Nuclear Fission and Fusion Processes
Nuclear fission and fusion are the two main types of nuclear reactions that convert mass into energy. Understanding these processes is central to nuclear chemistry, and they show up in everything from power plants to stellar physics. This guide covers how each process works, their applications in energy generation, and the challenges each one faces.

Understanding Nuclear Fission
Nuclear fission is a process where a heavy, unstable nucleus splits into two or more smaller nuclei, releasing a large amount of energy in the process. The energy comes from the conversion of a small amount of mass into energy, described by Einstein's equation .
Key Concepts
Heavy nuclei like (Uranium-235) and (Plutonium-239) are the most common fission fuels. These isotopes are described as fissile, meaning they can sustain a fission chain reaction when struck by a neutron.
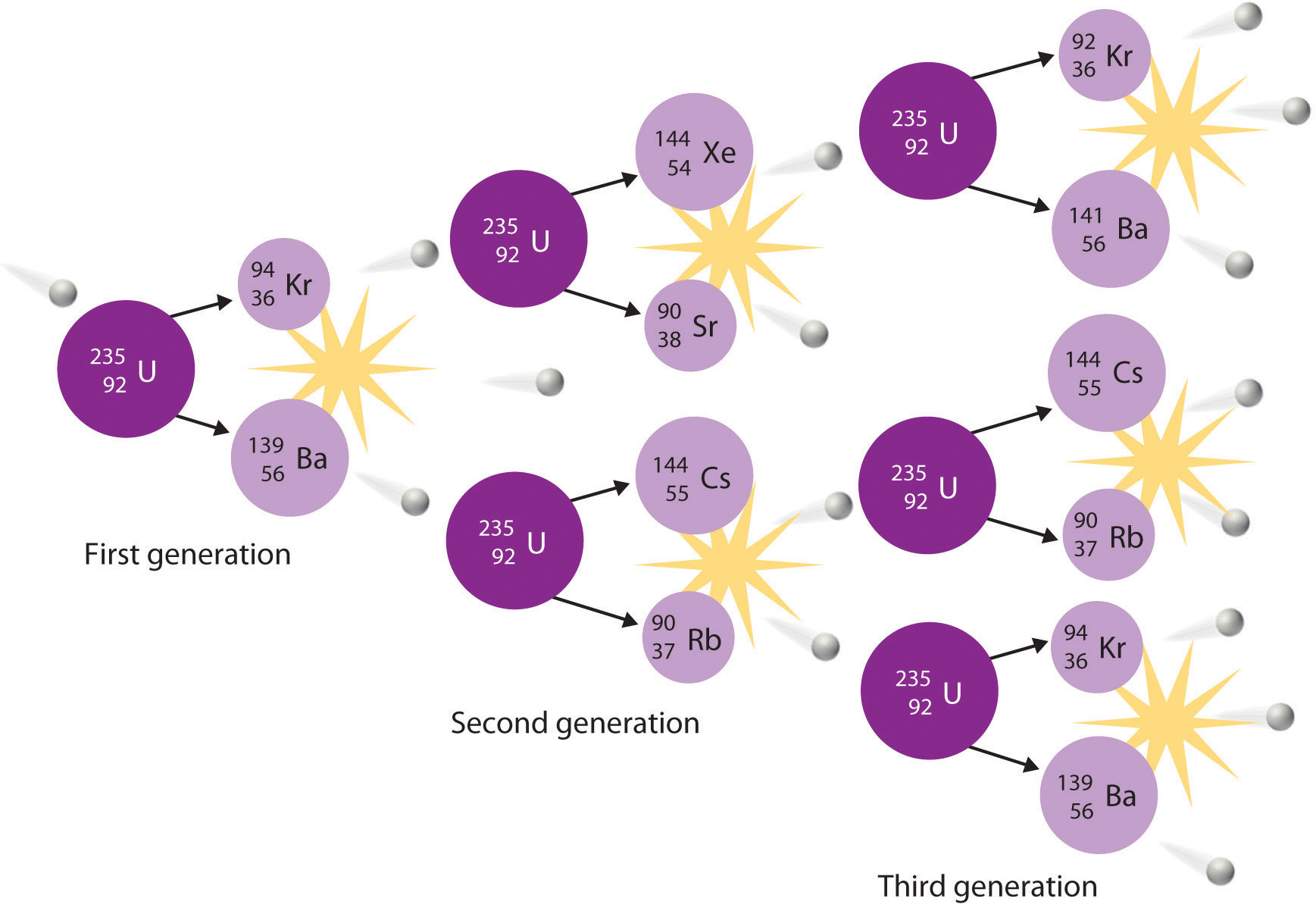
Here's how fission works step by step:
- A slow-moving (thermal) neutron strikes a heavy nucleus like .
