Absolute Zero and the Third Law
Absolute zero is the lowest temperature that can exist: 0 K (−273.15 °C). At this point, a system reaches its minimum possible energy and maximum order. The Third Law of Thermodynamics builds on this idea by stating that a perfect crystal at absolute zero has exactly zero entropy, giving us a fixed reference point for measuring the entropy of any substance.
Definition of Absolute Zero
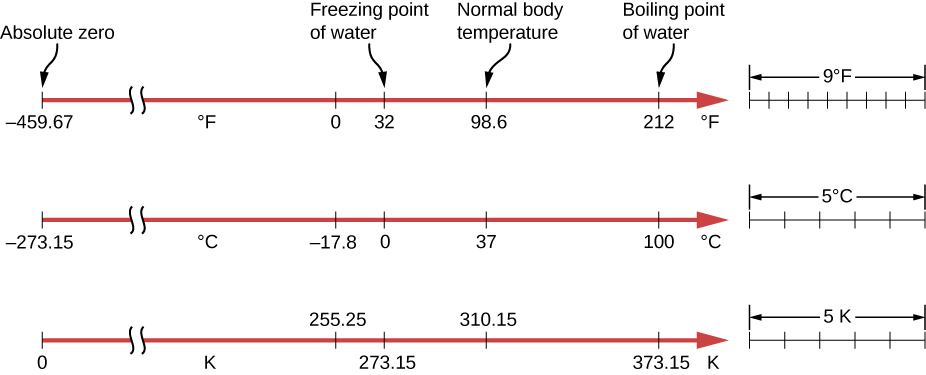
Absolute zero is defined as 0 K, which equals −273.15 °C. At this temperature, particles have the lowest kinetic energy allowed by quantum mechanics, and the system reaches maximum order.
- All classical molecular motion ceases at absolute zero. Particles stop translating, rotating, and vibrating in the classical sense (though quantum zero-point energy remains).
- The Kelvin scale uses absolute zero as its starting point, which is why thermodynamic equations rely on Kelvin rather than Celsius or Fahrenheit. Relative scales like Celsius can produce negative values that break equations such as the ideal gas law.
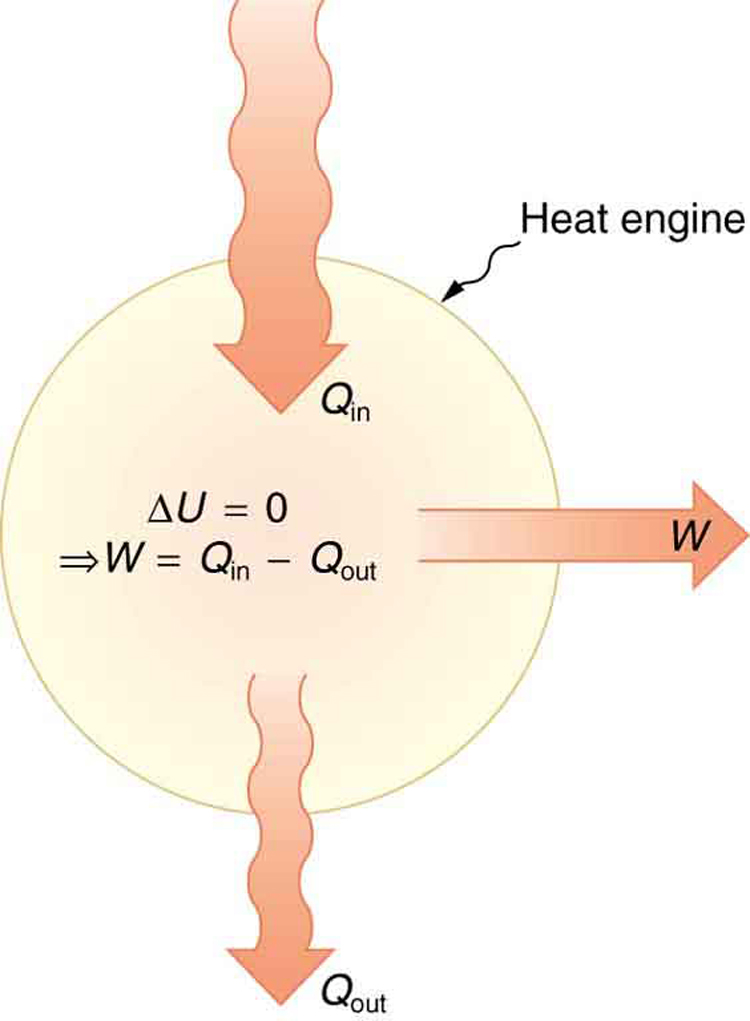
- Practical examples: heat engine efficiency calculations (like Carnot efficiency, ) and the ideal gas law () both require temperature in Kelvin to give correct results.
