Overview of Biomolecules
This guide covers the major organic reaction types and mechanisms you'll encounter in honors chemistry: substitution, addition, elimination, and rearrangement. Understanding how and why these reactions proceed the way they do is essential for predicting products and designing synthetic pathways.

Basic Organic Reactions and Mechanisms
Nucleophilic Substitution Reactions (SN1 and SN2)
In nucleophilic substitution, a nucleophile (electron-rich species) replaces a leaving group on a carbon atom. The "S" stands for substitution, the "N" for nucleophilic, and the number (1 or 2) tells you how many species are involved in the rate-determining step.
SN1: Unimolecular Nucleophilic Substitution
The SN1 mechanism happens in two steps: first the leaving group departs on its own to form a carbocation intermediate, then the nucleophile attacks.
- The rate depends only on the substrate concentration because the slow step involves just the substrate losing its leaving group.
- Substrate preference: Tertiary > Secondary > Primary. Tertiary substrates react fastest because their carbocation intermediates are stabilized by three alkyl groups donating electron density through hyperconjugation and inductive effects.
- The carbocation intermediate is planar (flat), so the nucleophile can attack from either side. This leads to racemization, where you get a roughly equal mix of both stereochemical configurations.
- A racemic mixture contains equal amounts of both enantiomers (mirror-image forms), so the mixture shows no net optical activity.
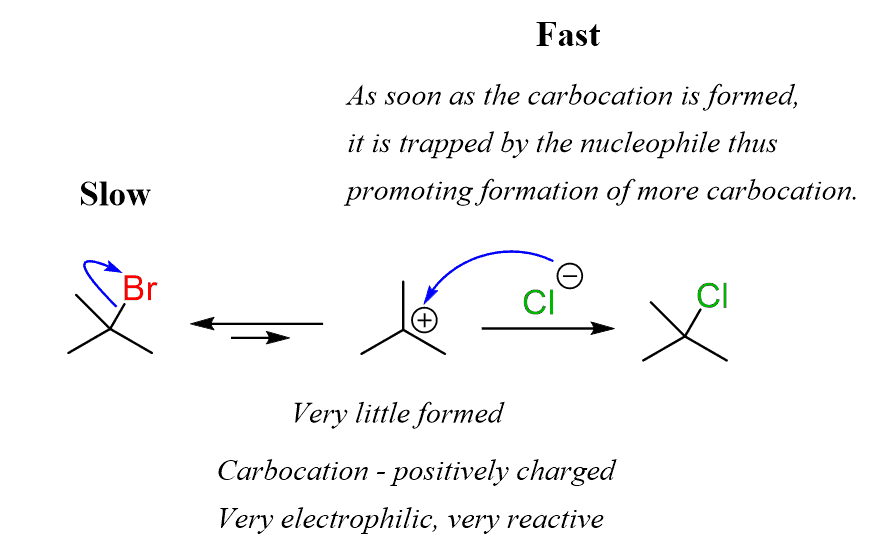
Image shows the SN1 Reaction Mechanism
Image courtesy of chemistry steps
SN2: Bimolecular Nucleophilic Substitution
The SN2 mechanism is a concerted, one-step process: the nucleophile attacks at the same time the leaving group departs. There's no intermediate.
- The rate depends on the concentrations of both the substrate and the nucleophile.
- Substrate preference: Methyl > Primary > Secondary. Tertiary substrates almost never undergo SN2 because bulky groups block the nucleophile's approach (steric hindrance).
- The nucleophile always attacks from the backside (opposite the leaving group), causing inversion of configuration. Think of an umbrella flipping inside out in the wind.
