Structure and Properties of Carboxylic Acids
Carboxylic acids contain a carbonyl group (C=O) directly bonded to a hydroxyl group (-OH), forming the carboxyl group (-COOH). This combination gives them distinctive physical properties and makes them significantly more acidic than alcohols. Understanding why they behave this way comes down to hydrogen bonding patterns and resonance stabilization.
Cyclic Dimers of Carboxylic Acids
Most carboxylic acids don't exist as isolated molecules in the liquid or solid phase. Instead, two molecules pair up through intermolecular hydrogen bonding to form cyclic dimers.
Here's how it works: the O-H of one carboxyl group donates a hydrogen bond to the C=O oxygen of a second molecule, and that second molecule does the same thing back. The result is a ring-shaped structure held together by two hydrogen bonds.
- Formic acid () and acetic acid () both form these dimers readily
- Because you need to break two hydrogen bonds per pair of molecules, boiling points are higher than you'd predict from molecular weight alone
- For example, propanoic acid (, MW 74) has a boiling point of 141 °C, while 1-butanol (MW 74) boils at 118 °C, even though both can hydrogen bond
This dimer formation is why carboxylic acids can be tougher to separate by distillation than you might expect.

Acidity of Carboxylic Acids vs. Alcohols
Carboxylic acids are far stronger acids than alcohols, and the values make this clear:
- Typical carboxylic acid : 4–5 (e.g., acetic acid )
- Typical alcohol : 16–18 (e.g., ethanol )
That's a difference of roughly 11–12 units, which means carboxylic acids are about to times more acidic. The reason comes down to what happens after the proton leaves.
When a carboxylic acid loses a proton, it forms a carboxylate ion (), where the negative charge is delocalized across two oxygen atoms through resonance. This spread-out charge is much more stable than a concentrated one.
When an alcohol loses a proton, it forms an alkoxide ion (), where the negative charge sits entirely on a single oxygen. That localized charge makes the alkoxide less stable and more eager to grab a proton back.
There's also an inductive effect at play: the electronegative carbonyl oxygen withdraws electron density from the O-H bond, making the proton easier to lose.
Charge Delocalization in Carboxylate Ions
The stability of the carboxylate ion () is the single biggest reason carboxylic acids are strong enough to behave as acids in aqueous solution. Two equivalent resonance structures explain this stability:
- A C–O single bond to one oxygen (which carries the formal negative charge) and a C=O double bond to the other oxygen
- The reverse: C=O to the first oxygen and C–O to the second
The actual structure is a resonance hybrid of both. In practice, this means:
- The negative charge is evenly distributed between the two oxygens (each carries roughly half)
- Both C–O bonds have a bond order of 1.5, intermediate between a single and double bond
- You can see this experimentally: both C–O bond lengths in acetate ion () are identical at about 1.27 Å, between a typical C–O single bond (1.43 Å) and C=O double bond (1.23 Å)
This even distribution of charge is what makes the carboxylate ion so much more stable than an alkoxide, and it's the core concept behind carboxylic acid acidity.
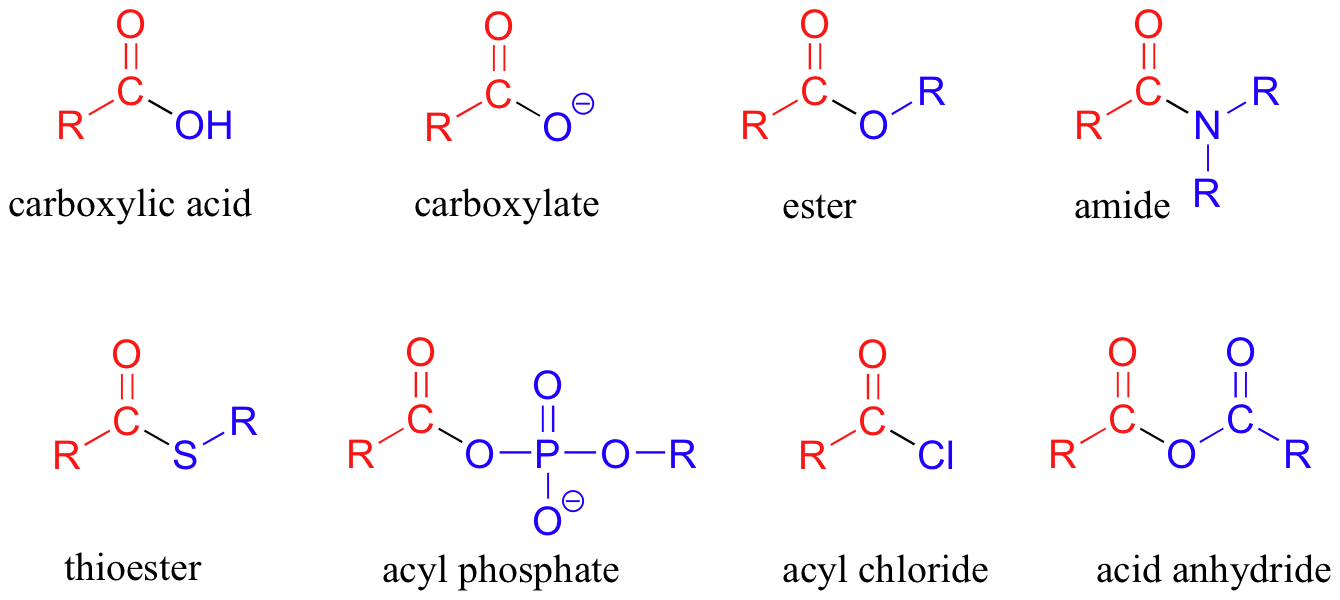
Structure and Reactivity of Carboxylic Acids
A few structural features of carboxylic acids are worth keeping in mind as you move into their reactions:
- The carbonyl carbon is electrophilic because the electronegative oxygen pulls electron density away from it. This makes it a target for nucleophilic attack, which is how carboxylic acid derivatives (esters, amides, anhydrides, acyl chlorides) are formed.
- The alpha hydrogen, on the carbon adjacent to the carbonyl, is more acidic than a typical C-H bond () due to stabilization of the resulting enolate. This matters for reactions you'll see later, like alpha-substitution.
- The carbonyl oxygen acts as a hydrogen bond acceptor, which contributes to the dimer formation discussed above and also influences solubility in water. Short-chain carboxylic acids (up to about four carbons) are fully miscible with water.