Structure of Atoms and Subatomic Particles
Atoms are the building blocks of all matter, and understanding their internal structure is essential for everything else in chemistry. The arrangement of subatomic particles determines what element you're dealing with, how it behaves in reactions, and why the periodic table is organized the way it is.

Subatomic Particles and Their Roles
Atoms are made up of three main types of particles: protons, neutrons, and electrons. Each one plays a distinct role.
Protons (p⁺)
- Positively charged
- Found in the nucleus
- Determine the atomic number (Z), which defines what element an atom is
- For example, an atom with 1 proton is always hydrogen; an atom with 6 protons is always carbon
Neutrons (n⁰)
- Have no charge (neutral)
- Also located in the nucleus alongside protons
- Contribute to the mass number (A), which is the sum of protons and neutrons
- Differences in neutron count create isotopes of the same element
- For example, has 8 protons and 8 neutrons, while has 8 protons and 10 neutrons
- Help stabilize the nucleus. Too many or too few neutrons relative to protons can make a nucleus radioactive.
Electrons (e⁻)
- Negatively charged
- Move around the nucleus in regions called electron shells or electron clouds
- Much less massive than protons or neutrons (about 1/1836 the mass of a proton), so they contribute negligibly to an atom's mass
- Drive chemical behavior because they participate in bonding between atoms
Atomic Structure Arrangement
The Nucleus
The nucleus sits at the center of the atom and contains all the protons and neutrons packed tightly together. Despite holding nearly all of the atom's mass, the nucleus is incredibly small compared to the overall size of the atom.
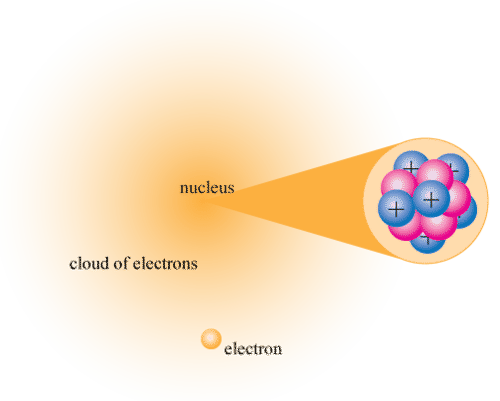
A dense nucleus containing closely packed protons and neutrons.
Image courtesy of CK-12 Foundation
Electron Shells (Bohr Model)
In the Bohr model (also called the planetary model), electrons orbit the nucleus in fixed energy levels called shells. Each shell can hold a maximum number of electrons given by the formula , where is the shell number.
- Shell 1 (): holds up to 2 electrons
- Shell 2 (): holds up to 8 electrons
- Shell 3 (): holds up to 18 electrons
This model is a useful simplification, but it doesn't fully capture how electrons actually behave.
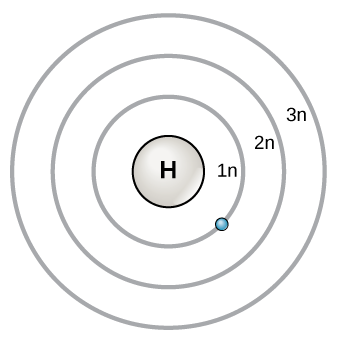
A diagram with concentric circles around a central nucleus depicting electron shells.
Image courtesy of Lumen Learning
Quantum Mechanical Model
The modern quantum mechanical model replaces fixed orbits with orbitals, which are regions of space where an electron has a high probability of being found. Rather than traveling in neat circular paths, electrons exist in a cloud-like distribution around the nucleus. This model is more accurate and is the basis for understanding electron configuration in detail.
