Thermodynamics is the science of energy and its transformations. It governs how heat, work, and energy interact in everything from power plants and engines to refrigerators and fuel cells. This section covers the core definitions you'll need as a foundation for the rest of the course.
Introduction to Thermodynamics
Concept of Thermodynamics
Thermodynamics studies the relationships between heat, work, and other forms of energy. At its core, it's about understanding how energy converts from one form to another: mechanical to thermal, chemical to electrical, and so on.
This field has direct engineering applications:
- Power generation: steam power plants, gas turbines, internal combustion engines
- Climate control: refrigeration and air conditioning systems
- Heat management: heat exchangers and heat transfer equipment
- Emerging technologies: fuel cells, renewable energy systems
- Transport engineering: aerospace and automotive design

Energy vs. Work vs. Heat
These four terms get used constantly in thermodynamics, and mixing them up causes real problems. Here's how they differ:
- Energy is the capacity to do work or cause change. It's measured in joules (J) or equivalent units. Energy is a property a system has.
- Work is energy transfer that happens when a force acts through a distance: , where is force and is displacement. Work is something a system does.
- Heat is energy transfer driven by a temperature difference. It always flows from a higher-temperature object to a lower-temperature one. Heat is measured in joules (J) or calories (cal).
- Temperature is a measure of the average kinetic energy of particles in a substance. It indicates how hot or cold something is and is measured in Kelvin (K), Celsius (°C), or Fahrenheit (°F).
The distinction between heat and temperature trips people up. Temperature is a state of a substance; heat is energy in transit because of a temperature difference. A large lake at 30°C holds far more thermal energy than a cup of water at 80°C, even though the cup is at a higher temperature.
Energy Forms and Conservation
Forms of Energy
Energy takes several forms, and thermodynamics often involves tracking how energy shifts between them.
Kinetic energy is associated with motion:
- Translational: , where is mass and is velocity
- Rotational: , where is moment of inertia and is angular velocity
Potential energy is associated with position or configuration:
- Gravitational: , where is mass, is gravitational acceleration ( near Earth's surface), and is height
- Elastic: , where is the spring constant and is displacement from equilibrium
Other important forms:
- Internal energy: the total kinetic and potential energy of all particles within a system (molecular motion, vibration, intermolecular forces)
- Chemical energy: energy stored in chemical bonds, released or absorbed during reactions
- Electrical energy: energy associated with the flow of electric charges
- Electromagnetic energy: energy carried by electromagnetic waves (light, radio waves, etc.)
Energy regularly converts between these forms. Friction converts mechanical energy (kinetic + potential) into thermal energy. Batteries convert chemical energy into electrical energy. Electric motors convert electrical energy into mechanical energy. Tracking these conversions is a central task in thermodynamics.
Energy Conservation in Systems
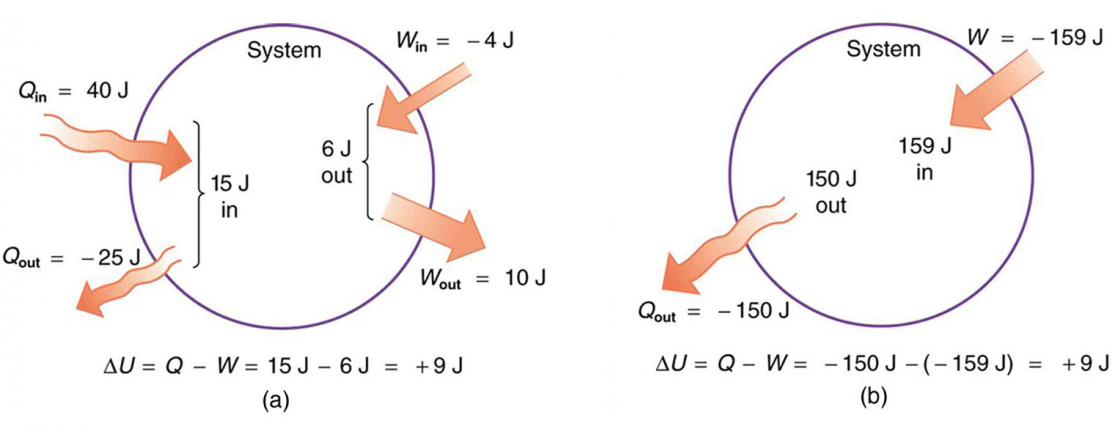
The First Law of Thermodynamics states that energy cannot be created or destroyed, only converted from one form to another. Mathematically:
where is the change in internal energy of the system, is the heat added to the system, and is the work done by the system.
To apply this law, you need to know what kind of system you're dealing with. Systems are classified by what can cross their boundaries:
- Closed system: no mass crosses the boundary, but energy (heat and work) can. The change in internal energy depends only on the initial and final states, not the path taken between them.
- Open system: both mass and energy can cross the boundary. The energy balance must account for heat, work, and the energy carried by mass flowing in and out.
- Isolated system: neither mass nor energy crosses the boundary. Total energy inside stays constant.
One more useful category: a steady-state system is one whose properties don't change over time. In steady state, the rate of energy entering equals the rate of energy leaving. Many real engineering devices (turbines running at constant speed, heat exchangers at operating conditions) are analyzed as steady-state open systems.