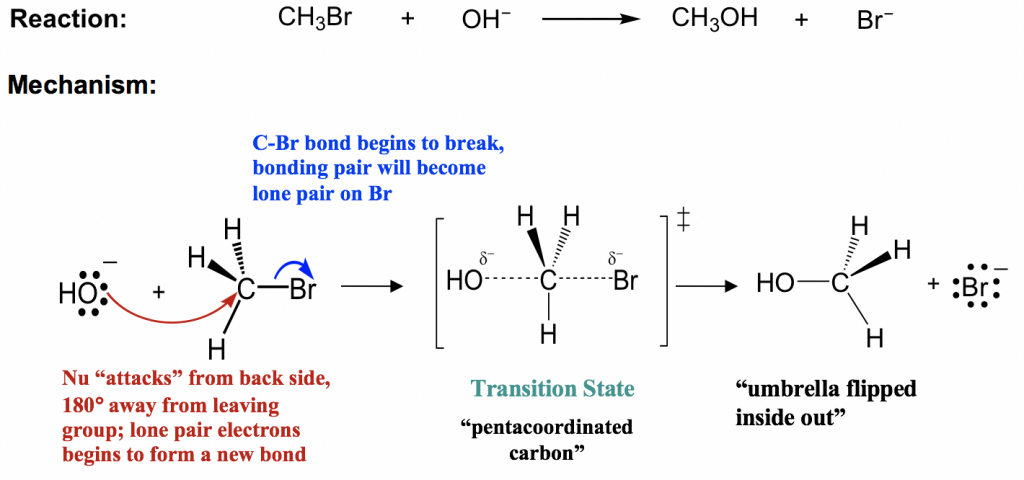
Image shows SN2 Reaction with descriptions of each state.
Image Courtesy of Kwantlen Polytechnic University
SN1 vs SN2: How to Decide
Four factors determine which mechanism dominates:
-
Substrate Structure
- SN1: Favored with tertiary substrates that form stable carbocations.
- SN2: Favored with methyl and primary substrates where the carbon is accessible.
-
Nucleophile and Leaving Group
- SN1: Nucleophile strength matters less because the nucleophile isn't involved in the rate-determining step.
- SN2: Requires a strong nucleophile capable of backside attack. Bulky nucleophiles slow SN2 down.
-
Solvent
- SN1: Polar protic solvents (water, alcohols) stabilize the carbocation intermediate through solvation.
- SN2: Polar aprotic solvents (acetone, DMF) are preferred because they don't solvate the nucleophile, keeping it reactive.
-
Steric Hindrance
- SN1: Less affected, since the leaving group departs before the nucleophile arrives.
- SN2: Highly sensitive. Bulky groups around the reactive carbon block nucleophilic attack.
Practice Question
Predict whether the following reaction would proceed via an SN1 or SN2 mechanism:
Answer: SN2, for three reasons:
- Substrate: is a primary alkyl bromide, which favors SN2.
- Nucleophile: is a strong nucleophile, well-suited for direct backside attack.
- Steric hindrance: Minimal crowding around the carbon bearing the leaving group.
Electrophilic Addition Reactions
Alkenes and alkynes have electron-rich bonds that attract electrophiles (electron-poor species). In electrophilic addition, atoms add across the double or triple bond, breaking the bond and forming new bonds.
Key Concepts
- Markovnikov's rule: When HX adds to an unsymmetrical alkene, the hydrogen attaches to the carbon that already has more hydrogens. The electrophile () adds to form the more stable (more substituted) carbocation intermediate.
- Anti-Markovnikov addition: Under certain conditions (such as the presence of peroxides with HBr), the addition reverses. A radical mechanism operates instead, placing the hydrogen on the carbon with fewer hydrogens.
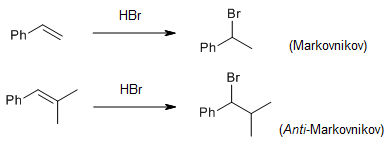
Image Description: Markovnikov and anti-Markovnikov rule.
Image courtesy: Organic Chemistry Portal.
Practice Question
What products would you expect from addition of HBr to 3-methyl-1-butene under (a) standard conditions and (b) peroxide conditions?
Answer:
- (a) Standard conditions (Markovnikov): adds to C-1 (the carbon with more hydrogens), and adds to C-2. The major product is 2-bromo-3-methylbutane.
- (b) Peroxide conditions (anti-Markovnikov): A radical mechanism places Br on C-1 instead. The major product is 1-bromo-3-methylbutane.
Elimination Reactions (E1 and E2)
Elimination reactions remove a leaving group and a proton from adjacent carbons to form a bond (alkene). Like substitution, there are two mechanisms depending on whether the steps happen together or separately.