- The nucleus absorbs the neutron and becomes highly unstable.
- The unstable nucleus splits into two mid-sized nuclei (called fission fragments), plus 2-3 additional neutrons.
- Those released neutrons can strike other nuclei, causing more fission events.
- This self-propagating process is a chain reaction.
The fact that each fission event produces multiple neutrons is what makes chain reactions possible. If at least one neutron from each event triggers another fission, the reaction sustains itself. This is called reaching criticality.
Example Reaction
Notice that both mass number and atomic number are conserved on each side of the equation. Always check this when balancing nuclear equations.
Nuclear fission reaction causing the split of a Uranium atom
Image courtesy of Chemistry LibreTexts
Understanding Nuclear Fusion
Nuclear fusion is the opposite of fission: two light nuclei combine to form a heavier nucleus, releasing energy. Fusion is the process that powers stars, including our Sun.
Key Concepts
Light nuclei are the fuel for fusion. The most studied reaction uses deuterium () and tritium (), which are isotopes of hydrogen.
Fusion requires extreme conditions because atomic nuclei are all positively charged, and like charges repel. This electrostatic repulsion (called the Coulomb barrier) must be overcome before nuclei can get close enough for the strong nuclear force to bind them together. That's why fusion requires temperatures exceeding K and very high pressures.
Example Reaction
Fusion releases even more energy per unit mass of fuel than fission does. A single gram of deuterium-tritium fuel releases roughly four times the energy of a gram of .
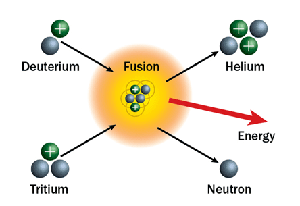
Merging of two hydrogen isotopes results in a helium nucleus and neutron.
Image courtesy of Science News
Applications in Energy Generation
Both fission and fusion can generate electricity without producing carbon dioxide during operation, making them relevant to discussions about clean energy.
Fission in Nuclear Reactors
Fission is already widely used for power generation. The basic idea: controlled fission heats water, the steam drives a turbine, and the turbine generates electricity.
- Reactor types include light-water reactors (LWRs), which come in two main designs: pressurized water reactors (PWRs) and boiling water reactors (BWRs). PWRs keep the primary coolant under high pressure to prevent boiling, while BWRs allow the water to boil directly in the reactor core.
- Control rods made of neutron-absorbing materials (like cadmium or boron) are inserted into or withdrawn from the reactor core to speed up or slow down the chain reaction.
- Coolant (usually water) circulates through the core to carry away heat and transfer it to the steam generation system.
- Multiple layers of containment structures and emergency shutdown systems prevent the release of radioactive material.
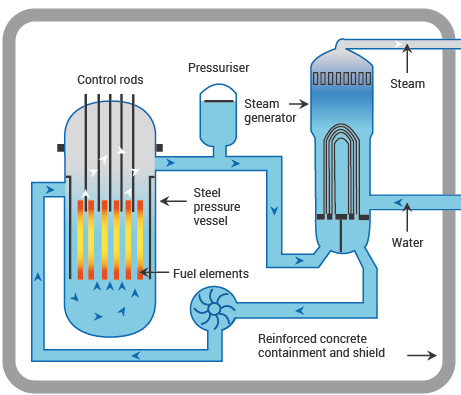
Diagram showing inside a nuclear reactor core
Image courtesy of World Nuclear Association
Fusion Prospects
Controlled fusion for power generation has not yet been achieved commercially, but research is active.