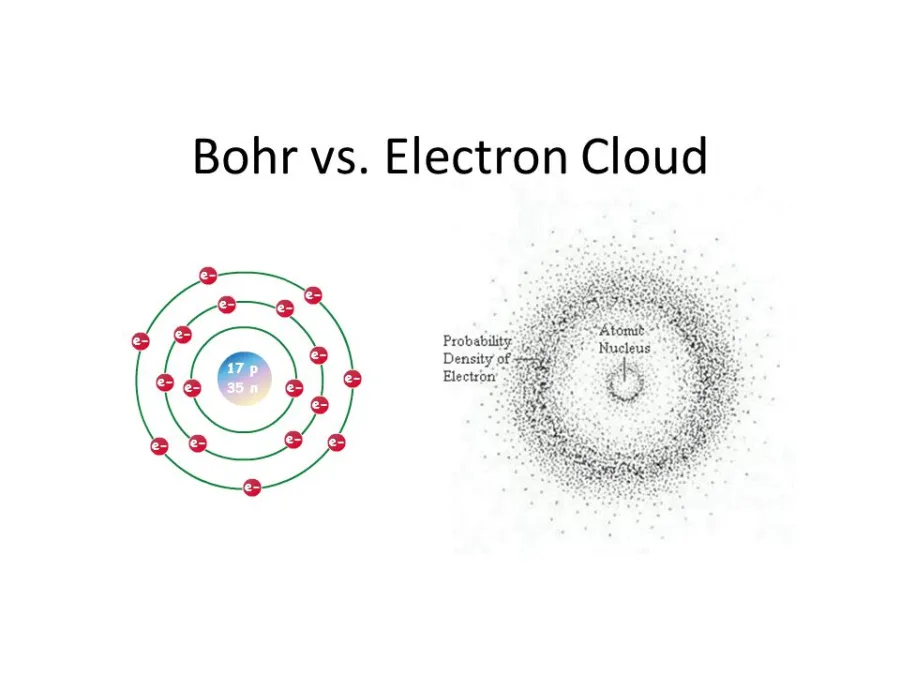
Image depicting the difference between the Bohr, or planetary, model of the electron shell vs the modern quantum mechanical model.
Image courtesy of ZME Science
Atomic Number and Mass Number
Every element is identified by two key numbers:
Atomic Number (Z)
- Equal to the number of protons in the nucleus
- Defines the element's identity. Change the number of protons, and you have a different element entirely.
Mass Number (A)
- Equal to the number of protons plus the number of neutrons:
- Different isotopes of the same element have different mass numbers
Atomic Mass Concepts
Atomic Mass
The atomic mass of a specific atom equals the combined count of its protons and neutrons (since electron mass is negligible). This value varies from isotope to isotope.
Average Atomic Mass
In nature, elements exist as mixtures of their isotopes. The average atomic mass is a weighted average that accounts for each isotope's mass and its natural abundance. This is the number listed on the periodic table for each element.
For example, chlorine has two main isotopes: (about 75.8% abundance) and (about 24.2% abundance). The weighted average gives chlorine an average atomic mass of approximately 35.45 amu.
Isotopes: Variations of Elements
Isotopes are atoms of the same element that differ in their number of neutrons.
- Same number of protons (same atomic number)
- Different number of neutrons (different mass number)
Isotopes can be stable or radioactive, depending on their neutron-to-proton ratio. Radioactive isotopes undergo decay over time, releasing particles or energy. This decay process is the basis for techniques like carbon-14 dating, which is used to determine the age of ancient artifacts and fossils.
Electron Configuration & Chemical Behavior
How electrons are arranged around the nucleus directly determines an element's chemical properties.
Valence Electrons
Valence electrons are the electrons in the outermost shell of an atom. They're the ones involved in chemical bonding.
Atoms tend toward having a full outer shell because that's the most stable configuration (this is sometimes called the octet rule for elements that want 8 valence electrons). To achieve this, atoms will transfer or share electrons with other atoms.