E1: Unimolecular Elimination
E1 proceeds in two steps:
- The leaving group departs, forming a carbocation intermediate (slow, rate-determining step).
- A base removes a proton from a carbon adjacent to the carbocation, forming the alkene (fast step).
E2: Bimolecular Elimination
E2 is concerted (one step): the base abstracts a proton at the same time the leaving group departs, and the double bond forms simultaneously. Both the substrate and the base are involved in the rate-determining step.
Zaitsev's Rule: In both E1 and E2, the more substituted alkene is generally the major product because it's more thermodynamically stable. "More substituted" means more alkyl groups are attached to the carbons of the double bond.
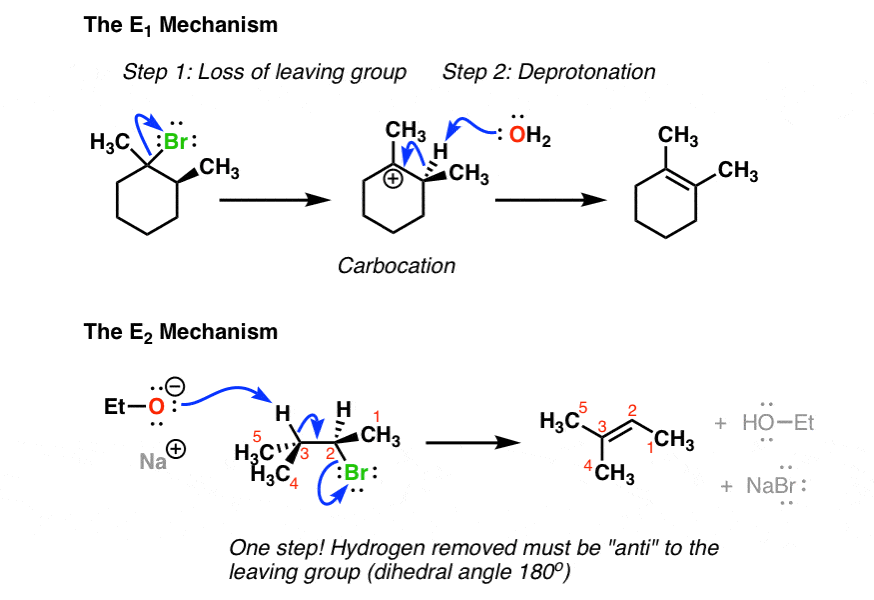
Image showing side-by-side comparison of E1 and E2 reactions.
Image courtesy of Master Organic Chemistry
E1 vs E2: How to Decide
-
Substrate Structure
- E1: Favored with tertiary substrates that form stable carbocations.
- E2: Works well with primary and secondary substrates. Also occurs with tertiary substrates when a strong base is present.
-
Base Strength
- E1: Weak bases or neutral conditions. The base only needs to remove a proton after the carbocation forms.
- E2: Strong bases are required to pull off the proton in the concerted step.
-
Steric Hindrance
- E1: Less sensitive to steric effects since the carbocation forms first.
- E2: Bulky groups can slow the reaction, but bulky bases actually favor elimination over substitution.
-
Solvent
- E1: Polar protic solvents stabilize the carbocation intermediate.
- E2: Polar aprotic solvents keep the base unsolvated and reactive.
Practice Question
Predict whether E1 or E2 will occur: 1-bromobutane () reacts with in liquid ammonia.
Answer: E2, for three reasons:
- Base strength: is a very strong base, which strongly favors E2.
- Steric hindrance: The primary substrate has minimal crowding, allowing the base easy access.
- Solvent: Liquid ammonia is a polar aprotic solvent, which doesn't solvate the base and favors E2.
Rearrangement Reactions
In rearrangement reactions, atoms or groups shift positions within a molecule to form a more stable structure. The most common type you'll see involves carbocation rearrangements, where a hydride () or methyl group migrates to an adjacent carbon to convert a less stable carbocation into a more stable one.
For example, a secondary carbocation can rearrange to a tertiary carbocation through a 1,2-hydride shift. This matters in SN1 and E1 reactions because both proceed through carbocation intermediates.
