Heat Engines and Components
Heat engines convert thermal energy into mechanical work by exploiting the temperature difference between a hot source and a cold sink. They show up everywhere: car engines, power plants, jet turbines. Understanding how they work is central to the Second Law of Thermodynamics, because the Second Law is what tells us why no engine can perfectly convert heat into work.
Thermal efficiency quantifies how well an engine performs this conversion. It compares the useful work you get out to the heat you put in. The Second Law places a hard ceiling on this efficiency, and this section covers how that ceiling arises.
Definition and Operation
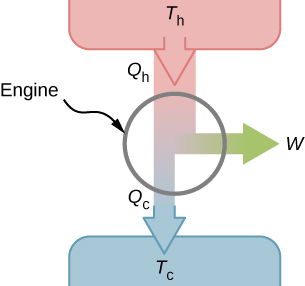
A heat engine is a thermodynamic system that converts thermal energy from a high-temperature source into mechanical work. It does this by cycling a working substance through a series of thermodynamic processes (compression, heating, expansion, cooling), transferring heat from the hot reservoir to the cold reservoir while producing net work output along the way.
The key word here is cyclically. The working substance returns to its initial state at the end of each cycle, so the engine can run continuously.
Primary Components
Every heat engine has four essential parts:
- Hot reservoir (heat source): Supplies thermal energy at high temperature (e.g., a combustion chamber, a boiler)
- Cold reservoir (heat sink): Absorbs rejected heat at low temperature (e.g., the atmosphere, a cooling tower)
- Working substance: The fluid that carries energy through the cycle (steam, air, combustion gases)
- Work-producing mechanism: Converts the working substance's energy into mechanical output (a piston, a turbine)
Examples
- Internal combustion engines (gasoline, diesel)
- External combustion engines (steam engines, Stirling engines)
- Gas turbines (jet engines, power generation turbines)
Thermal Efficiency of Heat Engines
Definition and Formula
Thermal efficiency () measures how much of the heat input actually becomes useful work. It's defined as the ratio of net work output to heat input from the hot reservoir:
This is a dimensionless quantity, often expressed as a percentage.

Calculation Using Heat Input and Output
You can derive a more practical form of the efficiency equation in two steps:
- Apply the First Law to the engine cycle. Since the working substance returns to its initial state each cycle (no change in internal energy), the net work equals the difference between heat absorbed and heat rejected:
- Substitute this into the efficiency definition:
This second form is especially useful because it shows efficiency in terms of the two heat transfers, and . If you could somehow reject zero heat (), you'd get 100% efficiency. But as the next point explains, that never happens.
Limitations on Efficiency
Thermal efficiency is always less than 1 (less than 100%). The Second Law of Thermodynamics requires that some heat must be rejected to the cold reservoir for the engine to complete its cycle. There is no way around this: a device that converts heat entirely into work with no other effect violates the Kelvin-Planck statement of the Second Law.
Factors Influencing Efficiency
Temperature Difference and Working Substance
The temperature difference between the hot and cold reservoirs is the primary driver of thermal efficiency. A larger gap means more of the input heat can be converted to work. This is why power plants operate their boilers at the highest practical temperature and reject heat at the lowest available temperature.
The working substance also matters. Properties like specific heat capacity and compressibility affect how much heat the fluid can absorb and how much work it can deliver during expansion. Different fluids are better suited to different temperature ranges and engine designs.
Irreversibilities and Design
Real engines always fall short of their theoretical maximum efficiency because of irreversibilities:
- Friction between moving parts converts some work into waste heat
- Heat losses through engine walls to the surroundings
- Fluid leakage past seals and valves
- Incomplete combustion of fuel, leaving chemical energy unconverted
Engine design choices directly target these losses. Compression ratio, valve timing, turbine blade geometry, and insulation all influence how close the real engine gets to its ideal cycle efficiency.
Operating Conditions
Efficiency isn't fixed for a given engine. It shifts with operating conditions:
- Speed and load: Engines are typically optimized for a specific operating range; running outside that range increases relative losses
- Ambient temperature: A hotter environment raises the cold reservoir temperature, shrinking the temperature difference and reducing efficiency
- Heat transfer rates: At very high speeds, there may not be enough time for complete heat transfer, reducing the effective
Types of Heat Engines: Comparison
Internal Combustion Engines (ICEs)
Internal combustion engines burn fuel directly inside the engine cylinder. The combustion gases themselves are the working substance, and their expansion drives a piston. ICEs are compact and responsive, which is why they dominate transportation, but they tend to have lower thermal efficiencies (typically 25–35% for gasoline, 35–45% for diesel).
- Gasoline engines use a spark to ignite a premixed fuel-air charge. They operate on the Otto cycle: isentropic compression, isochoric (constant-volume) heat addition, isentropic expansion, isochoric heat rejection.
- Diesel engines compress air to a high enough temperature that injected fuel auto-ignites. They operate on the Diesel cycle: isentropic compression, isobaric (constant-pressure) heat addition, isentropic expansion, isochoric heat rejection. The higher compression ratios give diesel engines better efficiency than gasoline engines.
External Combustion Engines (ECEs)
External combustion engines burn fuel outside the engine to heat a separate working fluid. This separation allows them to use a wider variety of fuels and heat sources, and they can potentially achieve higher efficiencies, though they tend to be larger and slower to respond than ICEs.
- Steam engines heat water in a boiler to produce high-pressure steam, which drives a piston or turbine. They operate on the Rankine cycle: isentropic compression (pump), isobaric heat addition (boiler), isentropic expansion (turbine), isobaric heat rejection (condenser). Most large-scale power plants use this cycle.
- Stirling engines use a sealed gas (often air or helium) that is alternately heated and cooled to drive a piston. They operate on the Stirling cycle: isothermal compression, isochoric heat addition, isothermal expansion, isochoric heat rejection. In theory, the Stirling cycle can match Carnot efficiency, but real Stirling engines face practical limitations.
Gas Turbines
Gas turbines compress incoming air, mix it with fuel, and ignite the mixture. The resulting hot gases expand through a turbine to produce work. They offer high power-to-weight ratios, which makes them ideal for aircraft propulsion and peaking power plants.
Gas turbines operate on the Brayton cycle: isentropic compression (compressor), isobaric heat addition (combustion chamber), isentropic expansion (turbine), isobaric heat rejection (exhaust). In power generation, gas turbines are often paired with a steam cycle in a combined cycle plant, using the hot exhaust gases to drive a Rankine cycle and pushing overall efficiency above 60%.