Types of Radioactivity and Nuclear Reactions
Radioactivity is the process by which unstable atomic nuclei release energy by emitting radiation. Understanding the different types of radioactive decay is central to nuclear chemistry because each type changes the nucleus in a specific, predictable way. This section covers the major decay modes, how nuclear reactions differ from chemical reactions, and what factors determine nuclear stability.

Types of Radioactivity
Alpha Decay (α-decay)
What is an alpha particle?
An alpha particle is made up of two protons and two neutrons, making it identical to a helium-4 nucleus (). Alpha emission typically occurs in heavy elements (atomic number ≥ 83), such as uranium and radium, because their large nuclei experience strong proton-proton repulsion that destabilizes them.
How does it interact with matter?
Alpha particles have the lowest penetrating power of the common radiation types. Their large mass and +2 charge cause them to interact strongly with surrounding matter, so they lose energy quickly. A single sheet of paper or even the outer layer of skin can stop them.
Atomic changes:
When a nucleus emits an alpha particle, the mass number decreases by 4 and the atomic number decreases by 2. In equation form:
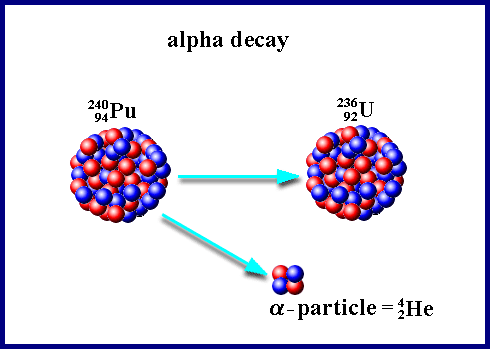
Image of alpha decay with a helium nucleus being ejected from a larger atom, resulting in a new element.
Image Courtesy of Nuceng
Beta Decay
Beta-minus Decay (β⁻-decay)
What happens during β⁻-decay?
A neutron inside an unstable, neutron-rich nucleus converts into a proton while emitting an electron (the beta particle) and an antineutrino. The key result: the atomic number increases by 1, but the mass number stays the same because the total number of nucleons hasn't changed.
Beta-plus Decay (β⁺-decay or Positron Emission)
What happens during β⁺-decay?
A proton inside an unstable, proton-rich nucleus converts into a neutron, releasing a positron (the antimatter counterpart of an electron) and a neutrino. The atomic number decreases by 1, while the mass number remains constant.
Comparing the two: β⁻ decay fixes a neutron excess (too many neutrons), while β⁺ decay fixes a proton excess (too many protons). Both shift the nucleus toward a more favorable neutron-to-proton ratio.
Gamma Decay (γ-decay)
Gamma rays are high-energy photons emitted when a nucleus drops from an excited energy state to a lower one. This type of decay does not change the mass number or the atomic number. It simply carries away excess energy, often occurring right after an alpha or beta decay leaves the daughter nucleus in an excited state.
