Resonance Forms
Resonance forms represent the different ways you can draw the electron distribution in a molecule without moving any atoms. No single Lewis structure captures where the electrons actually are; the real molecule is a resonance hybrid, a blend of all valid resonance forms. Understanding the rules for drawing these forms is essential for predicting stability, reactivity, and charge distribution throughout organic chemistry.
Resonance Forms vs. Molecular Structures
A resonance form is a hypothetical Lewis structure that shows one possible arrangement of electrons. The actual molecule doesn't flip between these forms. Instead, the true structure is a hybrid, with bond lengths, bond strengths, and charge distributions that are intermediate between the individual forms you draw.
- Resonance forms share the same arrangement of atoms but differ in how electrons (bonds and lone pairs) are distributed.
- The real molecular structure has bond lengths and partial charges that are averages of the contributing forms. For example, benzene's six C–C bonds are all the same length (1.40 Å), halfway between a typical single bond (1.54 Å) and a double bond (1.34 Å).
- A double-headed arrow () placed between structures indicates resonance. Don't confuse this with equilibrium arrows (), which describe two separate species interconverting.
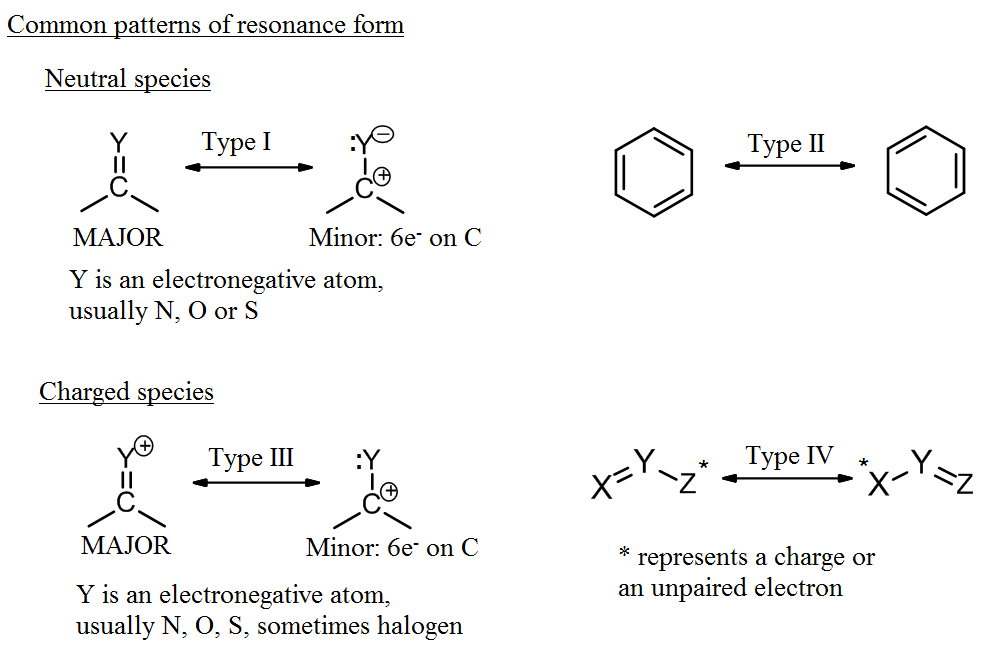
Electron Movement in Resonance
Curved arrows are the language for showing how electrons redistribute between resonance forms. The atoms stay locked in place; only electrons move.
- The tail of a curved arrow starts at the electron source (a lone pair, a bond, or sometimes a charge).
- The head of the arrow points to where those electrons go (forming a new bond or becoming a lone pair).
- A double-headed curved arrow moves two electrons. This is the most common type in resonance and represents lone pair donation into a bond or a pi bond shifting to an adjacent position.
- A single-headed (fishhook) arrow moves one electron. You'll see these in radical chemistry, not in typical resonance drawings.
A common beginner mistake is drawing arrows that move atoms. If your arrow implies an atom has changed position, the structure isn't a valid resonance form.

Equivalent vs. Non-Equivalent Resonance Forms
Equivalent resonance forms have the same arrangement of bonds and lone pairs, just on different atoms. They contribute equally to the hybrid and have identical energies. Benzene is the classic example: its two Kekulé structures are mirror images of each other and contribute equally.
Non-equivalent resonance forms differ in how bonds and charges are distributed, so they don't contribute equally. The more stable form contributes more to the hybrid. For instance, in the carbonate ion (), all three resonance forms are actually equivalent by symmetry, but in a molecule like an enolate, the forms placing negative charge on oxygen vs. carbon are non-equivalent.
To rank non-equivalent resonance forms from most to least stable, apply these rules in rough order of importance:
- More covalent bonds = more stable. A form with a greater number of bonds is lower in energy because covalent bonds release energy when they form.
- Complete octets on all atoms (especially C, N, O). Forms where second-row atoms satisfy the octet rule are strongly preferred. A carbon with only six electrons (a carbocation) is a significant destabilizing feature.
- Fewer formal charges = more stable. A resonance form with no formal charges is generally better than one with separated positive and negative charges.
- Negative charge on the more electronegative atom. If a form must carry a negative formal charge, it's more stable when that charge sits on a more electronegative atom (e.g., oxygen rather than carbon). Likewise, positive charge is better tolerated on less electronegative atoms.
The resonance hybrid is always more stable than any single contributing form. This extra stabilization is called resonance stabilization energy.
Additional Resonance Concepts
- Conjugation occurs when alternating single and multiple bonds allow electron delocalization over a larger portion of the molecule. The more extended the conjugation, the greater the stabilization (e.g., 1,3-butadiene vs. an isolated diene).
- Aromaticity is a special case of cyclic conjugation. A molecule is aromatic when it is cyclic, planar, fully conjugated, and contains pi electrons (Hückel's rule). Aromatic compounds like benzene have unusually large resonance stabilization.
- The mesomeric effect describes how electron density is pushed or pulled through a conjugated pi system by electron-donating or electron-withdrawing groups.
- Hyperconjugation is a subtler stabilization that arises from overlap between filled sigma bonding orbitals and adjacent empty or partially filled p orbitals. It helps explain why more substituted carbocations are more stable.