Atomic Theory
This guide traces how our understanding of the atom evolved over roughly 2,500 years, from ancient Greek philosophy to modern quantum mechanics. Knowing this history helps you see why each model was proposed and what evidence forced scientists to revise their thinking.

🤔 Early Philosophical Concepts of Atoms
Around 2,500 years ago, most Greek philosophers believed matter was continuous and infinitely divisible. Democritus disagreed. He introduced the idea of atomos (Greek for "uncuttable"), proposing that if you kept dividing matter, you'd eventually reach a smallest piece that could not be broken down further. These particles, he argued, were indivisible and indestructible.

Democritus' atomic model.
Image Courtesy of Sutori.
Despite this insight, Democritus' idea didn't catch on. Aristotle proposed instead that all matter was composed of four elements: earth, air, fire, and water. Because of Aristotle's enormous influence, this four-element view dominated Western thought for nearly 2,000 years, and the concept of atoms was largely set aside. 🌎
💫 The Chemical Revolution
By the 18th century, chemistry had matured into a formal, experimental science. Two foundational laws emerged that pointed toward the existence of atoms:
- ⚖️ Law of Conservation of Mass (Antoine Lavoisier): In a chemical reaction, the total mass of the reactants equals the total mass of the products. Mass is neither created nor destroyed. This hinted that reactions involve rearranging indestructible particles rather than creating or destroying matter.
- ➗ Law of Definite Proportions (Joseph Proust): A given chemical compound always contains the same elements in the same mass ratio. For example, water is always 11.2% hydrogen and 88.8% oxygen by mass. This suggested that compounds form from atoms combining in fixed ways.
Both laws are still central to the stoichiometry calculations you'll do throughout this course.
🎱 Dalton's Atomic Theory (1803)
John Dalton built on these laws to propose a formal atomic theory, sometimes called the billiard ball model because he pictured atoms as tiny, solid spheres. His key postulates:
- All matter is made up of tiny, indivisible atoms.
- Atoms of a given element are identical in mass and properties; atoms of different elements differ.
- Atoms combine in whole-number ratios to form compounds (confirming Proust's law).
- In chemical reactions, atoms are rearranged but never created or destroyed (confirming Lavoisier's law).
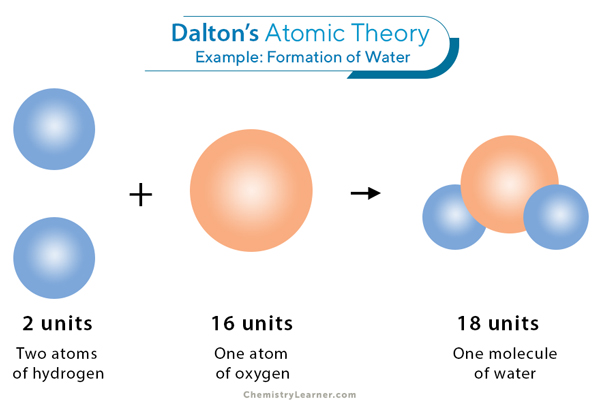
Atoms forming a molecule of water demonstrating Dalton's atomic theory.
Image courtesy of Chemistry Learner
Dalton's theory was a huge step forward, but it needed refinement. Later experiments showed that atoms could be subdivided into smaller particles, and that atoms of the same element don't always have identical masses (these variants are called isotopes, which you'll study soon).
⚛️ Discoveries in Atomic Structure
🍮 J.J. Thomson (1897)
Thomson used a cathode ray tube to discover the electron, proving that atoms were not indivisible after all.