Gamma radiation has the highest penetrating power of the three common types. It takes several centimeters of lead or thick concrete to significantly reduce its intensity.
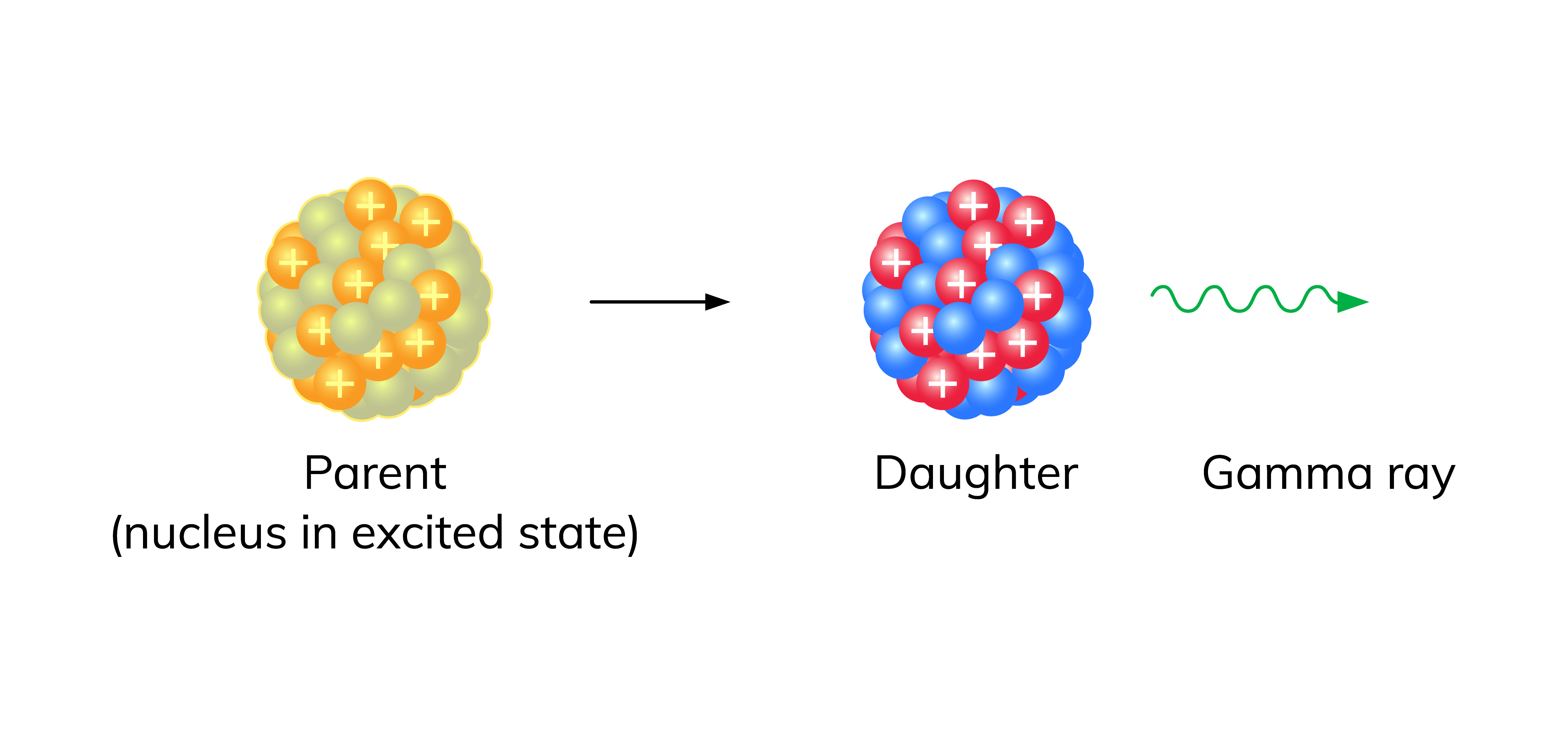
Image of gamma decay with high-energy photons being released from an excited nucleus returning to its ground state.
Image Courtesy of Wizeprep
Electron Capture
In electron capture, an inner orbital electron is pulled into the nucleus, where it combines with a proton to form a neutron and release a neutrino. The result is the same net change as β⁺ decay: the atomic number decreases by 1 and the mass number stays the same. Electron capture tends to occur in proton-rich nuclei, especially heavier ones where positron emission is energetically less favorable.
Neutron Emission
Some extremely neutron-rich nuclei can achieve greater stability by directly ejecting one or more neutrons. This decreases the mass number by 1 (per neutron lost) but leaves the atomic number unchanged, since no protons are involved.
Nuclear Reactions vs. Chemical Reactions
Nuclear reactions involve changes within the nucleus itself, while chemical reactions only rearrange the electrons surrounding the nucleus. A few key differences to remember:
- Energy scale: Nuclear reactions release or absorb millions of times more energy than chemical reactions. A single fission event releases roughly a million times the energy of burning one molecule of fuel.
- Particles involved: Chemical reactions involve valence electrons. Nuclear reactions involve protons, neutrons, and the particles emitted during decay.
- Element identity: Chemical reactions never change one element into another. Nuclear reactions can, because they alter the number of protons.
Fission
Fission is the splitting of a heavy nucleus (such as ) into two smaller nuclei, along with a few free neutrons and a large release of energy. This is the process used in nuclear power plants and nuclear weapons. The free neutrons can trigger further fission events, creating a chain reaction.
Fusion
Fusion is the combining of lighter nuclei (such as hydrogen isotopes) to form a heavier nucleus, releasing even more energy per unit mass than fission. Fusion powers stars like our Sun, where extreme temperatures (millions of degrees) and pressures force nuclei close enough to overcome their electrostatic repulsion.