For example, sodium (Na) has 1 valence electron and chlorine (Cl) has 7. Sodium transfers its single valence electron to chlorine, giving both atoms full outer shells. This produces the ionic compound NaCl (table salt).
Periodic Trends
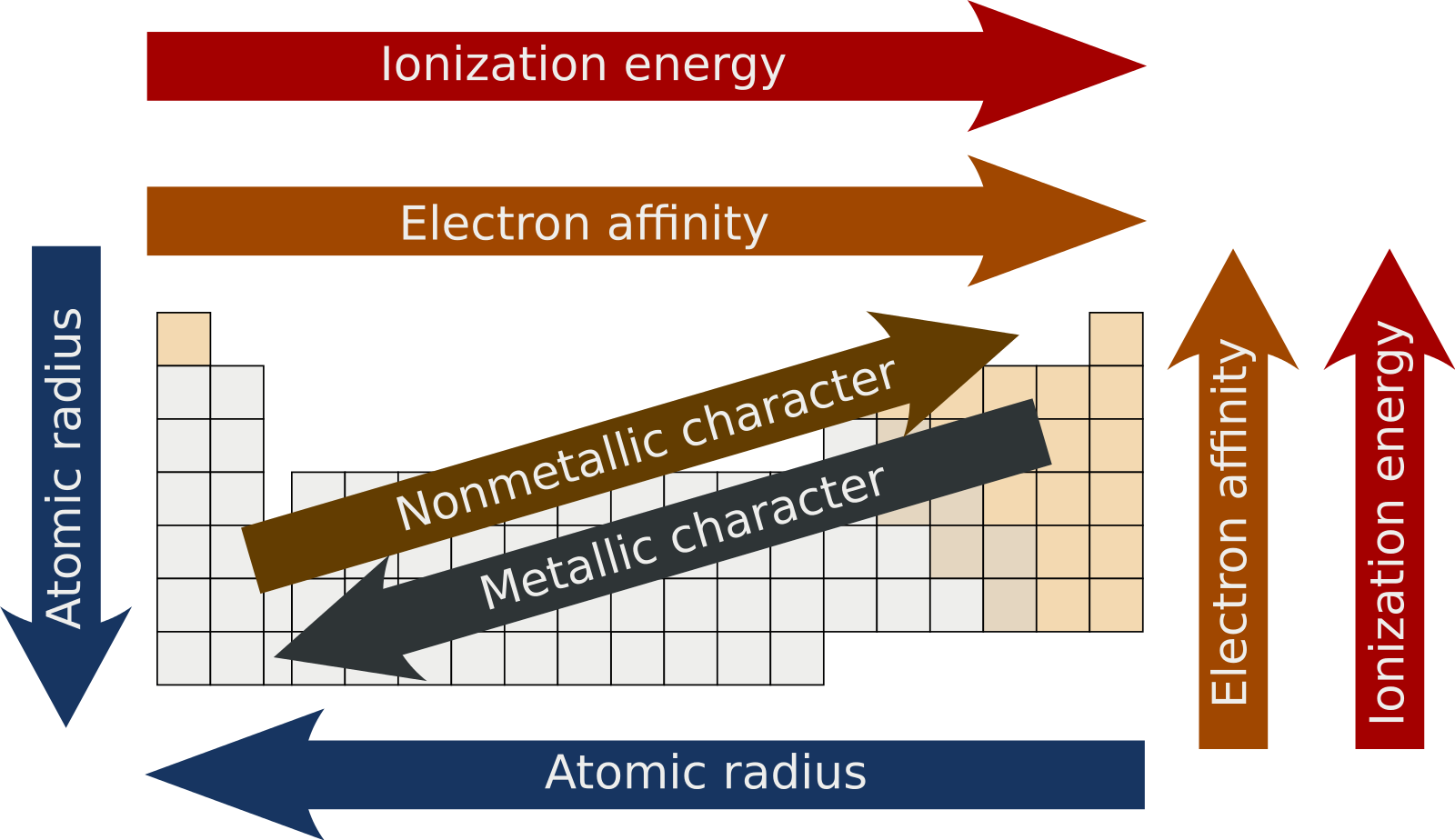
A periodic table highlighting trends such as electron affinity, atomic radius, ionization energy, and metallic character.
Image courtesy of Wikimedia Commons
Electron configuration creates predictable patterns across the periodic table:
- Atomic radius: The average distance from the nucleus to the outermost electron. Increases going left and down the table, because you're adding shells (going down) and because there's less effective nuclear pull on outer electrons (going left).
- Ionization energy: The energy required to remove one electron from a neutral atom. Increases going right and up. Smaller atoms with more protons hold their electrons more tightly.
- Electron affinity: The energy released when an atom gains an electron. Generally increases going right and up. Nonmetals tend to have high electron affinities because gaining an electron brings them closer to a full outer shell.
- Metallic character: The tendency of an element to lose electrons and form positive ions (cations). Increases going left and down.
- Nonmetallic character: The tendency to gain electrons and form negative ions (anions). Increases going right and up. This trend runs diagonally opposite to metallic character.
Quantum Numbers & Orbital Shapes
The quantum mechanical model uses four quantum numbers to describe the location and behavior of each electron in an atom.
-
Principal quantum number (): Indicates the energy level or shell. Higher means higher energy and greater distance from the nucleus.
-
Angular momentum quantum number (): Describes the shape of the orbital. Values range from 0 to . Each value corresponds to an orbital type: is s, is p, is d, is f.
-
Magnetic quantum number (): Specifies the orientation of the orbital in space. Values range from to .
-
Spin quantum number (): Indicates the direction of electron spin, either or . Each orbital can hold at most 2 electrons with opposite spins.
Orbitals have distinct shapes: s-orbitals are spherical, p-orbitals are dumbbell-shaped, and d- and f-orbitals have more complex geometries. These shapes describe where electrons are most likely to be found.
