Half-life and Radioactive Decay
Half-life describes how long it takes for half of a radioactive sample to decay. It's one of the most practical concepts in nuclear chemistry because it connects to everything from dating ancient fossils to treating cancer. This guide covers the core concept, the math behind decay calculations, and real-world applications.

⌛The Concept of Half-Life in Radioactive Decay
What is Half-Life?
Half-life () is the time required for half of the radioactive nuclei in a sample to undergo decay. Radioactive decay is not linear; it's exponential. That means the quantity of undecayed material decreases by half during each half-life period, producing a rapid decline early on that gradually slows over time.
💡 Imagine you had 1000 atoms of a radioactive isotope. After one half-life, 500 remain. After two half-lives, 250 remain. After three, 125. Each interval cuts the remaining amount in half.
The decay constant () represents the probability per unit time that a given atom will decay. It's inversely related to half-life:
A large decay constant means the isotope decays quickly (short half-life). A small decay constant means it's more stable (long half-life).
Different Isotopes, Different Half-Lives
Half-lives vary enormously across isotopes. Polonium-214 has a half-life of about 164 microseconds, while uranium-238's half-life is roughly 4.5 billion years. This range reflects huge differences in nuclear stability.
This variability matters for practical applications. For radiometric dating, scientists choose an isotope whose half-life is appropriate for the timescale they're measuring. Carbon-14 ( ≈ 5,730 years) works well for archaeological artifacts up to ~50,000 years old, while uranium-lead dating handles geological timescales of billions of years.
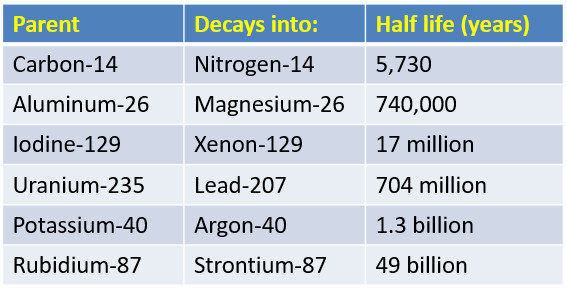
Chart showcasing different isotopes with their corresponding half-lives.
Image Courtesy of StickMan Physics
✍🏼Calculations Involving Half-Life
The Formula for Radioactive Decay
The exponential decay formula lets you calculate how many undecayed atoms remain after any amount of time:
Where:
- is the number of undecayed atoms at time
- is the initial number of atoms
- is the decay constant (in units of inverse time, like )
- is the time elapsed
You can also write this in terms of half-lives, which is often more intuitive:
Both formulas give the same result. The second version is especially handy when the elapsed time is a whole number of half-lives.
❓Practice Problem: If you start with 10,000 atoms and , how many atoms remain undecayed after 5 hours?
Step 1: Identify your values.
Step 2: Notice that means . So 5 hours is exactly 5 half-lives.
Step 3: Substitute into the formula.
You can verify this with the half-life version: after 5 half-lives, . Since you can't have half an atom, approximately 313 atoms remain.
Activity and Decay Rates
Activity () is the rate at which a sample decays, measured in disintegrations per second. The SI unit is the becquerel (Bq), where 1 Bq = 1 decay per second.
Activity is directly proportional to the number of undecayed atoms. As atoms decay and decreases, the activity drops too. This is why radioactive samples become less "active" over time.
Since decreases exponentially, activity does too:

Image of example problem
Image Courtesy of Expii
☢️ Real-world Applications of Half-Life and Radioactive Decay
🗓️ Radiometric Dating Techniques
Scientists compare the current amount of a radioactive isotope in a sample to its expected initial quantity, then use the decay equations to calculate the sample's age. Carbon-14 dating works for organic materials up to about 50,000 years old. For much older geological samples, uranium-lead dating (using with its 4.5-billion-year half-life) can determine the age of rocks and even the Earth itself.
🏥 Medical Diagnostics and Treatment
Radioisotopes play a critical role in medicine. For diagnostics, PET scans use fluorine-18 ( ≈ 110 minutes), which decays fast enough to produce useful images but doesn't linger in the body. For treatment, iodine-131 ( ≈ 8 days) treats thyroid disorders because the thyroid selectively absorbs iodine, and the radiation destroys diseased cells while limiting damage to surrounding tissue.
🔋 Nuclear Energy Production
Understanding decay is central to nuclear energy:
- Fission splits large unstable nuclei (like ), releasing enormous energy. Nuclear reactors use controlled fission with control rods to regulate the reaction rate.
- Fusion combines small nuclei (like hydrogen isotopes), which is what powers the sun. Practical fusion reactors remain a major research goal.
🌱 Safety and Environmental Considerations
Half-life directly determines how long nuclear waste remains hazardous. Some fission products have half-lives of just days, while others (like plutonium-239, ≈ 24,100 years) require secure storage for thousands of years. Knowing an isotope's half-life is essential for designing storage facilities and setting safety timelines.
Think about it: if a waste isotope has a half-life of 30 years, roughly how many years until its activity drops to less than 1% of its original level? (Hint: when is about 7 half-lives, so around 210 years.)
❓Practice Questions for Half-life and Radioactive Decay
Problem: A radioactive material has a half-life of one hour. If the initial activity is 1200 Bq, what will its activity be after two hours?
Step 1: Write the decay formula in terms of half-life:
Step 2: Identify your values.
Step 3: Substitute and solve.
After two hours (two half-lives), the activity decreases to 300 Bq. You can check this intuitively: after one half-life it's 600 Bq, after two it's 300 Bq.
When working through decay problems, always confirm that your units for and match (both in hours, both in seconds, etc.). A unit mismatch is the most common source of errors in these calculations.