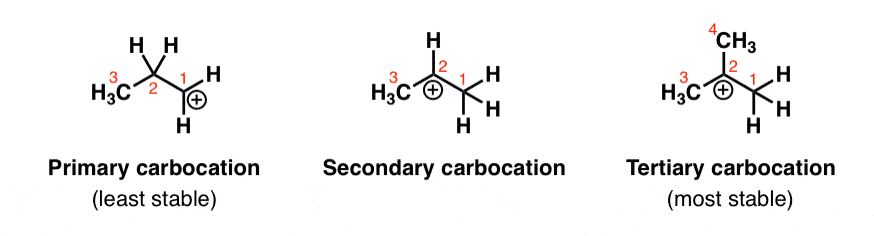
Diagram showing the most stable out of three molecules.
Image courtesy of Master Organic Chemistry
Reaction Intermediates & Transition States
Reaction Intermediates
An intermediate is a species that forms during a reaction and exists briefly before converting to the next species or the final product. It sits in an energy valley on a reaction energy diagram.
The four common intermediates:
- Carbocations (positively charged carbon): Stability order is tertiary > secondary > primary > methyl. More alkyl groups stabilize the positive charge.
- Carbanions (negatively charged carbon): Follow a roughly opposite stability trend from carbocations. Electron-withdrawing groups nearby help stabilize them.
- Free radicals (carbon with one unpaired electron): Stability follows the same trend as carbocations (tertiary > secondary > primary), but radicals are generally less reactive than carbocations because the carbon isn't formally charged.
- Carbenes (neutral carbon with two bonds and a lone pair): Highly reactive, short-lived species.
Transition States
A transition state is the highest-energy point along a reaction pathway. Unlike intermediates, transition states can't be isolated. They represent the exact moment bonds are partially breaking and partially forming. On an energy diagram, transition states appear as peaks.
The energy difference between reactants and the transition state is the activation energy (), which determines how fast the reaction proceeds.
Predicting Outcomes
Stereochemistry
Stereochemistry deals with the 3D arrangement of atoms in molecules. It directly affects reaction outcomes and product properties.
A molecule is chiral if it can't be superimposed on its mirror image (like your left and right hands). A carbon bonded to four different groups is a chiral center (also called a stereocenter).
Key stereochemical outcomes to track in reactions:
- Inversion of configuration: The stereochemistry flips at the reactive carbon (occurs in SN2).
- Retention of configuration: The stereochemistry stays the same.
- Racemization: You get a 50/50 mix of both configurations (occurs in SN1 due to the planar carbocation).
