Maxwell Relations and Their Applications
Maxwell relations let you calculate thermodynamic quantities that are difficult or impossible to measure directly by converting them into expressions involving measurable properties like temperature, pressure, and volume. They show up constantly in thermodynamic derivations, so understanding where they come from and how to use them is worth the effort.
Derivation of Maxwell Relations
The four Maxwell relations all come from one mathematical fact: for any well-behaved function of two variables, the order of mixed second partial derivatives doesn't matter. For a function :
You apply this property to each of the four thermodynamic potentials, starting from their differential forms:
| Potential | Differential Form | Natural Variables |
|---|---|---|
| Internal energy | ||
| Enthalpy | ||
| Helmholtz free energy | ||
| Gibbs free energy |
Each differential has the form , where and are the "conjugate" coefficients. Applying the equality of mixed partials () to each potential gives one Maxwell relation:
-
From :
-
From :
-
From :
-
From :
To derive any one of these yourself, identify the two natural variables and the two conjugate coefficients in the differential form, then set the cross-derivatives equal. The negative signs appear wherever or contributes a minus sign to one of the coefficients.
Physical Interpretation of Maxwell Relations
Each relation connects a partial derivative that's hard to visualize with one that has a clearer physical meaning.
Relation 1:
This applies to adiabatic (constant-entropy) processes. It tells you how temperature responds to a volume change during a reversible adiabatic expansion or compression, relating it to how pressure changes with entropy at fixed volume.
Relation 2:
Also for isentropic (reversible adiabatic) processes. The temperature change you get from changing pressure adiabatically equals the rate at which volume changes with entropy at constant pressure.
Relation 3:
This is one of the two most practically useful relations. At constant temperature, the entropy gained per unit volume increase equals the rate of pressure increase with temperature at constant volume. The right-hand side is directly measurable from an equation of state.
Relation 4:
The other workhorse relation. The right-hand side is just , where is the thermal expansion coefficient (). This means you can calculate how entropy changes with pressure isothermally using volume and thermal expansion data alone.
Relations 3 and 4 are the ones you'll use most often, because they connect entropy (which you can't measure directly) to -- data (which you can).
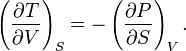
Applications in Thermodynamic Calculations
Expressing entropy changes using measurable quantities
Suppose you need the entropy change of a substance during an isothermal pressure change. Entropy isn't something you read off a gauge, but Maxwell relation 4 converts it:
Integrating from pressure to at constant :
If you know the equation of state (or have tabulated data), you can evaluate this integral directly.

Deriving relationships between heat capacities and equations of state
Maxwell relations also let you connect heat capacities to -- behavior. For example, starting from relation 3 and differentiating with respect to :
This tells you how depends on volume, expressed entirely in terms of the equation of state. For an ideal gas, , so , confirming that of an ideal gas is independent of volume.
Simplification of Thermodynamic Equations
The Clapeyron equation
The Clapeyron equation describes the slope of a phase boundary on a - diagram:
where and are the entropy and volume changes of the phase transition. Since is hard to measure directly, you substitute (valid at equilibrium, where ):
Now everything on the right is measurable: the latent heat , the transition temperature , and the volume change .
General strategy for applying Maxwell relations
When you encounter a partial derivative that involves entropy or another quantity you can't measure directly:
- Identify which thermodynamic potential has the relevant natural variables.
- Write the corresponding Maxwell relation.
- Replace the unmeasurable derivative with the equivalent measurable one.
- Integrate if needed, using equation-of-state data or known material properties (, , , etc.).
This strategy turns abstract thermodynamic expressions into quantities you can actually compute from experimental data.