- Tokamaks and stellarators are two reactor designs that use powerful magnetic fields to confine superheated plasma (the state of matter at fusion temperatures). The tokamak is the more widely studied design.
- Ignition refers to the point where a fusion reaction becomes self-sustaining, meaning the energy produced by fusion keeps the plasma hot enough to continue reacting without external heating. This remains an extremely difficult engineering challenge.
- Fusion fuel is abundant: deuterium can be extracted from seawater, and tritium can be bred from lithium. Fusion also produces far less long-lived radioactive waste than fission.
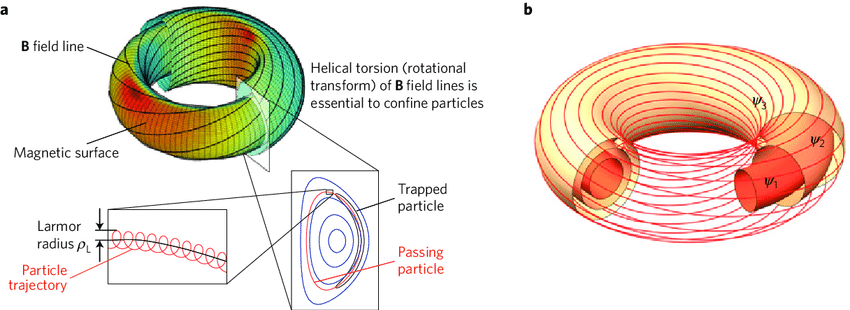
Magnetic confinement in a Tokamak.
Image courtesy of Research Gate
Challenges and Future Prospects
For Fission
- Radioactive waste management: Spent fuel contains isotopes that remain dangerously radioactive for thousands of years. Long-term storage solutions (like deep geological repositories) are still being developed.
- Nuclear proliferation: Fissile materials can potentially be diverted for weapons production, so strict international oversight is required.
- Economics: Nuclear plants have very high upfront construction costs, even though operating costs are relatively low.
For Fusion
- Extreme conditions: Plasma must be heated to over 150 million °C (about 10 times hotter than the Sun's core) because Earth-based reactors operate at much lower pressures than the Sun's interior, so they need higher temperatures to compensate.
- Net energy gain: The reactor must produce more energy from fusion than is consumed to heat and confine the plasma. Achieving this consistently is the central engineering hurdle.
- Timeline: Commercial fusion power is still likely decades away, though projects like ITER (an international tokamak being built in France) aim to demonstrate net energy gain.
Future Directions
- Small modular reactors (SMRs) are a newer fission technology that could be factory-built and deployed more flexibly than traditional large-scale plants.
- International collaboration through ITER and similar projects continues to push fusion research forward.
- Both public perception and government policy will shape how nuclear technology develops alongside other energy sources.
Practice Questions
1) If one fission event releases around 200 MeV of energy, how much energy would be released from 1 mole of Uranium-235 undergoing fission?
Explanation:
One mole of contains Avogadro's number of atoms: atoms.
Step 1: Convert the energy per fission event from MeV to joules.
Step 2: Multiply by the number of atoms in one mole.
That's roughly joules per mole, an enormous amount of energy from just 235 grams of uranium. For comparison, burning 235 grams of coal releases only about 7,000 J.
2) Why do scientists need temperatures even higher than the Sun's core to achieve fusion on Earth?
Explanation:
The Sun's core reaches about 15 million °C, but it also has immense gravitational pressure (about 250 billion atmospheres) that compresses nuclei together and increases the probability of fusion. On Earth, we can't replicate that pressure, so we compensate by using even higher temperatures (over 150 million °C) to give the nuclei enough kinetic energy to overcome the Coulomb barrier (electrostatic repulsion between positive charges) on their own. Without the Sun's gravity doing part of the work, temperature has to do all of it.