Accuracy, Precision, Percent Error
Every measurement in chemistry carries some degree of error. Understanding accuracy, precision, and percent error gives you the tools to evaluate how trustworthy your experimental data really is and to diagnose what went wrong when results are off.

Accuracy Versus Precision
Accuracy
Accuracy describes how close a measured value is to the true or accepted value. If the accepted density of aluminum is and you measure , that's highly accurate.
Systematic Errors
Systematic errors push all your measurements in the same direction, either consistently too high or consistently too low. They usually come from flawed equipment or a problem in your experimental setup. A classic example: if a balance isn't zeroed before use, every mass reading will be off by the same amount. Systematic errors hurt accuracy but don't necessarily affect precision.
Example: You're determining solution concentration by titration. If your titrant was prepared correctly and you hit the endpoint right at the expected volume, your calculated concentration is accurate because it's close to the true value.
Precision
Precision describes how close repeated measurements are to each other, regardless of whether they're close to the true value.
Random Errors
Random errors cause unpredictable variation between measurements. They might come from slight fluctuations in temperature, small differences in how you read a meniscus, or electrical noise in a digital instrument. Random errors reduce precision.
Example: You weigh a substance five times and get 2.05 g each time. Those measurements are precise because they're consistent with one another, even if the true mass turns out to be 2.10 g.
Comparing Accuracy and Precision
The dartboard analogy is the fastest way to lock this in:
- Accurate and Precise: All darts clustered tightly on the bullseye.
- Accurate but Not Precise: Darts are spread out, but their average position is the bullseye.
- Precise but Not Accurate: All darts cluster tightly together, but away from the bullseye.
- Neither Accurate nor Precise: Darts scattered all over the board with no pattern.
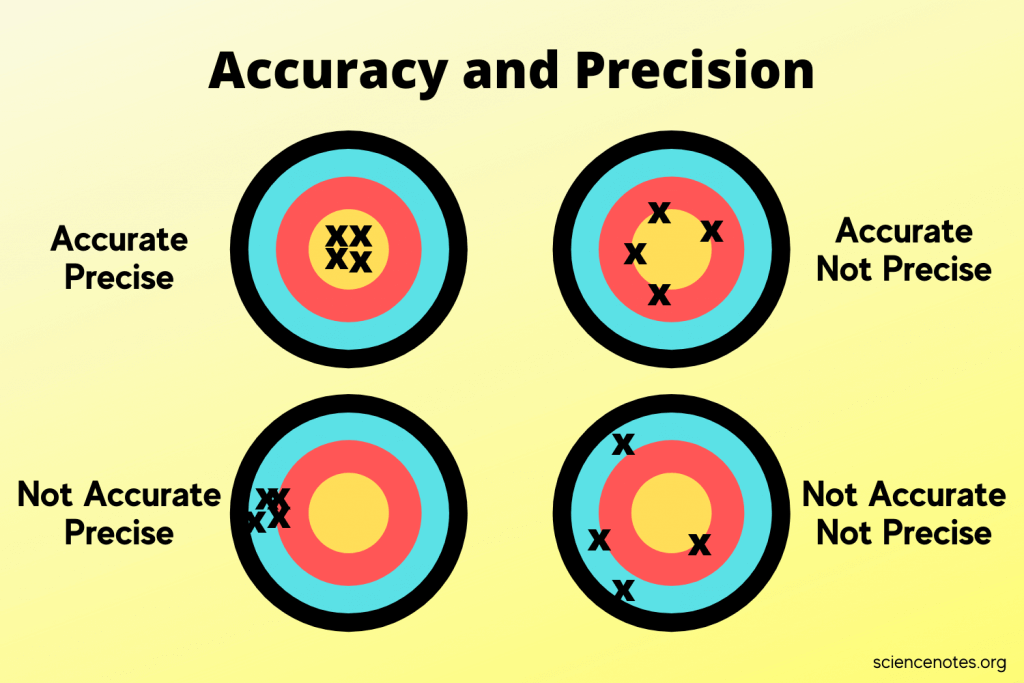
Graphic with four different targets, expressing the difference between accuracy and precision.
Image courtesy of ScienceNotes
The key distinction: precision is about reproducibility, accuracy is about correctness. You can be precise without being accurate (and vice versa).
Percent Error
Percent error quantifies how far your experimental value is from the accepted value, expressed as a percentage. Think of it as an accuracy score for a measurement.
Formula for Percent Error
The absolute value bars mean your answer is always positive. You're measuring the size of the error, not its direction.
Interpretation
- A low percent error (close to 0%) means your measurement is very close to the accepted value.
- A high percent error signals something went wrong. Check for systematic errors, procedural mistakes, or equipment issues.
Example Calculation
You measure the boiling point of water as 98°C. The accepted value at sea level is 100°C.
A 2.0% error is relatively small, so this measurement is fairly accurate.
Practice Problem
Calculate the percent error if you find a substance's density to be when its accepted density is .
That's less than 1%, which indicates a very accurate measurement.
Common mistake to avoid: The original version of this problem showed an answer of 88.3%, which is incorrect. Make sure you divide the absolute difference (0.07) by the true value (7.92) before multiplying by 100%.
Factors Influencing Measurement Reliability
Factors Affecting Accuracy
- Systematic Errors: Uncalibrated instruments or faulty equipment shift all results in one direction.
- Environmental Conditions: Temperature changes, humidity, or air currents can alter readings.
- Procedural Errors: Misreading a graduated cylinder, using improper technique, or skipping a step in a procedure.
Factors Affecting Precision
- Random Errors: Unpredictable variations with no consistent pattern, like slight hand tremors or electrical noise in sensors.
- Repeatability of Technique: Consistent technique across trials is essential for getting reproducible results.
- Equipment Resolution: An instrument that reads to 0.01 g can produce more precise data than one that reads to 0.1 g.
Improving Your Measurements
- Calibrate instruments regularly against known standards before collecting data.
- Replicate measurements by running multiple trials and averaging results, which helps reduce the impact of random errors.
- Refine your technique through practice so that each trial is performed as consistently as possible.