Amino Acids and Peptide Bonds
Peptide Sequence Construction
Amino acids are identified using two standard abbreviation systems. One-letter abbreviations use a single uppercase letter (G for Glycine, S for Serine), while three-letter abbreviations capitalize only the first letter (Gly for Glycine, Ser for Serine). You'll need to know both, since one-letter codes show up in compact sequence notation and three-letter codes appear in more detailed representations.
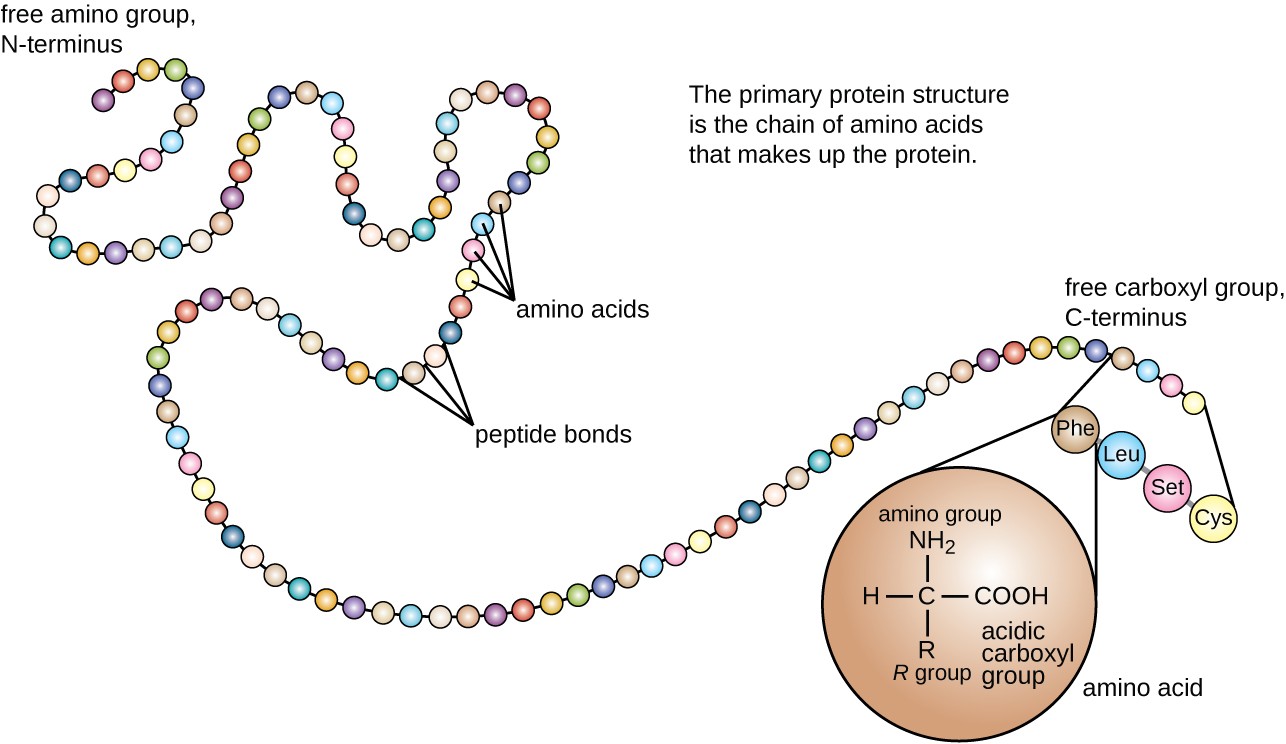
Peptide sequences are always written from the N-terminus (the end with a free amino group, ) to the C-terminus (the end with a free carboxyl group, ). This convention matters because reversing the sequence gives you a completely different molecule.
- One-letter example: AGCDE = Ala-Gly-Cys-Asp-Glu
- Three-letter example: Ala-Gly-Cys-Asp-Glu = Alanine-Glycine-Cysteine-Aspartic Acid-Glutamic Acid
The specific order of amino acids in a peptide or protein is its primary structure. Every higher level of protein organization depends on getting this sequence right.
Peptide Bond Formation
A peptide bond forms through a condensation (dehydration) reaction between two amino acids. Here's the step-by-step process:
- The carboxyl group () of one amino acid reacts with the amino group () of the next.
- The carboxyl group loses and the amino group loses .
- A water molecule () is released as a byproduct.
- A covalent C–N bond forms between the carbonyl carbon of the first amino acid and the nitrogen of the second.
A few things to note about the resulting bond:
- Peptide bonds have partial double bond character due to resonance between the carbonyl oxygen and the nitrogen lone pair. This means the bond is planar and doesn't rotate freely, which constrains the shape of the backbone.
- Repeating this reaction builds the peptide backbone, a chain of alternating units.
- Each amino acid within the chain is called an amino acid residue, and the side chains (R groups) extend outward from the backbone. These R groups are what give each residue its unique chemical properties.
Disulfide Bridges
Disulfide Bridges in Proteins
Cysteine (Cys, C) is the only standard amino acid with a thiol group () on its side chain, and this makes it uniquely capable of forming disulfide bridges.
A disulfide bridge forms when two cysteine thiol groups undergo oxidation, losing their hydrogens and creating a covalent sulfur-sulfur bond (). This reaction requires an oxidizing agent.
Disulfide bridges can occur in two contexts:
- Intramolecular: between two cysteines in the same chain, contributing to tertiary structure
- Intermolecular: between cysteines on different chains, helping stabilize quaternary structure by linking separate polypeptide subunits together
These bonds are important for locking a protein into its correct three-dimensional shape. They're especially common in secreted proteins (like insulin and antibodies) that need extra stability outside the cell. Disulfide bridges can be broken by reducing agents such as dithiothreitol (DTT), which converts the bond back into two free groups.

Protein Structure and Stability
Secondary Structure
Secondary structure refers to local, repeating folding patterns in the protein backbone. These patterns are stabilized by hydrogen bonds between backbone atoms (specifically, between the of one residue and the of another).
The two most common types:
- Alpha helix (-helix): The backbone coils into a right-handed spiral. Hydrogen bonds form between every and the four residues ahead along the chain, running parallel to the helix axis.
- Beta sheet (-sheet): The backbone stretches into extended strands that lie side by side. Hydrogen bonds form between adjacent strands (which can run in the same direction, called parallel, or in opposite directions, called antiparallel), creating a pleated appearance.
Factors Influencing Protein Stability
Two major forces keep a protein folded in its functional shape:
- Hydrophobic effect: Nonpolar amino acid side chains tend to cluster together in the interior of the protein, away from water. This burial of hydrophobic residues is the single largest driving force behind protein folding.
- Hydrogen bonding: Beyond stabilizing secondary structure, hydrogen bonds between side chains, between side chains and backbone atoms, and between the protein and surrounding water all contribute to overall stability.
Other stabilizing interactions include ionic interactions (salt bridges) between charged side chains and van der Waals forces between tightly packed residues, though for this unit, the hydrophobic effect and hydrogen bonding are the most important to understand.