The Nernst equation lets you calculate cell potential when concentrations aren't at standard conditions (1 M, 1 atm). Since real batteries and electrochemical systems almost never operate at standard conditions, this equation bridges the gap between textbook values and actual measured voltages.
Concentration cells take this idea further: they generate voltage purely from a difference in concentration between two half-cells containing the same redox couple. You'll see these principles at work in pH meters, biological membrane potentials, and ion-selective sensors.
Nernst Equation and Cell Potential
Nernst equation for nonstandard conditions
The Nernst equation adjusts the standard cell potential based on the actual concentrations (or pressures) of the species involved. The driving idea is that , the reaction quotient, tells you how far the system is from equilibrium, and that shift changes the voltage.
Each variable:
- = cell potential under actual (nonstandard) conditions
- = standard cell potential (looked up from tables, at 1 M / 1 atm)
- = ideal gas constant, 8.314 J/(mol·K)
- = temperature in Kelvin
- = number of moles of electrons transferred in the balanced redox equation
- = Faraday's constant, 96,485 C/mol
- = reaction quotient, calculated the same way as an equilibrium expression but using current concentrations, not equilibrium ones
At 25°C (298 K), the constants simplify to give a version you'll use most often:
Notice this version uses (base 10) instead of . The factor 0.0592 V comes from combining and converting to .
How to use it, step by step:
-
Write the balanced overall cell reaction and identify (the electrons transferred).
-
Look up or calculate it from standard reduction potentials: .
-
Write the expression for using the balanced equation. Remember: products over reactants, each raised to its stoichiometric coefficient. Pure solids and liquids don't appear in .
-
Plug concentrations (or pressures for gases) into .
-
Substitute into the Nernst equation and solve for .
Quick check on your answer: If (more reactants than products), the term is negative, so . If , the cell potential drops below standard. This makes intuitive sense: a system rich in reactants has more driving force to move forward.
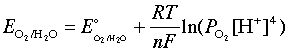
Cell potential and thermodynamic relationships
Cell potential, Gibbs free energy, and the equilibrium constant are three ways of describing the same thermodynamic reality. If you know one, you can find the others.
The key equations:
From these, you can also derive:
What the signs tell you:
| Condition | Reaction | ||
|---|---|---|---|
| Spontaneous (forward) | Positive | Negative | Proceeds toward products |
| At equilibrium | Zero | Zero | No net change |
| Nonspontaneous (forward) | Negative | Positive | Requires external energy |
A larger positive corresponds to a more negative and a larger . That means the reaction strongly favors products at equilibrium.
One thing that trips students up: at equilibrium, and . Plugging that into the Nernst equation is actually how you derive the relationship between and . It's not a separate fact to memorize; it falls right out of the equation.
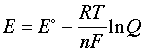
Concentration Cells
Concentration cells and applications
A concentration cell is an electrochemical cell where both half-cells use the same metal and the same ion, just at different concentrations. Because the electrodes are identical, . The entire voltage comes from the concentration difference.
How it works:
- The half-cell with higher ion concentration acts as the cathode (reduction occurs, pulling ions out of solution).
- The half-cell with lower ion concentration acts as the anode (oxidation occurs, pushing ions into solution).
- Electrons flow from the dilute side to the concentrated side through the external wire.
- The cell runs until both concentrations equalize, at which point .
Think of it this way: the system is trying to equalize the concentrations, and it generates electrical work in the process.
Practical applications:
- pH meters measure the voltage of a concentration cell where concentration differs between a reference solution and the sample. That voltage maps directly to pH.
- Solubility product determination: By setting up a concentration cell where one side contains a saturated solution of a sparingly soluble salt (like ), you can measure the cell potential and back-calculate the unknown ion concentration, which gives you .
- Biological systems: Nerve cells maintain different ion concentrations across their membranes. The resulting potential difference (similar to a concentration cell) drives nerve impulse transmission.
- Sensors: Glucose monitors and environmental ion sensors use concentration-dependent potentials to detect analyte levels.
Potential calculation of concentration cells
Since for a concentration cell, the Nernst equation simplifies to:
At 25°C:
where is the concentration of the metal ion at the cathode (the more concentrated side) and is the concentration at the anode (the more dilute side).
Example: A concentration cell has two half-cells. One contains 0.010 M , the other 1.0 M . Find the cell potential at 25°C.
-
The cathode is the concentrated side: .
-
The anode is the dilute side: .
-
For , .
-
Notice that the voltage is small (about 59 mV). Concentration cells typically produce modest voltages because the driving force is only a concentration gradient, not a difference in reduction potential between two different metals.
Key points to remember:
- The potential depends only on the concentration ratio, not on the identity of the redox couple.
- A larger ratio of cathode-to-anode concentration gives a larger voltage.
- You can work backwards: measure and one known concentration to find an unknown concentration.