Chemical equilibrium describes the point where a reaction's forward and reverse processes occur at equal rates, producing no net change in composition. Understanding this concept is central to predicting whether a reaction will proceed, how far it'll go, and how it responds to external changes. This section covers equilibrium from the Gibbs energy perspective, reaction coordinate diagrams, equilibrium calculations, and Le Chatelier's principle.
Chemical Equilibrium and Reaction Coordinates
Concept of chemical equilibrium
Chemical equilibrium is a dynamic state: both the forward and reverse reactions keep running, but they proceed at equal rates. Because of this, the concentrations of reactants and products remain constant over time, even though molecules are still reacting in both directions.
From a thermodynamic standpoint, equilibrium corresponds to the minimum in Gibbs free energy for the system at constant temperature and pressure. At that minimum, the Gibbs free energy change for the reaction is zero (). The system has no thermodynamic driving force to shift in either direction.
The standard Gibbs free energy change connects to the equilibrium constant through:
- is the standard Gibbs free energy change (all species in their standard states)
- is the universal gas constant (8.314 J/mol·K)
- is the absolute temperature in kelvins
- is the equilibrium constant
A large positive means is small (reactants favored), while a large negative means is large (products favored). For example, if at 298 K, you get , indicating the reaction strongly favors products at equilibrium.
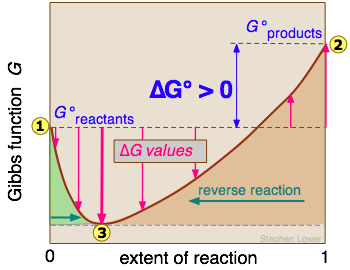
Interpretation of reaction coordinate diagrams
Reaction coordinate diagrams plot the Gibbs free energy of the system on the vertical axis against the extent of reaction (reaction coordinate) on the horizontal axis. Reactants sit on the left side and products on the right.
The key features to read from these diagrams:
- Transition state: The highest point on the curve between reactants and products. This peak represents the activation energy barrier the system must overcome for the reaction to proceed. A higher peak means a slower reaction (kinetics), but it says nothing about whether the reaction is thermodynamically favorable.
- Relative energy of reactants vs. products: If products sit lower in Gibbs energy than reactants, and the forward reaction is spontaneous. If products sit higher, and the reverse direction is spontaneous.
- Magnitude of the energy difference: A larger gap between reactants and products means a greater thermodynamic driving force and a larger equilibrium constant, which translates to a higher equilibrium yield of products (or reactants, depending on direction).
One common mistake: labeling a reaction with products lower than reactants as "exothermic." On a Gibbs energy diagram, lower products mean the reaction is spontaneous (), not necessarily exothermic. Exothermic vs. endothermic refers to enthalpy (), not Gibbs energy. A reaction can be spontaneous yet endothermic if the entropy increase is large enough.

Calculations with the law of mass action
The law of mass action defines the equilibrium constant expression. For a general reaction:
The equilibrium constant is:
The square brackets represent equilibrium concentrations in mol/L (M), and the exponents are the stoichiometric coefficients from the balanced equation. Pure solids and pure liquids don't appear in the expression because their activities are defined as 1.
To calculate equilibrium concentrations from initial conditions, use an ICE table (Initial, Change, Equilibrium):
- Write the balanced equation and the equilibrium constant expression.
- List the initial concentrations of all species.
- Define the change in concentration using a variable (typically ). Use stoichiometric ratios to express each species' change in terms of .
- Write equilibrium concentrations as (initial + change) for each species.
- Substitute these expressions into the expression and solve for . This often produces a quadratic equation; use the quadratic formula or simplifying approximations when is very small or very large.
- Check that your answer gives physically meaningful (non-negative) concentrations.
Effects on equilibrium state
Le Chatelier's principle states that when a system at equilibrium is disturbed, it shifts to partially counteract the disturbance and establish a new equilibrium.
Temperature changes affect the value of itself:
- For an endothermic reaction (), increasing temperature shifts equilibrium toward products. Think of heat as a "reactant" that you're adding.
- For an exothermic reaction (), increasing temperature shifts equilibrium toward reactants. Here, heat acts like a "product," and adding more of it pushes the reaction backward.
Pressure changes matter only when gaseous species are involved:
- Increasing pressure (or decreasing volume) shifts equilibrium toward the side with fewer total moles of gas. For example, in (4 moles of gas on the left, 2 on the right), higher pressure favors ammonia production.
- Decreasing pressure shifts equilibrium toward the side with more moles of gas.
- Adding an inert gas at constant volume does not change the partial pressures of the reacting species, so it has no effect on equilibrium.
Concentration changes do not alter , but they do shift the position of equilibrium:
- Adding more reactant drives the reaction forward (toward products).
- Adding more product drives the reaction backward (toward reactants).
- Removing a product (for instance, by condensation or precipitation) also drives the reaction forward, which is a common industrial strategy for increasing yield.
The reaction quotient is useful here: it has the same form as but uses current (non-equilibrium) concentrations. If , the reaction shifts forward. If , it shifts in reverse. At equilibrium, .