Definition of black body
A black body is an idealized object that perfectly absorbs and emits electromagnetic radiation. It doesn't exist in nature, but it's the theoretical benchmark for understanding how thermal radiation works, and studying it is what forced physicists to invent quantum mechanics.
Ideal absorber and emitter
A black body absorbs all incident electromagnetic radiation, regardless of wavelength or angle. It also emits radiation at every wavelength with the maximum possible intensity for its temperature. The key property: the emission spectrum depends only on temperature, not on what the object is made of or how its surface is structured.
No real object is a perfect black body, but some systems come close (more on cavity radiation below).
Perfect thermal equilibrium
A black body in thermal equilibrium maintains a constant temperature because it absorbs and emits energy at equal rates. This is a steady-state condition where energy input exactly balances energy output.
Each microscopic absorption process is balanced by a corresponding emission process. This is called the principle of detailed balance. The black body in equilibrium serves as the reference point against which non-equilibrium systems are compared.
Electromagnetic spectrum
Wavelength vs frequency
Wavelength and frequency are inversely related through:
where is the speed of light, is wavelength (in meters), and is frequency (in Hertz). The energy of a single photon is directly proportional to its frequency:
where J·s is Planck's constant. This means shorter-wavelength radiation (X-rays, gamma rays) carries more energy per photon than longer-wavelength radiation (radio waves, microwaves).
Visible light range
Visible light occupies a narrow band of the spectrum, roughly 380–740 nm, corresponding to frequencies between Hz and Hz. Human eye sensitivity peaks around 555 nm (green light). A black body at about 5800 K (the Sun's surface temperature) has its peak emission right in this visible range, which is not a coincidence from an evolutionary standpoint.
Historical context
Classical physics limitations
Before Planck, physicists tried to explain black body radiation using classical thermodynamics and electromagnetism. The Rayleigh-Jeans law worked well at low frequencies but made a catastrophic prediction at high frequencies: it said the spectral energy density should increase without bound as frequency increases.
The root of the problem was the classical equipartition theorem, which assigns an average energy of to each electromagnetic mode in a cavity. Since the number of modes grows as , the total energy diverges. This clearly contradicted experiments, which showed intensity dropping off at high frequencies.
Ultraviolet catastrophe
Paul Ehrenfest coined the term "ultraviolet catastrophe" for this failure. Classical theory predicted infinite energy emission at short wavelengths (ultraviolet and beyond), while experiments showed a clear peak in the spectrum followed by a rapid decrease at high frequencies. Resolving this contradiction required a completely new idea: energy quantization, introduced by Max Planck in 1900.
Planck's law
Planck's law is the central result of this topic. It gives the correct spectral energy distribution for black body radiation at all frequencies, and its derivation introduced quantization into physics.
Derivation and assumptions
Planck's key move was to assume that the oscillators in the cavity walls can only have energies that are integer multiples of a fundamental quantum:
where and is the oscillator frequency. He then used the Boltzmann distribution to compute the average energy of an oscillator at temperature . Instead of the classical result , he obtained:
This average energy goes to at low frequencies (recovering the classical result) but drops exponentially at high frequencies (suppressing the ultraviolet catastrophe). Multiplying by the density of electromagnetic modes gives the Planck spectral radiance:
Energy quantization concept
The radical idea here is that energy exchange between radiation and matter happens in discrete packets (quanta), each of size . At high frequencies, each quantum is large, so thermal fluctuations at temperature are unlikely to excite those modes. This is why the spectrum falls off at high frequencies rather than diverging. Quantization also provided the theoretical basis for understanding atomic spectra and the photoelectric effect.
Stefan-Boltzmann law
Total radiated power
Integrating Planck's law over all frequencies gives the total power radiated per unit area by a black body:
where W/(m²·K⁴) is the Stefan-Boltzmann constant and is the surface area. For real objects, you multiply by the emissivity (a number between 0 and 1):
Temperature dependence
The fourth-power dependence is steep. Doubling the temperature increases radiated power by a factor of . This explains why very hot objects cool rapidly through radiation, and it's the basis for estimating stellar luminosities. If you know a star's radius and surface temperature, you can calculate its total luminosity directly.
