Intermolecular Forces and Properties
Intermolecular forces (IMFs) are the attractions between molecules that determine physical properties like boiling point, solubility, and viscosity. Understanding IMFs is essential for predicting how substances behave, and this topic shows up constantly in later units on solutions, gases, and thermodynamics.

Identifying Intermolecular Forces (IMFs)
IMFs are the attractive forces between molecules (or between molecules and ions). They're much weaker than intramolecular forces (the covalent or ionic bonds within a molecule or compound), but they control physical state, boiling point, solubility, and more.
A useful hierarchy to memorize, from weakest to strongest:
London Dispersion Forces < Dipole-Dipole < Hydrogen Bonding < Ion-Dipole
Van der Waals Forces
London Dispersion Forces (LDFs) are present in all molecules and atoms, but they're the only IMF available to nonpolar substances. They arise from temporary, instantaneous dipoles: at any given moment, electrons aren't perfectly evenly distributed, so one side of a molecule becomes slightly negative and the other slightly positive. That temporary dipole induces a dipole in a neighboring molecule, creating a brief attraction.
Two things to remember about LDFs:
- Larger molecules (higher molar mass) = stronger LDFs. More electrons means larger, more easily formed temporary dipoles. That's why is a solid at room temperature while is a gas.
- Molecular shape matters. Long, straight molecules have more surface area for contact than compact, spherical ones, so they experience stronger LDFs. For example, n-pentane (straight chain) has a higher boiling point than neopentane (compact/branched), even though they have the same molecular formula ().
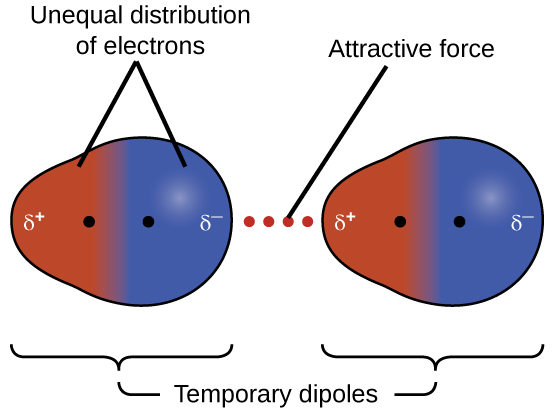
Image showing temporary dipoles and the temporary attractive force.
Image courtesy of Wikimedia Commons
Dipole-Dipole Interactions occur between polar molecules that have permanent dipoles. The partially positive end () of one molecule is attracted to the partially negative end () of a neighboring molecule. These are stronger than LDFs (for molecules of comparable size) because the attraction is constant, not temporary.
For a molecule to have dipole-dipole interactions, it must be polar, meaning it has polar bonds and an asymmetric geometry so the bond dipoles don't cancel out. For example, is polar and exhibits dipole-dipole interactions, while is nonpolar (linear, dipoles cancel) and does not.
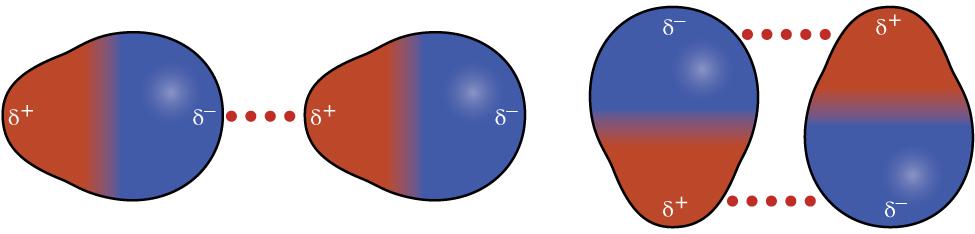
Image showing the permanent dipoles attracting adjacent oppositely charged regions
Image courtesy of NDLA.
Special Cases
Hydrogen Bonding is a particularly strong type of dipole-dipole interaction. It occurs when hydrogen is bonded directly to nitrogen, oxygen, or fluorine (remember: "N, O, F"). These three elements are so electronegative that they pull electron density far away from hydrogen, leaving H with a large partial positive charge. That exposed H is then strongly attracted to a lone pair on an N, O, or F of a neighboring molecule.
⚠️ Hydrogen bonds are NOT actual covalent bonds. The name is misleading. They're still intermolecular attractions, just unusually strong ones. Water () is the classic example: each water molecule can form up to four hydrogen bonds with its neighbors, which is why water has an unusually high boiling point (100°C) for such a small molecule.