Third Law of Thermodynamics
The Third Law states that the entropy of a perfect crystal at absolute zero is exactly zero. Entropy measures the number of microscopic arrangements (microstates) available to a system. A perfect crystal at 0 K has only one possible arrangement, so its entropy is zero.
- As temperature approaches 0 K, the entropy of any system approaches a constant minimum value. For a perfect crystal, that minimum is zero.
- This gives us an absolute reference point for entropy. Unlike energy, where we often work with changes, the Third Law lets us assign actual entropy values to substances. Standard molar entropies listed in data tables are built on this foundation.
- Real substances that aren't perfect crystals (glasses, mixed crystals) can retain a small amount of residual entropy at 0 K because their atoms are frozen into a disordered arrangement. Ice is a classic example: the hydrogen bonds can orient in multiple ways even at very low temperatures.
Unattainability of Absolute Zero
The Third Law has an important consequence: it's impossible to reach exactly 0 K in a finite number of steps.
Here's why this happens, step by step:
- Each cooling step removes some entropy from the system.
- As the system gets colder, the remaining entropy shrinks, and each successive step removes a smaller fraction of what's left.
- The work required to extract heat grows larger and larger as temperature drops, increasing without bound as you approach 0 K.
- You'd need an infinite number of steps to actually reach absolute zero.
Advanced cooling techniques can get remarkably close:
- Adiabatic demagnetization uses magnetic fields applied to paramagnetic salts to reach temperatures in the microkelvin range.
- Laser cooling slows atoms with carefully tuned photons, reaching temperatures on the order of nanokelvins.
Even with these methods, zero-point energy sets a quantum mechanical floor. Every system retains some minimum energy even in its ground state, so complete removal of all energy is not physically possible.
System Behavior Near Absolute Zero
As temperature drops toward 0 K, materials undergo dramatic changes in their physical and thermodynamic properties.
Physical property changes:
- Superconductivity: Certain materials (lead below 7.2 K, niobium below 9.3 K) lose all electrical resistance, allowing current to flow indefinitely without energy loss.
- Superfluidity: Liquid helium-4 below about 2.17 K flows with zero viscosity, meaning it can climb container walls and pass through tiny pores without friction.
- Thermal expansion decreases and approaches zero, consistent with the Third Law's requirement that entropy changes vanish at 0 K.
Thermodynamic property changes:
- Specific heat capacity drops toward zero. The Debye model predicts that the heat capacity of solids follows a dependence at very low temperatures, meaning it takes vanishingly small amounts of energy to change the temperature.
- Thermal conductivity behavior depends on the material. In crystalline solids like diamond, thermal conductivity can peak at low temperatures before dropping, while amorphous materials like rubber behave quite differently.
- Gas behavior deviates strongly from the ideal gas law (). At very low temperatures, real gases condense into liquids or solids, and quantum statistics (Bose-Einstein or Fermi-Dirac) replace classical descriptions.
Quantum effects become dominant:
- Zero-point energy, the minimum energy a quantum system can have, becomes the most significant contribution to a system's total energy. For a quantum harmonic oscillator, this equals .
- Phenomena like quantum tunneling and Bose-Einstein condensation (where particles collectively occupy the lowest quantum state) emerge and govern material behavior in ways that classical physics cannot explain.