Phase diagrams are essential tools for understanding alloy systems. They show how temperature and composition affect the equilibrium phases in materials, which lets you predict microstructures and properties. The Gibbs phase rule connects components, phases, and degrees of freedom, giving you a framework for interpreting what's happening on any phase diagram.
Phase Diagrams and Alloy Systems
Interpretation of phase diagrams
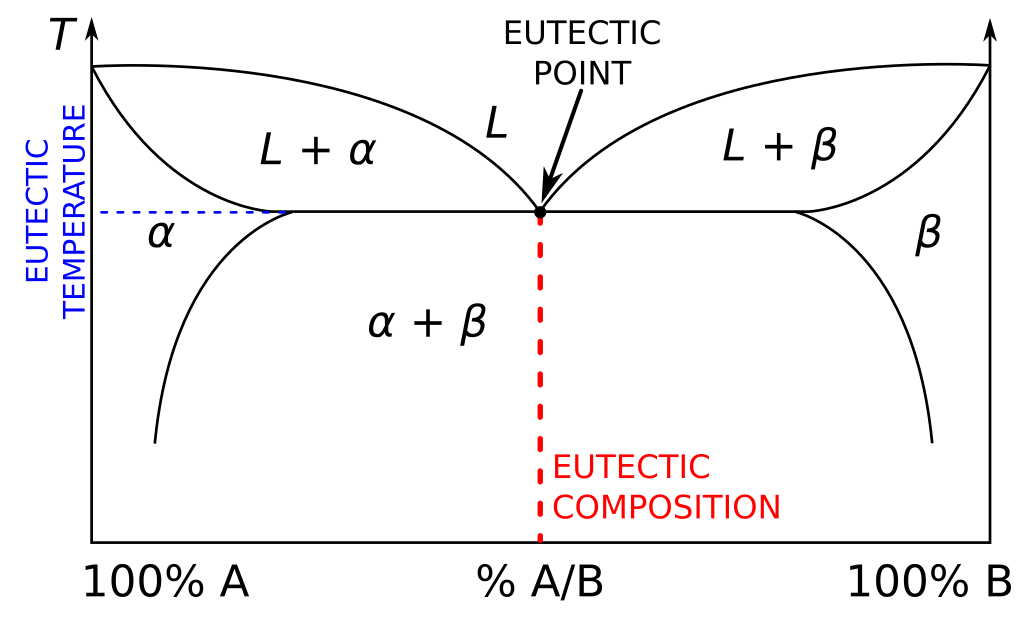
Binary phase diagrams graphically represent equilibrium phases in two-component systems (e.g., Cu-Ni). Composition sits on the x-axis and temperature on the y-axis. Distinct regions separated by phase boundaries denote different phases.
- Tie lines are horizontal lines drawn across two-phase regions at a given temperature. They connect the compositions of the two phases in equilibrium (e.g., liquid + solid). You read the endpoints of a tie line to find the composition of each phase, and you use the lever rule to find the relative amounts of each phase.
- Microstructure evolution depends on which equilibrium phases exist at a specific composition and temperature.
- Slow cooling gives the system time to reach equilibrium phases.
- Rapid cooling can produce non-equilibrium phases (like martensite in steel) or supersaturated solid solutions, because atoms don't have time to diffuse and rearrange.
Ternary phase diagrams depict equilibrium in three-component systems (e.g., Fe-Cr-Ni). Composition is plotted on an equilateral triangle, and you typically view either isothermal sections (a slice at constant temperature) or vertical sections along a specific composition path.
Application of Gibbs phase rule
The Gibbs phase rule is:
where is the number of degrees of freedom, is the number of components, and is the number of phases in equilibrium. Degrees of freedom are the independent variables (temperature, pressure, composition) you can change without altering the number of equilibrium phases present.
For binary systems (), the maximum degrees of freedom is 2:
- Single-phase regions: . You can independently vary both temperature and composition while staying in that single phase.
- Two-phase regions (phase boundaries): . Once you fix temperature, the compositions of both phases are determined.
- Invariant points (e.g., eutectic, peritectic): . Temperature, compositions, and phases are all fixed. Nothing can change without a phase disappearing.
For ternary systems (), the maximum degrees of freedom is 3, following the same logic: single-phase regions have , two-phase regions have , and invariant points have .
Thermodynamics of intermetallics and solutions
Intermetallic compounds are ordered phases with specific stoichiometry (e.g., NiAl) and a defined crystal structure. They form when the Gibbs free energy of the compound is lower than that of the constituent elements separately.
- Enthalpy of formation : strong bonding between unlike atoms releases energy, making the compound favorable.
- Entropy of formation : the ordered atomic arrangement reduces disorder compared to a random mixture.
- Overall stability is governed by . Because both and are negative, the compound is most stable at lower temperatures where the favorable enthalpy term dominates. At high temperatures, the term (which is positive here) can destabilize the compound.
Solid solutions are single-phase solids where solute atoms dissolve into a solvent matrix (e.g., Cu dissolved in Ni). They form when the Gibbs free energy of mixing is negative: .
- depends on relative bond strengths. If unlike-atom bonds are stronger than like-atom bonds, and mixing is enthalpically favorable.
- is always positive because mixing increases disorder. This means higher temperatures always promote solid solution formation through the term.
- Solid solubility is also governed by the Hume-Rothery rules: atomic size difference (< ~15%), similar electronegativity, similar crystal structure, and comparable valence electron concentration all favor extensive solid solubility.
Factors in alloy phase transformations
Three main variables control which phases form and how they transform:
- Composition determines the equilibrium phases, their relative amounts, solubility limits, and the temperatures and compositions of invariant reactions (e.g., the eutectic point).
- Temperature influences which phases are stable, how much solute a solid solution can hold (higher temperature generally increases solubility), and the kinetics of transformation (diffusion is faster at higher temperatures).
- Pressure has a smaller effect than composition or temperature for most alloy systems, but it can shift phase boundaries. Phases with higher density are favored at higher pressures, following Le Chatelier's principle.
Phase transformations directly alter material properties:
- Mechanical properties (strength, ductility) depend on which phases are present, their volume fractions, and microstructural features like grain size. A fine-grained two-phase microstructure will behave very differently from a coarse single-phase one.
- Physical properties (density, thermal/electrical conductivity) are determined by the crystal structure and bonding character of the phases present.
- Corrosion resistance is affected by phase composition and distribution. If two phases with different electrochemical potentials are in contact, galvanic coupling can accelerate corrosion at the more anodic phase.