- Formation of new chiral centers: Addition reactions can create new stereocenters, and you need to predict which configuration(s) will form.
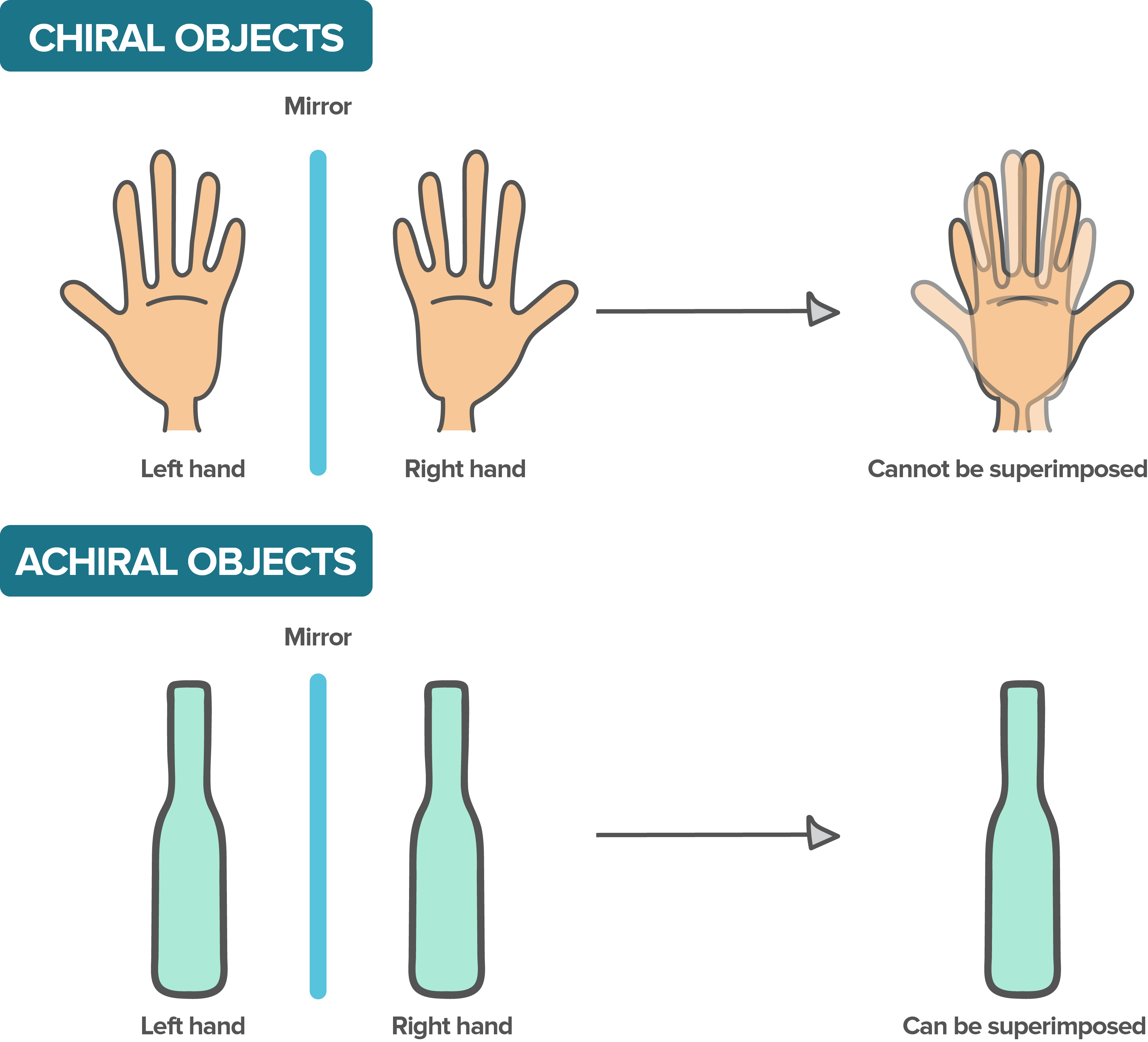
Image Description: Depicting chiral molecules vs achiral molecules
Image courtesy: Khan Academy.
Practice Question
Predict the major product: 1-bromo-2-methylcyclohexane undergoes elimination with alcoholic KOH.
Answer: The major product is 1-methylcyclohexene.
Step-by-step reasoning:
- Identify the reaction type. Alcoholic KOH is a strong base, so this favors E2 elimination over E1.
- Determine the mechanism. The substrate is secondary, and the strong base drives a concerted E2 process.
- Apply Zaitsev's rule. The base () abstracts a proton from the adjacent carbon, and the leaving group () departs simultaneously to form an alkene. The more substituted alkene is favored.
- Identify the major product. 1-Methylcyclohexene is more substituted than the alternative (methylenecyclohexane), making it the Zaitsev product.
- Check for rearrangements. E2 is concerted with no carbocation intermediate, so no rearrangement occurs.
Synthetic Strategies & Conditions
When planning a synthesis, you need to consider how reaction conditions affect which pathway a reaction takes:
- Catalysts lower the activation energy and can direct a reaction toward a specific product without being consumed.
- Temperature affects reaction rate and can shift selectivity. Higher temperatures tend to favor elimination over substitution.
- Solvent choice influences mechanism (polar protic vs. polar aprotic, as discussed above).
- Retrosynthetic analysis is the strategy of working backward from your target molecule to identify simpler starting materials. You break bonds in the target to reveal what reactions and reagents could have built it.
The key to predicting outcomes is combining your knowledge of mechanism, stereochemistry, and reaction conditions. Work through practice problems systematically: identify the substrate type, the reagent/base/nucleophile strength, the solvent, and then determine which mechanism applies.