Chemical equilibrium describes the steady state where the concentrations of reactants and products no longer change over time. Calculating the actual concentrations at equilibrium is one of the most practical skills in Gen Chem II, since it connects the abstract idea of an equilibrium constant to real, measurable quantities like pH.
Equilibrium Concentrations and Pressures
Calculations with the equilibrium constant
The equilibrium constant (K) expresses the ratio of product concentrations to reactant concentrations at equilibrium, each raised to the power of its stoichiometric coefficient.
For a general reaction , the equilibrium expression is:
where , , , and are molar concentrations and , , , are the stoichiometric coefficients.
There are two main types of problems you'll see with this expression:
Finding K from known equilibrium concentrations. Plug the values straight into the expression. For the reaction , if the equilibrium concentrations are , , , and :
Notice that is squared because its coefficient is 2. Forgetting to apply exponents is one of the most common mistakes on exams.
Finding an unknown concentration when K is known. Rearrange the equilibrium expression to isolate the unknown. Using the same reaction with , , , and :
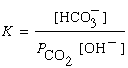
ICE table method
The ICE table is your go-to tool when you know the initial concentrations and the value of K, but not the equilibrium concentrations. ICE stands for:
- Initial (I): Concentrations before any reaction occurs.
- Change (C): How much each species increases or decreases as the system reaches equilibrium.
- Equilibrium (E): The final concentrations, found by adding the Initial and Change rows.
Here's how to work through an ICE problem step by step:
- Write the balanced equation and the K expression.
- Fill in the Initial row with the given starting concentrations. Products that haven't formed yet start at 0.
- Define the Change row using a variable x. The signs and coefficients come from stoichiometry. Reactants decrease (negative), products increase (positive), and each change is multiplied by that species' coefficient.
- Write the Equilibrium row as the sum of Initial + Change for each species.
- Substitute the Equilibrium expressions into the K expression and solve for x.
- Calculate all equilibrium concentrations using the value of x.
Example: For with , , , and no products initially:
| A | B | C | D | |
|---|---|---|---|---|
| Initial | 0.1 | 0.2 | 0 | 0 |
| Change | ||||
| Equilibrium |
Substituting into the K expression:
This equation can be solved using the quadratic formula (or sometimes by simplifying with approximations). If solving gives , then:
Quick check: Always verify that your x value doesn't make any equilibrium concentration negative. If it does, you've made an error somewhere.

Multiple equilibria systems
Some systems involve more than one equilibrium happening at the same time. The concentrations of shared species link the equilibria together, so you can't solve them independently.
To handle these problems:
- Write the equilibrium constant expression for each reaction.
- Set up an ICE table for each equilibrium, using separate variables (x, y, etc.) for each reaction's extent.
- Substitute the equilibrium expressions into each K expression.
- Solve the resulting system of equations simultaneously.
- Use the solved variables to find all equilibrium concentrations.
Example: Consider two coupled equilibria with and no B or C initially:
- with
- with
| Equilibrium 1 | A | B | |
|---|---|---|---|
| Initial | 0.1 | 0 | |
| Change | |||
| Equilibrium |
B produced in the first reaction then participates in the second:
| Equilibrium 2 | B | C | |
|---|---|---|---|
| Initial | 0 | ||
| Change | |||
| Equilibrium |
The two equations are:
From : , so , giving .
Substituting into : , which simplifies to . Solving: , so , giving .
Then: .
Note that the net equilibrium concentration of B is , since B is produced by the first reaction and consumed by the second. The final concentrations:
You can verify these by checking that ... which doesn't equal 2. This tells you the "sequential ICE table" approach used here is an approximation. For a more rigorous treatment, you'd set up a single ICE table where A loses total, B has a net amount of , and C gains , then solve both K expressions simultaneously from the start. Your instructor will clarify which approach they expect.
Equilibrium in acid-base reactions
Acid-base equilibria are just a specific application of everything above, using their own named constants.
- The acid dissociation constant describes how much a weak acid ionizes:
- The base dissociation constant describes how much a weak base reacts with water:
Note that water doesn't appear in the expression because it's the solvent (pure liquids are excluded from equilibrium expressions).
Connecting equilibrium to pH:
- At 25°C:
Example: Finding the pH of 0.1 M acetic acid (, )
| Initial | 0.1 | 0 | 0 |
| Change | |||
| Equilibrium |
Because is very small relative to the initial concentration, you can often use the small-x approximation: assume . This simplifies the math to:
Since :
When is the small-x approximation valid? A common rule of thumb: if is less than 5% of the initial concentration, the approximation holds. Here, , so you're safe. If the percentage exceeds 5%, solve the full quadratic instead.