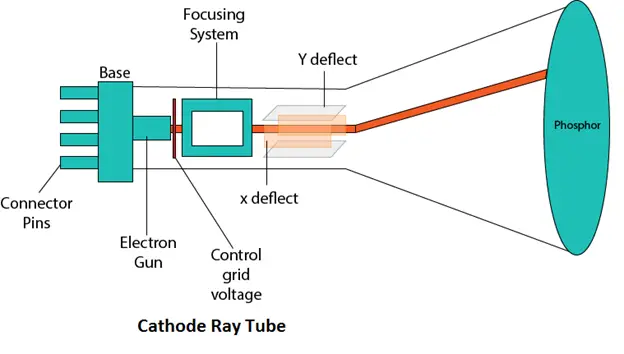
An image of the Cathode Ray Tube used in Thomson's experiments.
Image courtesy of Laws of Nature
His experiments proceeded in three stages:
- He observed that rays emitted from the cathode were deflected toward the positive pole of a magnet, showing the rays carried negative charge.
- He confirmed this by deflecting the rays with an external electric field.
- Using the deflection data, he calculated the mass-to-charge ratio of the particles. The particles turned out to be about 1,000 times lighter than a hydrogen atom, so Thomson concluded they were subatomic particles (later named electrons).
To account for electrons inside an electrically neutral atom, Thomson proposed the plum pudding model: a sphere of uniformly distributed positive charge with tiny negative electrons embedded throughout, like raisins in a pudding.

A visual of the plum pudding model created by J.J. Thomson.
Image courtesy of Wikimedia Commons
🥇 Ernest Rutherford (1911)
Rutherford tested Thomson's model by firing alpha particles (positively charged) at a thin sheet of gold foil and detecting where they landed on a surrounding screen.
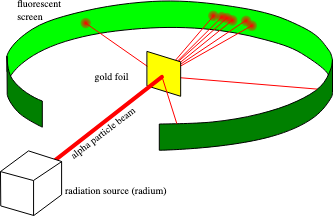
Rutherford's gold foil experiment
Image courtesy of Wikimedia Commons
If the plum pudding model were correct, the positive charge would be spread so thinly that all alpha particles should pass straight through. Instead, Rutherford observed that:
- Most particles passed through undeflected (the atom is mostly empty space).
- Some were deflected at large angles.
- A few bounced almost straight back (they hit something small, dense, and positively charged).
These results led to three conclusions:
- Atoms have a small, dense, positively charged nucleus at the center.
- Most of the atom's volume is empty space.
- Electrons occupy the space around the nucleus but take up very little of the atom's total volume.
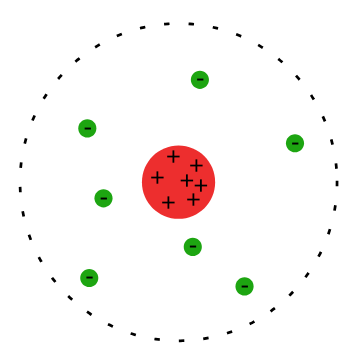
The Rutherford atomic model
Image courtesy of Wikimedia Commons
♾️ Niels Bohr (1913)
Bohr refined Rutherford's model by introducing quantized energy levels. He proposed that electrons orbit the nucleus only at specific, fixed distances, each corresponding to a particular energy. Electrons can jump between these levels by absorbing or emitting specific amounts of energy, which explains why elements produce characteristic line spectra (distinct colors of light).
Bohr's model worked well for hydrogen but couldn't fully explain atoms with more electrons. Still, it was a critical stepping stone toward quantum mechanics.
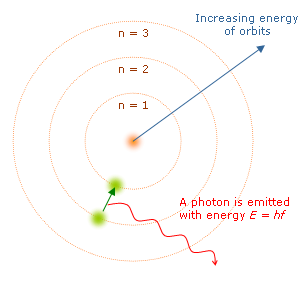
Representation of the different energy levels discovered by Bohr
Image courtesy of Enoch Lau
🌐 From Bohr to Quantum Mechanics
After Bohr, the quantum mechanical model took shape through several key contributions:
-
Werner Heisenberg formulated the Uncertainty Principle: you cannot simultaneously know both the exact position and exact momentum of an electron. The more precisely you measure one, the less precisely you can know the other.
