Scientific Notation and Metric System
Scientific notation and the metric system are two foundational tools in chemistry that let you work with numbers ranging from the subatomic to the astronomical. Mastering both will make nearly every calculation in this course faster and less error-prone.

Importance of Scientific Notation
Chemistry constantly throws extreme numbers at you. The mass of a single proton is about . The number of atoms in a sample can exceed . Writing these out every time is impractical, so we use scientific notation.
What is Scientific Notation?
Scientific notation expresses a number as a coefficient (a value between 1 and 10) multiplied by a power of ten. For example:
That's Avogadro's number, the number of particles in one mole of a substance. The coefficient is 6.02, and the exponent tells you how many places the decimal point has been shifted.
Why Use Scientific Notation?
- Simplifies calculations: Multiplying by is far easier than working with all those zeros longhand.
- Improves readability: You can immediately see the scale of a number from its exponent.
- Aids comparison: Comparing to is straightforward because you check exponents first, then coefficients.
Working with Numbers in Scientific Notation
Converting Standard Form to Scientific Notation
For numbers greater than or equal to 10:
Move the decimal point left until you have a number between 1 and 10. The number of places you moved becomes your positive exponent.
(The decimal moved 3 places to the left.)
For numbers less than 1:
Move the decimal point right until you have a number between 1 and 10. The number of places you moved becomes your negative exponent.
(The decimal moved 4 places to the right.)
Arithmetic Operations
Addition and Subtraction: The exponents must match before you can add or subtract the coefficients. If they don't match, adjust one of the numbers first.
Multiplication: Multiply the coefficients and add the exponents.
Notice that isn't proper scientific notation (the coefficient must be between 1 and 10), so you adjust it to .
Division: Divide the coefficients and subtract the exponents.
Application Exercises
- If there are molecules in one mole, how many are in half a mole?
- A solution has a concentration of . What is its concentration when diluted to twice its original volume?
When you double the volume without adding more solute, the concentration is halved:
(Note: M stands for molarity, which is .)
Mastering Metric Unit Conversions
The metric system is built on base units: grams (mass), liters (volume), and meters (length). Prefixes scale these base units by powers of ten, which makes converting between them straightforward.
Understanding Metric Prefixes
| Prefix | Symbol | Meaning | Example |
|---|---|---|---|
| kilo- | (1,000 ×) | 1 km = 1,000 m | |
| hecto- | (100 ×) | 1 hg = 100 g | |
| deka- | (10 ×) | 1 daL = 10 L | |
| (base) | — | (1 ×) | m, g, L |
| deci- | (0.1 ×) | 1 dg = 0.1 g | |
| centi- | (0.01 ×) | 1 cm = 0.01 m | |
| milli- | (0.001 ×) | 1 mg = 0.001 g | |
| micro- | 1 μm = 0.000001 m | ||
| nano- | 1 nm = 0.000000001 m |
A classic mnemonic for the bolded prefixes kilo through milli: King Henry Died By Drinking Chocolate Milk. The "B" stands for the base unit (meter, liter, gram, etc.).
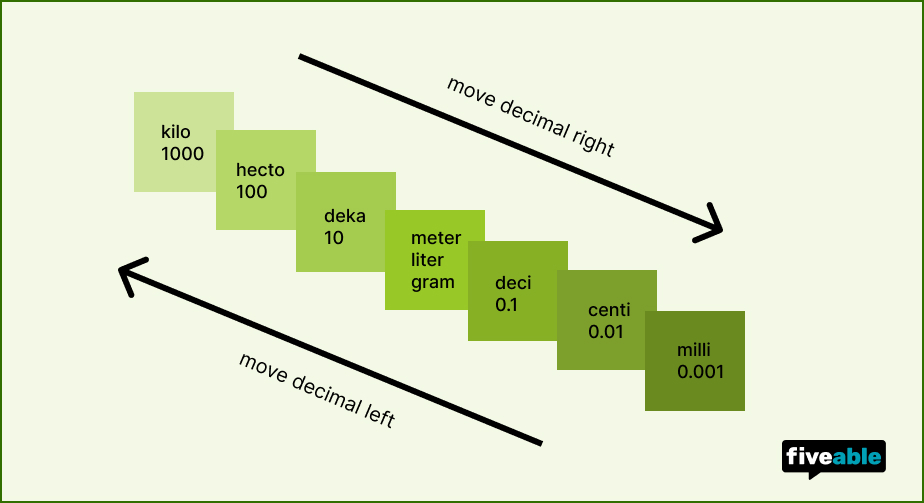
Visual ladder method showing conversion steps between units.
Image Credit to Fiveable
Conversion Techniques
Conversion Factors Method
Set up a ratio so the unwanted unit cancels and the desired unit remains.
Example: Convert to meters, using .
The "cm" cancels top and bottom, leaving you with meters.
The Ladder Method
Each step on the prefix ladder represents one power of ten. Count the number of steps between your starting prefix and your target prefix, then move the decimal point that many places.
Example: Kilometers to meters is 3 steps down the ladder (kilo → hecto → deka → base), so you multiply by , moving the decimal 3 places to the right.
Practice Exercise
Convert to grams and express the result in scientific notation. Since milli- means , there are 1,000 mg in 1 g.
Integration in Chemical Problem Solving
Real chemistry problems often combine metric conversions with scientific notation in a single calculation. The cleanest approach is to handle them one step at a time:
- Convert all measurements to the units you need (using conversion factors or the ladder method).
- Perform the arithmetic in scientific notation.
- Adjust your final coefficient so it falls between 1 and 10, and double-check that your units are correct.
Final Tips
- Watch your exponents during arithmetic. Adding when you should subtract (or vice versa) is the most common source of errors.
- Always include units in your final answer. A number without a unit is meaningless in chemistry.
- Practice converting in both directions (large to small and small to large). Speed with conversions saves real time on exams.
- Re-check proper form. Your coefficient should always be ≥ 1 and < 10. If it isn't, adjust the coefficient and exponent together.