Electrolytes
Electrolytes are substances that produce ions when dissolved in water, which allows the solution to conduct electricity. Understanding how and why different substances ionize in water is central to predicting the behavior of solutions.
Concept and Examples of Electrolytes
When a substance dissolves in water and breaks apart into ions, those ions carry electrical charge through the solution. That's what makes something an electrolyte: it creates a conducting solution.
Electrolytes fall into categories based on how completely they ionize:
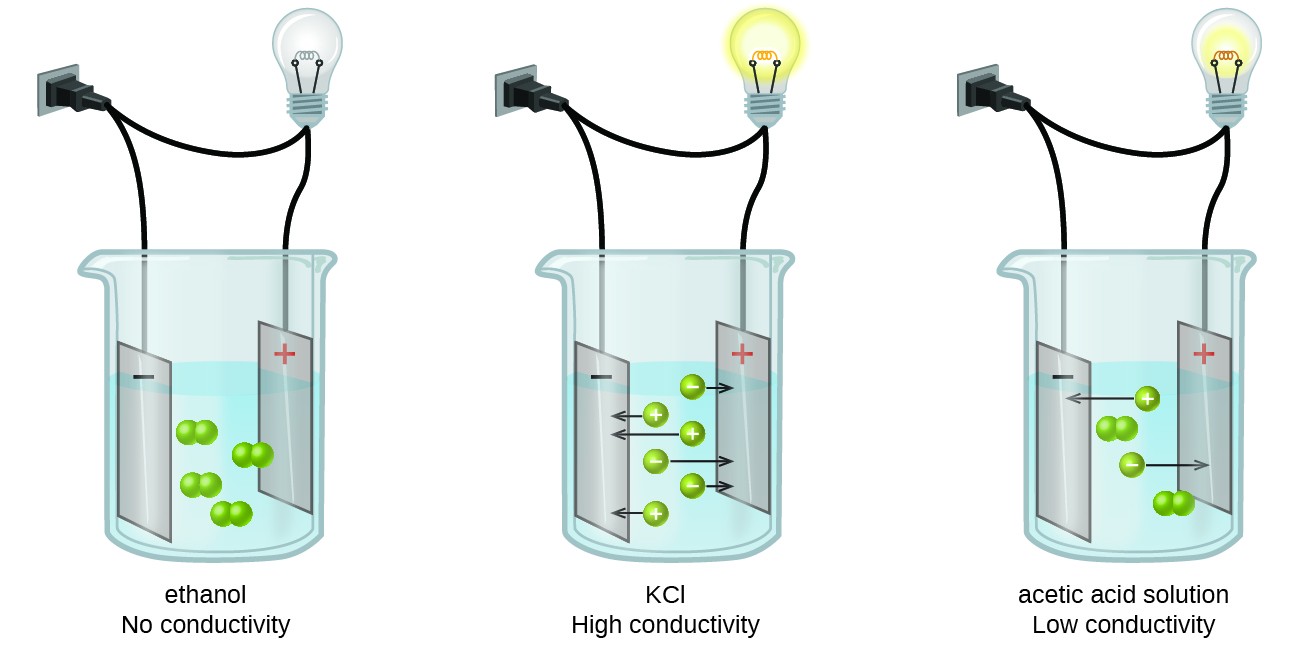
- Strong electrolytes completely dissociate into ions. Virtually no intact formula units remain in solution. Examples: , ,
- Weak electrolytes only partially dissociate, so the solution contains a mixture of ions and undissociated molecules. Examples: (acetic acid), (ammonia)
- Non-electrolytes don't produce ions at all, so their solutions don't conduct electricity. Examples: table sugar (), ethanol ()
A quick way to think about it: the more ions floating around in solution, the better the solution conducts and the stronger the electrolyte.
Dissolution of Ionic vs. Covalent Electrolytes
Ionic and covalent electrolytes produce ions through different mechanisms.
Ionic electrolytes (salts) are already made of ions in the solid state. When they dissolve, water molecules pull the cations and anions apart through ion-dipole attractions:
This is called dissociation: the compound separates into pre-existing ions.
Covalent electrolytes (acids and bases) are made of molecules, not ions. They must react with water to produce ions. This process is called ionization.
- Strong acids and bases react completely with water:
- Weak acids and bases react only partially, establishing an equilibrium between ions and undissociated molecules. The double arrow () signals that the reaction doesn't go to completion:
The key distinction: ionic compounds dissociate (split apart), while covalent compounds ionize (react to form new ions).
Solute-Solvent Interactions and Electrolyte Strength
How strongly a solute interacts with the solvent determines whether it behaves as a strong or weak electrolyte.
The main interaction at work is the ion-dipole interaction between ions and polar water molecules. Water's partial charges are attracted to dissolved ions, which helps pull solutes apart and stabilize the resulting ions in solution.
- Strong ion-dipole interactions lead to complete dissociation. Ions with high charge density (small radius and/or high charge, like or ) attract water molecules strongly, favoring full dissociation.
- Weaker ion-dipole interactions lead to partial dissociation. Larger ions with lower charge density don't attract water as effectively.
The solvent itself matters too:
- Polar solvents like water are good at solvating ions because their molecular dipoles can surround and stabilize charged particles. This promotes dissociation and ionization.
- Non-polar solvents like hexane can't effectively stabilize ions, so even strong electrolytes may not dissociate well in them.
Key Processes in Electrolyte Solutions
Four terms come up repeatedly when discussing electrolyte solutions. They're related but distinct:
- Dissociation: An ionic compound separates into its component ions in solution. No chemical reaction occurs; the ions were already there in the solid.
- Ionization: A molecular (covalent) compound reacts with the solvent to create ions that didn't exist before.
- Solvation: Solvent molecules surround and stabilize dissolved ions or molecules. This is the general term that applies to any solvent.
- Hydration: Solvation specifically by water. When you see after an ion, that ion is hydrated.
Dissociation and ionization describe how ions form. Solvation and hydration describe what happens to those ions once they're in solution: solvent molecules cluster around them, which keeps them dissolved and stable.