1H NMR Spectroscopy
Interpretation of 1H NMR Spectra
A 1H NMR spectrum encodes four pieces of structural information, and you need all four to solve a structure:
- Number of signals tells you how many unique hydrogen environments exist in the molecule. Equivalent hydrogens (related by symmetry) give a single signal.
- Integration tells you the relative number of hydrogens producing each signal. A 1:2:3 integration ratio means the signals correspond to groups of, say, 1H, 2H, and 3H.
- Chemical shift () in ppm reflects the electronic environment around each set of hydrogens. Nearby electronegative atoms or pi systems shift signals downfield (higher ), while electron-donating alkyl groups keep signals upfield (lower ).
- Splitting pattern reveals how many neighboring, non-equivalent hydrogens are coupled to the signal. This follows the rule: a hydrogen with neighboring hydrogens splits into lines.
Common splitting patterns:
- Singlet (s): no adjacent non-equivalent H's
- Doublet (d): one neighboring H
- Triplet (t): two neighboring H's
- Quartet (q): three neighboring H's
- Multiplet (m): complex or overlapping splitting (common for aromatic rings)
A classic example is the ethyl group (): the appears as a quartet (3 neighboring H's from ) and the appears as a triplet (2 neighboring H's from ), with a 2:3 integration ratio.
Chemical Shift Trends and Functional Group Identification
Chemical shift values are your main tool for identifying functional groups. Here are the key ranges to know:
| Region ( ppm) | Type of Hydrogen | Examples |
|---|---|---|
| 0–2 | Alkyl (, , ) | Cyclohexane (~1.4), TMS (0.0) |
| 2–4.5 | H's on C next to electronegative atoms or systems | , , benzylic H's |
| 4.5–6.5 | Vinylic () | Styrene vinyl H's |
| 6.5–8.5 | Aromatic | Benzene (7.27), toluene, naphthalene |
| 9–10 | Aldehyde () | Benzaldehyde (~9.9) |
| 10–12 | Carboxylic acid () | Acetic acid (~11.4) |
A few functional groups deserve special attention:
- Alcohols () and amines () have exchangeable protons. Their chemical shifts are variable (solvent-dependent, concentration-dependent) and they often appear as broad singlets. A shake will make these peaks disappear, which is a useful diagnostic trick.
- Aldehydes are easy to spot: a distinctive singlet or very small doublet far downfield around 9–10 ppm.

Coupling Constants and Stereochemistry
The coupling constant (), measured in Hz, carries stereochemical information beyond what chemical shift alone provides.
Vicinal coupling () depends on the dihedral angle between the two coupled hydrogens, as described by the Karplus relationship:
- Trans alkenes: large values, typically 12–18 Hz
- Cis alkenes: smaller values, typically 6–12 Hz
- Freely rotating single bonds: intermediate , usually 6–8 Hz
Be careful with the cis/trans coupling constant values. For alkenes, trans coupling is larger than cis. This is the opposite of what some students assume.
Geminal coupling () occurs between two hydrogens on the same carbon when those hydrogens are diastereotopic (non-equivalent due to a nearby chiral center). For example, the group in lactic acid has diastereotopic protons that couple to each other.
Distinguishing Isomers by NMR
One of the most practical uses of 1H NMR is telling isomers apart.
Constitutional isomers have different connectivity, so their spectra usually look completely different. The four isomers of butanol (1-butanol, 2-butanol, isobutanol, tert-butanol) each produce a distinct spectrum with different numbers of signals, splitting patterns, and chemical shifts. Tert-butanol, for instance, shows a large 9H singlet for its three equivalent methyl groups.
Symmetry matters. Para-disubstituted benzenes have fewer unique aromatic H environments than ortho-disubstituted ones, so they produce fewer signals in the aromatic region.
Diastereomers are stereoisomers that are not mirror images. They have genuinely different chemical environments, so their spectra differ in chemical shifts and coupling constants. Cis- and trans-decalin, for example, show different values for ring junction protons.
Enantiomers are mirror images and produce identical NMR spectra in ordinary (achiral) solvents. To distinguish enantiomers by NMR, you need a chiral shift reagent (like Eu(hfc)₃) or a chiral derivatizing agent (like Mosher's acid, MTPA), which converts enantiomers into diastereomers with distinguishable spectra.
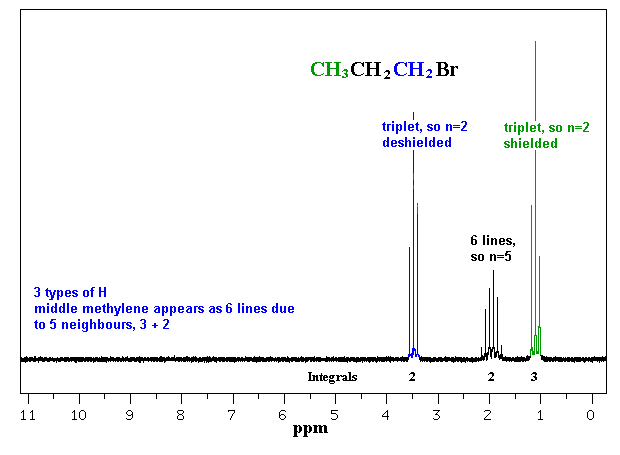
Confirming Reaction Products and Regiochemistry
Comparing the NMR spectrum of a product to that of the starting material is one of the most direct ways to confirm that a reaction worked and to determine its regiochemistry.
- Disappearance of a signal indicates that a functional group was consumed. For example, oxidation of a secondary alcohol to a ketone removes the signal and the signal.
- Appearance of a new signal confirms formation of a new functional group. An aldehyde signal near 9–10 appearing after an oxidation reaction confirms the product.
- Shifts in existing signals reflect changes in the electronic environment around hydrogens that weren't directly involved in the reaction but are nearby.
- Integration ratios in the product spectrum can confirm stoichiometry. If two regioisomers form (as in electrophilic aromatic substitution), their ratio can be measured directly from integration of distinct product signals.
For regiochemistry, the splitting pattern of the product is especially telling. In Markovnikov addition of HBr to propene, the product (2-bromopropane) shows a septet-doublet pattern, while the anti-Markovnikov product (1-bromopropane) shows a distinctly different triplet-based pattern.
NMR Spectroscopy Fundamentals
A few practical details underpin every 1H NMR experiment:
- Tetramethylsilane (TMS) is the universal reference compound, defined as ppm. Its 12 equivalent hydrogens produce a single sharp peak far upfield from most organic signals.
- Deuterated solvents (like , , or DMSO-) are used because regular solvents contain that would overwhelm the spectrum. The deuterium () nuclei don't resonate at the same frequency as .
- Shielding and deshielding are the core concepts behind chemical shift. Electrons around a hydrogen generate a small local magnetic field that opposes the external field, shielding the nucleus and shifting it upfield. Electron-withdrawing groups pull electron density away, deshielding the nucleus and shifting it downfield.
- The Larmor frequency is the specific radiofrequency at which a nucleus resonates in a given magnetic field. It's proportional to the field strength, which is why stronger magnets give better spectral resolution.