Biosynthesis of Squalene and Lanosterol
Steroid biosynthesis begins with the construction of squalene from two molecules of farnesyl diphosphate (FPP), followed by its conversion to lanosterol. Lanosterol is the first molecule with the characteristic four-ring steroid skeleton, making it the direct precursor to cholesterol and all downstream steroid hormones. Understanding these steps connects the isoprenoid pathway you've already seen to the production of biologically critical steroids.
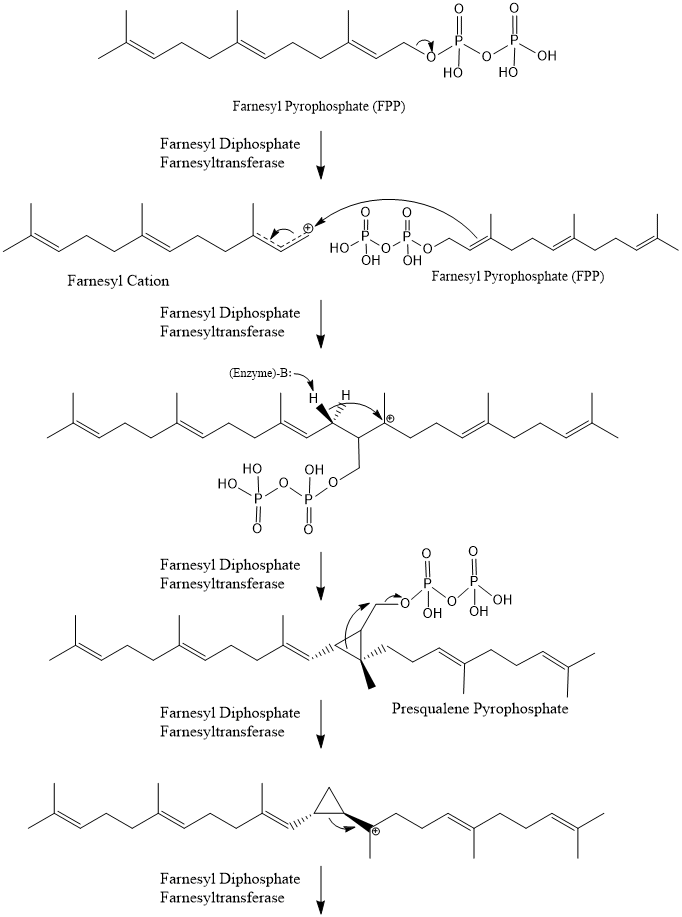
Biosynthesis of Squalene
Squalene is a 30-carbon linear triterpene assembled from two 15-carbon FPP molecules. The enzyme squalene synthase catalyzes this transformation in two distinct stages:
- Two FPP molecules are joined in a head-to-head fashion. The diphosphate group is eliminated from one FPP molecule, and the two carbon skeletons are linked to form presqualene diphosphate (PSPP), a cyclopropane-containing intermediate.
- PSPP then undergoes a reductive rearrangement: NADPH donates a hydride that reduces and eliminates the remaining diphosphate group, the cyclopropane ring opens, and the 30-carbon chain of squalene is released.
The head-to-head joining here is distinct from the head-to-tail condensations used earlier in the isoprenoid pathway (e.g., when building FPP from isopentenyl units). That difference is worth remembering for exams.
Mechanism of Squalene Epoxidation
Before cyclization can occur, squalene must be activated by epoxidation at its 2,3-double bond. The enzyme squalene epoxidase, a flavoprotein monooxygenase, carries out this stereospecific reaction.
The catalytic cycle works as follows:
- The enzyme's flavin cofactor (FAD) is reduced by NADPH, forming .
- Molecular oxygen () binds to the reduced flavin, generating a reactive flavin C4a-hydroperoxide intermediate.
- One oxygen atom from the hydroperoxide is transferred to the 2,3-double bond of squalene, forming 2,3-oxidosqualene (an epoxide).
- The second oxygen atom is reduced to water, regenerating the oxidized FAD cofactor.
The epoxidation is stereospecific: the oxygen atom is delivered to the si face of the 2,3-double bond. This stereochemistry is critical because it dictates how oxidosqualene folds and cyclizes in the next step.
Cyclization of Oxidosqualene to Lanosterol
The conversion of the linear oxidosqualene into the tetracyclic lanosterol skeleton is one of the most remarkable single-enzyme reactions in biochemistry. Lanosterol synthase (also called oxidosqualene cyclase) catalyzes the entire process.
The reaction proceeds through concerted electrophilic additions followed by carbocation rearrangements:
-
The enzyme protonates the epoxide ring of oxidosqualene, opening it to generate a carbocation at C-2.
-
This carbocation triggers a cascade of electrophilic additions across the folded polyene chain, forming the A, B, and C rings of the steroid nucleus in rapid succession.
-
The D ring is formed through a final electrophilic addition, initially producing a protosteryl cation with the complete four-ring system.
-
A series of 1,2-shifts then rearrange the protosteryl cation to give the lanosterol skeleton:
- Hydride shift from C-17 to C-20
- Methyl shift from C-14 to C-13
- Hydride shift from C-9 to C-8
-
The final carbocation is quenched by deprotonation, yielding lanosterol.
These 1,2-shifts convert the initial anti-Markovnikov ring junction geometry of the protosteryl cation into the thermodynamically more stable trans-fused ring system found in lanosterol. The enzyme controls every stereochemical outcome by holding the substrate in a precise chair-boat-chair-boat conformation within its active site.
Lanosterol then serves as the branch point for further modifications: in animals, it is converted to cholesterol through roughly 19 additional enzymatic steps, and cholesterol itself is the precursor to testosterone, estradiol, cortisol, and other steroid hormones.
Regulation of Steroid Biosynthesis
The flux through steroid biosynthesis is tightly controlled at the level of the mevalonate pathway, which supplies the isoprenoid building blocks for squalene.
- HMG-CoA reductase catalyzes the rate-limiting step of the mevalonate pathway (reduction of HMG-CoA to mevalonate). This is the target of statin drugs, which lower cholesterol by inhibiting this enzyme.
- Sterol regulatory element-binding proteins (SREBPs) are transcription factors that sense intracellular cholesterol levels. When cholesterol is low, SREBPs are proteolytically activated and travel to the nucleus, where they upregulate genes encoding HMG-CoA reductase, squalene synthase, and other biosynthetic enzymes.
- When cholesterol is abundant, SREBPs remain inactive, and HMG-CoA reductase is also subject to accelerated degradation and phosphorylation-dependent inactivation.
Together, these mechanisms form a feedback loop that matches steroid production to the cell's actual needs.