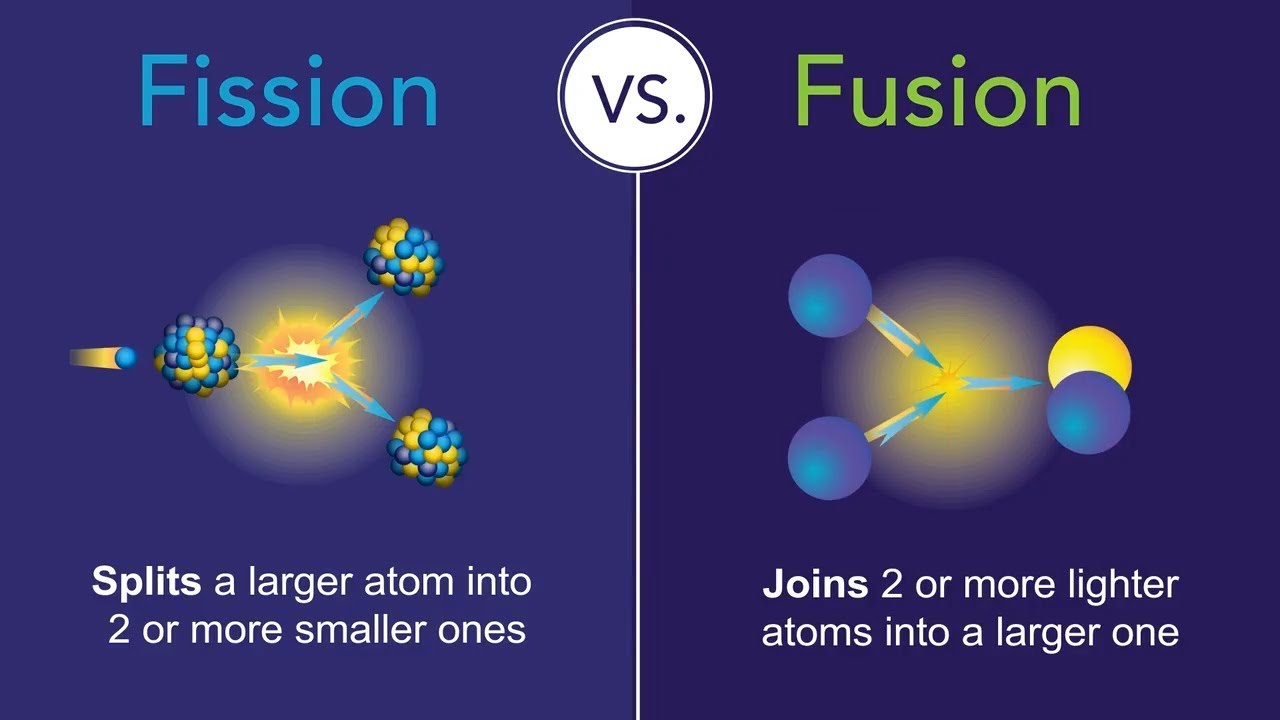
Fusion vs Fission diagram
Image Courtesy of Department of Energy
Stability and Decay Factors
Several factors determine whether a given isotope is stable or radioactive.
Nuclear Binding Energy
Binding energy is the energy required to completely separate all the protons and neutrons in a nucleus. The higher the binding energy per nucleon, the more stable the nucleus. Iron-56 () sits near the peak of the binding energy curve, which is why nuclei lighter than iron tend to undergo fusion and nuclei heavier than iron tend to undergo fission to move toward greater stability.
Magic Numbers
Certain numbers of protons or neutrons produce unusually stable nuclei. These magic numbers (2, 8, 20, 28, 50, 82, and 126) correspond to completely filled nuclear energy shells, similar in concept to filled electron shells in atomic structure. Nuclei with a magic number of protons and a magic number of neutrons (called "doubly magic") are exceptionally stable. For example, has 2 protons and 2 neutrons, both magic numbers.
External Factors Influencing Nuclear Reactions
Under normal Earth conditions, radioactive decay rates are essentially constant and unaffected by temperature, pressure, or chemical environment. However, extreme conditions found elsewhere in the universe can drive nuclear reactions that don't occur naturally on Earth.
Stellar Nucleosynthesis
Inside stars, temperatures reach millions of kelvin and pressures are enormous. Under these conditions, fusion reactions build heavier elements from lighter ones. Elements up to iron are produced in the cores of massive stars, while elements heavier than iron are primarily formed during supernova explosions through rapid neutron capture processes.
Radioactivity & Nuclear Reactions Practice Questions
- A sample of americium-241 undergoes alpha decay. What are the products of this reaction?
Explanation: When americium-241 emits an alpha particle (), subtract 2 from the atomic number and 4 from the mass number of the parent:
- Atomic number: (neptunium)
- Mass number:
Answer: The products are neptunium-237 () and an alpha particle ().
- Describe how beta decay affects the stability of a nucleus. Consider both β⁻ and β⁺ decays.
Explanation: Both forms of beta decay adjust the neutron-to-proton (n/p) ratio to move the nucleus closer to the band of stability.
- β⁻ decay occurs in neutron-rich nuclei (n/p ratio too high). Converting a neutron into a proton lowers the ratio toward a stable value.
- β⁺ decay occurs in proton-rich nuclei (n/p ratio too low). Converting a proton into a neutron raises the ratio toward a stable value.
In both cases, the nucleus ends up with a more balanced n/p ratio and greater overall stability.
- Explain why neutron emission might occur more frequently in elements that lie above the band of stability on the chart of nuclides.
Explanation: Nuclei that plot above the band of stability have too many neutrons relative to their protons. Emitting a neutron directly reduces the neutron count, lowering the n/p ratio and shifting the nucleus back toward the band of stability. This is the most direct way for an extremely neutron-rich nucleus to correct its imbalance.
- Why do we say that elements with magic numbers are especially stable?
Explanation: Magic numbers (2, 8, 20, 28, 50, 82, 126) represent completely filled nuclear shells, analogous to noble gas electron configurations in atomic chemistry. A filled shell means nucleons are in a particularly low-energy, tightly bound arrangement. Nuclei with magic numbers of protons or neutrons require more energy to remove a nucleon, making them resistant to decay and exceptionally stable.