Buffer Systems
Buffer systems keep the pH of a solution stable when small amounts of acid or base are added. They show up constantly in chemistry and biology, from controlling reaction conditions in the lab to maintaining blood pH in your body. This section covers how buffers work, how to calculate their pH, and what determines how well they perform.
Resistance of Buffers to pH Changes
A buffer is a solution that resists pH changes when small amounts of strong acid or strong base are added. Every buffer contains two key components: a weak acid and its conjugate base (or a weak base and its conjugate acid). These two components work as a team to neutralize whatever gets added.
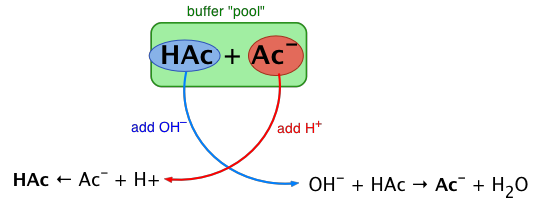
Here's how that works in practice, using an acetic acid/acetate buffer as an example:
When strong acid () is added:
- The conjugate base () reacts with the added to form the weak acid ()
- This consumes the added , so pH barely drops
When strong base () is added:
- The weak acid () reacts with the added to form the conjugate base () and water
- This consumes the added , so pH barely rises
The ratio of weak acid to conjugate base controls the buffer's pH:
- More weak acid relative to conjugate base → lower pH
- More conjugate base relative to weak acid → higher pH
Because both components are always present, the buffer can respond to additions in either direction.

Henderson-Hasselbalch Equation for pH
The Henderson-Hasselbalch equation gives you a direct way to calculate the pH of a buffer solution:
Where:
- = the negative log of the acid dissociation constant () of the weak acid
- = concentration of the conjugate base
- = concentration of the weak acid
Steps to calculate buffer pH:
- Identify the weak acid and its conjugate base in the buffer.
- Look up (or calculate) the of the weak acid.
- Determine the concentrations of the conjugate base () and weak acid ().
- Plug into the equation and solve.
Example: A buffer contains 0.1 M acetic acid () and 0.2 M sodium acetate.
Notice that when , the log term equals zero and . That's a useful shortcut to remember.
Buffer Capacity and Effective Range
Buffer capacity refers to how much strong acid or base a buffer can absorb before its pH changes significantly. Two factors control it:
- Concentration of the buffer components. A buffer made from 1.0 M acetic acid and 1.0 M acetate can neutralize far more added acid or base than one made from 0.01 M of each. Higher concentrations mean greater capacity.
- Ratio of weak acid to conjugate base. A ratio close to 1:1 gives the maximum buffer capacity because you have plenty of both components available to respond to either acid or base additions.
Effective pH range is the window of pH values where the buffer actually works well. For most buffers, this is roughly:
pH unit
Outside this range, you've used up too much of one component, and the buffer can no longer resist pH changes effectively. This is why choosing the right weak acid matters: its should be close to your target pH.
To design or evaluate a buffer, check three things:
- Is the of the weak acid close to the desired pH?
- Are the concentrations of both components high enough for the job?
- Is the acid-to-conjugate-base ratio reasonably close to 1:1?
Buffer Equilibrium and Stability
Buffers work because they maintain an equilibrium between the weak acid and its conjugate base. For an acetic acid buffer, that equilibrium is:
When you add strong acid or base, you're disturbing this equilibrium. Le Chatelier's principle explains the buffer's response: the system shifts to partially counteract the disturbance, consuming the added or and restoring near-original conditions.
The equilibrium constant determines where this balance settles. A buffer doesn't prevent pH change entirely; it just minimizes it by shifting the equilibrium. The common ion effect also plays a role here: the presence of the conjugate base suppresses dissociation of the weak acid, helping keep both components available in solution.