-
Erwin Schrödinger developed mathematical wave equations that describe electrons not as particles in fixed orbits, but as probability distributions. This is the electron cloud model you'll use going forward. Regions where the cloud is denser represent higher probability of finding an electron.
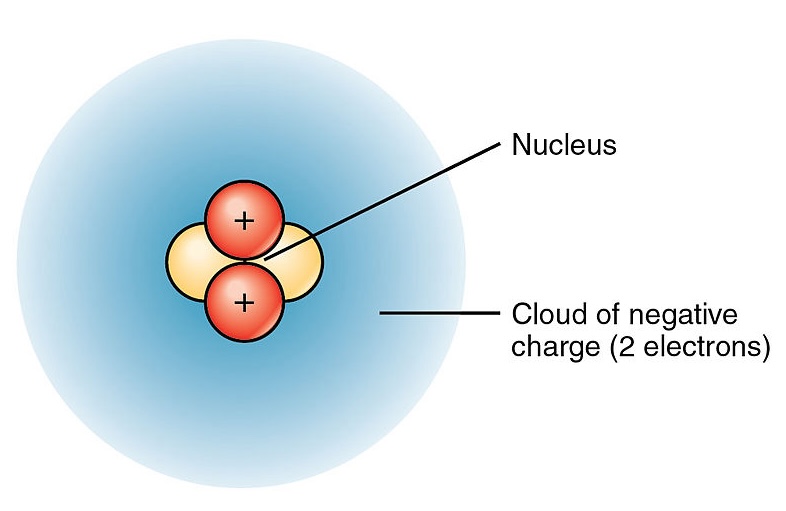
Image depicting the electron cloud model of Schrödinger and his contemporaries.
Image courtesy of Wikimedia Commons
-
James Chadwick (1932) discovered the neutron, a neutral particle in the nucleus alongside protons. This completed the picture of the three subatomic particles: protons (positive), neutrons (neutral), and electrons (negative).
✏️ Practice Questions
- The word for atom comes from the Greek word atomos, meaning…
- Invisible
- Undivided
- Indivisible
- Indestructible
- What is the positively charged subatomic particle?
- The neutron
- The positron
- The proton
- The electron
- What is the negatively charged subatomic particle?
- The neutron
- The positron
- The proton
- The electron
- What is the neutrally charged subatomic particle?
- The neutron
- The positron
- The proton
- The electron
- Who used gold foil to discover the nucleus?
- Ernest Rutherford
- Niels Bohr
- Democritus
- Schrödinger
- Compare and contrast the plum pudding model, the Bohr model, and the electron cloud model.
📌 Solutions
- C, indivisible
- C, the proton
- D, the electron
- A, the neutron
- A, Ernest Rutherford
- The plum pudding model (Thomson) pictures electrons scattered throughout a uniform sphere of positive charge, with no nucleus. The Bohr model incorporates Rutherford's discovery of a dense, positively charged nucleus and places electrons in fixed circular orbits at specific energy levels around it. The electron cloud model (Schrödinger) keeps the nucleus but replaces fixed orbits with probability clouds, recognizing that you can only predict the likelihood of finding an electron in a given region. All three models agree that atoms contain negatively charged electrons, but they differ in how those electrons are arranged and whether a distinct nucleus exists.
⚛️ Wrapping Up Atomic Theory
The modern understanding of the atom goes even deeper than the electron cloud model. The Standard Model of particle physics describes protons and neutrons as being made of smaller particles called quarks, held together by fundamental forces. For this course, though, the key takeaway is the progression: Dalton's solid sphere → Thomson's plum pudding → Rutherford's nuclear model → Bohr's quantized orbits → the quantum mechanical electron cloud.
This guide covered the history of how atomic theory came to be, and having this knowledge will be fundamental in moving to the next topic on atom structures and subatomic particles.