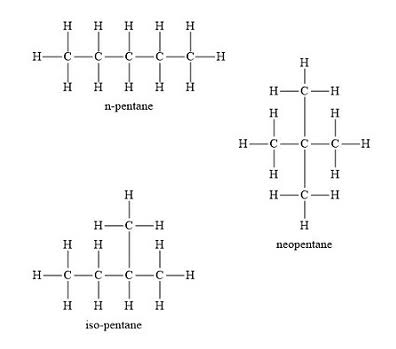
Pentane isomers
Image courtesy of Quora
(2) Name this molecule according to IUPAC rules:
Start by drawing out the structure. Carbons connect to each other along the chain, with hydrogens on the outside. Parentheses indicate branches.
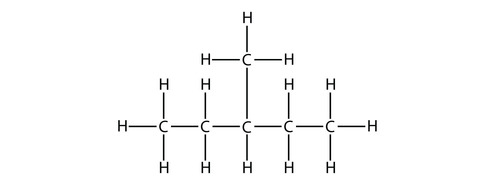
3-methylpentane
Image courtesy of Kathy_Wellman22 (Quizlet)
-
Longest chain: The horizontal chain has five carbons (pent-). All bonds are single, so it's an alkane (-ane).
-
Substituent: There's one branch, which is a methyl group (meth- = 1 carbon).
-
Numbering: The substituent sits on the 3rd carbon from either end (it's in the middle), so numbering from either direction gives the same result.
-
Final name: 3-methylpentane
(3) Why does 1-butene have a lower boiling point than butane even though both have four carbon atoms?
This comes down to intermolecular forces. Butane (, an alkane) is saturated, meaning it has more hydrogen atoms than 1-butene (, an alkene). More atoms means more electrons, which means stronger London dispersion forces (the intermolecular forces that hold nonpolar molecules together).
Because butane has stronger London dispersion forces than 1-butene, you need to add more energy to pull butane molecules apart from each other. That's why butane has a slightly higher boiling point. In general, alkanes have slightly higher boiling points than alkenes with the same number of carbons.
⭐ Conclusion
Organic chemistry has its own vocabulary and set of rules, but once you get comfortable with carbon's bonding behavior, functional groups, and IUPAC naming, the rest builds naturally from there. Practice drawing structures and naming compounds until the process feels automatic.
Chemical structure of ethanol
Image courtesy of Quora