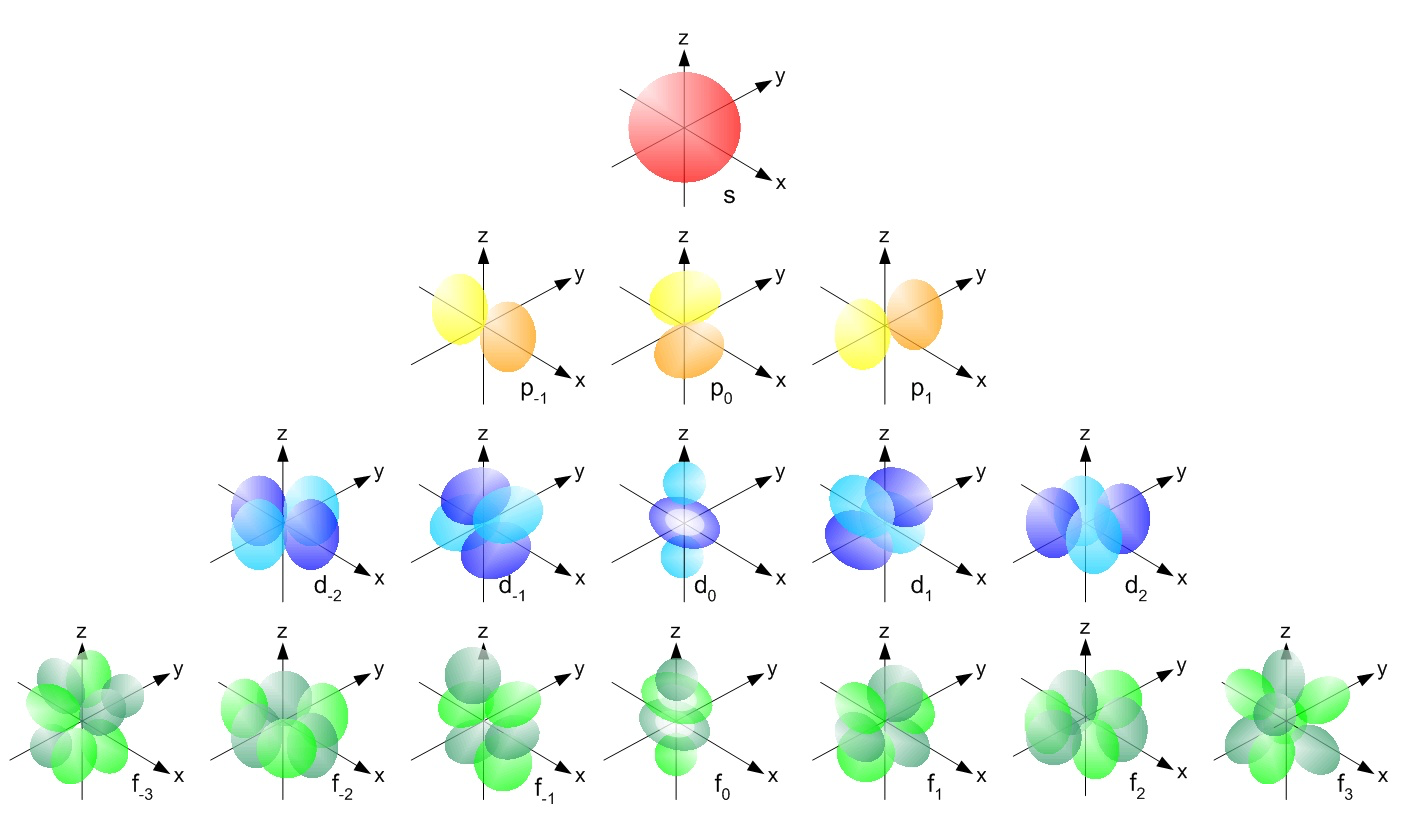
Showing different shapes for s, p, d, f orbitals.
Image courtesy of LibreTexts Chemistry
Practice Questions
Questions
- What is the atomic number for an element with 13 protons?
- Calculate the mass number for an element with 15 neutrons, 12 protons, and 12 electrons.
- Describe why isotopes have different atomic masses but not different atomic numbers.
- Sketch out what you think a p-orbital would look like.
Solutions
- The atomic number (Z) equals the number of protons, so it's 13 (this is aluminum).
- Mass number is the sum of protons and neutrons: . Electrons don't count toward mass number.
- Isotopes of a given element all have the same number of protons, which is what defines them as that element. However, they have different numbers of neutrons, which changes the mass number without affecting the atomic number.
- A p-orbital has a dumbbell or figure-eight shape in three dimensions. There are three p-orbitals per energy level (for ), each oriented along a different axis (x, y, z).
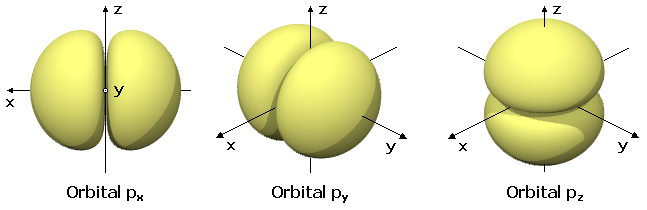
Sketch of a p-orbital in three different orientations.
Image courtesy of Wikimedia Commons