Solubility equilibrium and the common ion effect explain how dissolved substances behave when specific ions are already present in solution. These principles let you predict when adding a particular ion will decrease solubility and when a precipitate will form.
Precipitation reactions and selective precipitation build directly on these ideas. By comparing ion concentrations to solubility limits, you can predict and control the formation of solids from solutions, which is essential for separating and identifying ions in mixtures.
Solubility Equilibrium and the Common Ion Effect
Common ion effect on solubility
The common ion effect happens when a solution already contains one of the ions produced by a dissolving solute. That extra ion shifts the dissolution equilibrium to the left (back toward the solid), which decreases the solute's solubility.
For example, dissolves to produce and . If you dissolve in a solution that already contains from , the extra pushes the equilibrium toward solid , so less dissolves than it would in pure water.
This is a direct application of Le Chatelier's principle: adding a common ion is a stress on the equilibrium, and the system responds by shifting in the direction that consumes that ion.
One thing to keep straight: the solubility product constant () does not change when a common ion is present. depends only on temperature. What changes is the molar solubility of the solute, because the equilibrium concentrations adjust to keep satisfied.
Ion concentrations with common ions
To calculate ion concentrations when a common ion is present:
- Write the balanced dissociation equation for the solute:
- Set up an ICE table, using the common ion's concentration as the initial value for that ion.
- Substitute equilibrium expressions into the equation and solve.
Worked example: Find in a solution of 0.1 M with excess . ()
- fully dissociates, so M before any dissolves.
- Let = moles per liter of that dissolve. Then at equilibrium: and .
- Plug into the expression:
- Because is tiny, will be negligible compared to 0.1, so approximate :
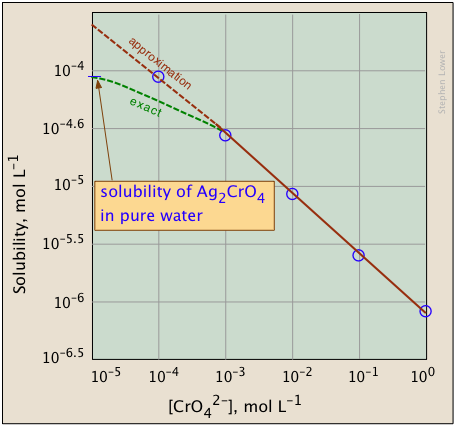
Compare this to the solubility of in pure water ( M). The common ion reduced solubility by roughly four orders of magnitude.
When can you use the approximation? The "drop the " shortcut works when the common ion concentration is much larger than the expected solubility. A good rule of thumb: if the common ion concentration is at least 100 times larger than would be, the approximation introduces negligible error. You can always verify by checking that your calculated is indeed small relative to the common ion concentration.
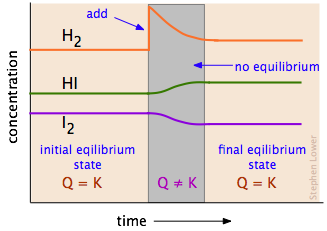
Precipitation Reactions and Selective Precipitation
Precipitation reactions using Q vs Ksp
To predict whether a precipitate forms when two solutions are mixed, compare the reaction quotient () to :
- : The solution is supersaturated. Precipitation occurs until drops to equal .
- : The solution is unsaturated. No precipitate forms; if solid is present, more can dissolve.
- : The system is at equilibrium. No net change.
To calculate , use the initial ion concentrations (after mixing but before any reaction). For a salt that dissociates into cations and anions:
Worked example: You mix 50.0 mL of M with 50.0 mL of M . Does precipitate? ()
- Account for dilution. The total volume doubles (50 mL + 50 mL = 100 mL), so each concentration is halved:
- Calculate :
- Compare: , so yes, precipitates.
A common mistake on exams: forgetting to account for dilution when two solutions are mixed. If you combine equal volumes, each ion concentration is halved. If volumes are unequal, use for each ion.
Selective precipitation in analysis
Selective precipitation uses differences in values to separate ions from a mixture. The ion that forms the least soluble compound with a given reagent precipitates first.
Here's how it works in practice:
- Choose a precipitating agent that forms insoluble compounds with some target ions but not others.
- Add the reagent gradually. The ion forming the compound with the smallest precipitates first.
- Filter to separate the solid precipitate from the remaining solution.
- Repeat with a different reagent to precipitate the next ion.
For example, suppose a solution contains both and . Adding dilute precipitates () while () stays mostly in solution because its is much larger. You can then filter off the and test the remaining solution for using a different reagent like .
To figure out how much reagent to add, you can calculate the minimum needed to start precipitating each ion. For : . For : (since ). The large gap between these threshold concentrations is what makes the separation possible.
This approach allows systematic identification of ions in complex mixtures, which is exactly what happens in qualitative analysis schemes you may encounter in lab.