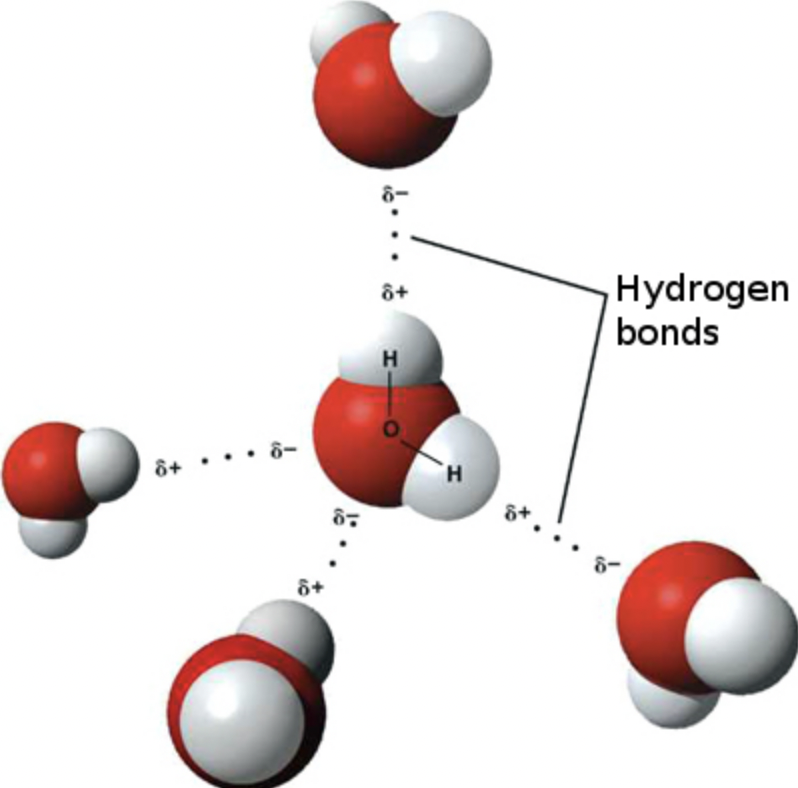
Image of hydrogen bonding between water molecules
Image courtesy of Picryl
Ion-Dipole Forces are the strongest IMF you'll encounter in this unit. They occur when an ion interacts with a polar molecule. The full charge on the ion creates a much stronger attraction than a partial charge would. This is the key force at work when ionic compounds dissolve in water: ions attract the oxygen end of water, and ions attract the hydrogen end.
IMFs in Action
Boiling Point and Melting Point
Boiling and melting require overcoming intermolecular forces to separate molecules from each other. Stronger IMFs mean more energy (heat) is needed, which means higher boiling and melting points.
When comparing boiling points of different substances:
- Identify the IMFs present in each substance.
- If the IMF types differ, the substance with the stronger type of IMF generally has the higher boiling point.
- If the IMF types are the same (e.g., both are nonpolar with only LDFs), the substance with the higher molar mass typically wins.
Solubility
The guiding principle is "like dissolves like." Polar solvents (like water) dissolve polar and ionic solutes well because the solvent molecules can form IMFs with the solute that are comparable in strength to the ones being broken. Nonpolar solvents (like hexane) dissolve nonpolar solutes for the same reason.
This is why oil and water don't mix: oil is nonpolar and can't form strong enough interactions with polar water molecules to overcome water's hydrogen bonding network.
Viscosity and Surface Tension
Viscosity is a liquid's resistance to flow. Stronger IMFs hold molecules together more tightly, making the liquid flow more slowly. Honey has high viscosity (strong IMFs, plus large tangled molecules), while gasoline flows easily (weak LDFs, small molecules).
Surface tension is the tendency of a liquid's surface to resist being broken. Molecules at the surface are pulled inward by IMFs from molecules below and beside them, creating a "skin" effect. Stronger IMFs produce higher surface tension. Water has unusually high surface tension because of its extensive hydrogen bonding, which is why small insects can walk on it.
Practice Questions
- Which has a higher boiling point: methane () or water (), and why?
- Why does salt () dissolve well in water but not in vegetable oil?
Answer Key
- Water has a much higher boiling point (100°C vs. ). Water molecules experience hydrogen bonding, which requires significantly more energy to overcome than the London dispersion forces between nonpolar molecules.
- is ionic. Water is polar and can stabilize the separated and ions through strong ion-dipole forces. Vegetable oil is nonpolar and cannot form comparably strong interactions with the ions, so the energy cost of breaking apart the ionic lattice isn't compensated.
Real-World Applications of IMFs
| Water's Unique Properties | Hydrogen bonding causes water to expand upon freezing (ice is less dense than liquid water) and gives it an unusually high boiling point for its molar mass. Both properties are critical for sustaining life on Earth. |
|---|---|
| Biological Systems | Hydrogen bonds hold together the two strands of the DNA double helix, and London dispersion forces contribute to protein folding by stabilizing hydrophobic cores. |
| Materials Science | Engineers manipulate IMFs to design materials with specific flexibility, strength, or biodegradability. For example, choosing polar vs. nonpolar polymer chains changes how a plastic behaves. |
| Environmental Science | Knowing a pollutant's IMFs helps predict whether it will dissolve in groundwater (polar) or accumulate in fatty tissue (nonpolar), which guides cleanup strategies. |
Conceptual Questions
-
Compare the boiling points of hexane () and ethanol (). What role do IMFs play, given that hexane actually has the higher molar mass?
-
Which would have greater surface tension: acetone () or glycerol ()? Why?
-
If DNA didn't use hydrogen bonding between base pairs, what consequences might that have for genetic information storage and replication?
-
How might knowledge of IMFs guide the design of a laundry detergent that needs to remove both grease stains and water-soluble stains?