Wien's displacement law
Peak wavelength calculation
Wien's law tells you where the black body spectrum peaks:
where m·K is Wien's displacement constant. Higher temperature means shorter peak wavelength.
Temperature vs wavelength relation
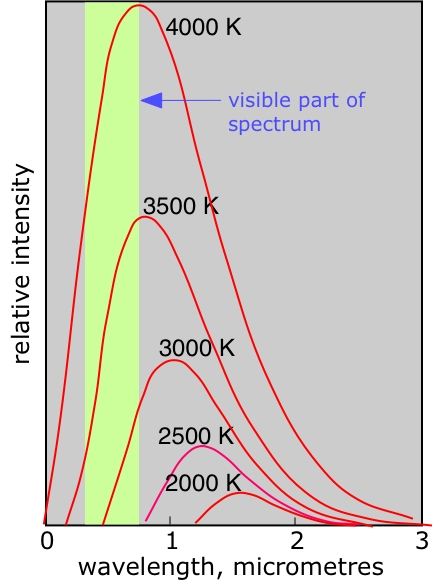
This inverse relationship explains why heated objects change color as they get hotter: a warm iron bar glows red (longer wavelength), and as temperature increases it shifts toward orange, yellow, and eventually white (a mix of many visible wavelengths). The Sun's peak emission near 500 nm corresponds to its surface temperature of about 5800 K. In astrophysics and remote sensing, measuring the peak wavelength of an object's spectrum is a direct way to determine its temperature.
Applications in astrophysics
Stellar temperature estimation
Stars are approximate black bodies, so their spectra roughly follow Planck's law. By measuring the peak wavelength or fitting the spectral shape, you can estimate a star's effective temperature (the temperature of a perfect black body with the same total luminosity). The Hertzsprung-Russell diagram organizes stars by effective temperature and luminosity, revealing distinct populations like main-sequence stars, giants, and white dwarfs.
Cosmic microwave background
The cosmic microwave background (CMB) is relic radiation from about 380,000 years after the Big Bang. It has the most perfect black body spectrum ever measured, peaking at a wavelength corresponding to K. Tiny anisotropies (variations at the level of 1 part in 100,000) in the CMB encode information about the density, composition, and geometry of the early universe.
Cavity radiation
Blackbody cavity properties
A small hole in an enclosed cavity is the classic physical realization of a black body. Any radiation entering the hole bounces around inside and is almost entirely absorbed before it can escape. The radiation inside the cavity reaches thermal equilibrium with the walls, and the spectrum of light escaping through the hole depends only on the cavity's temperature, not on what the walls are made of.
Laboratory devices called hohlraums are designed to approximate this ideal as closely as possible.
Kirchhoff's law
Kirchhoff's law of thermal radiation states that for a body in thermal equilibrium, the emissivity and absorptivity are equal at each wavelength and direction:
This means good absorbers are necessarily good emitters, and vice versa. The law is essential for understanding radiative heat transfer and for connecting the idealized black body to real materials.
Experimental measurements
Techniques and apparatus
- Cavity radiators (hohlraums) approximate ideal black body conditions for calibration and testing.
- Spectrophotometers measure spectral intensity as a function of wavelength or frequency.
- Bolometers measure total incident radiation power by detecting temperature changes in an absorbing element.
- Cryogenic techniques allow study of low-temperature black body radiation (relevant for CMB measurements).
Challenges in precision
Achieving uniform temperature throughout the cavity is difficult, and background radiation can contaminate measurements. Real materials have emissivity that varies with wavelength, temperature, and surface condition, so corrections are always needed. Detector calibration across a wide range of wavelengths and intensities remains a persistent experimental challenge.
Quantum mechanical interpretation
Energy states in cavity
In the full quantum treatment, each electromagnetic mode in the cavity is a quantum harmonic oscillator with discrete energy levels:
The term is the zero-point energy, present even when no photons occupy the mode. This contributes to vacuum fluctuations but does not affect the thermal radiation spectrum (it cancels out when computing energy differences).
The density of electromagnetic modes increases as , but the Bose-Einstein occupation factor decreases exponentially at high frequencies. The competition between these two effects produces the characteristic peaked shape of the Planck spectrum.
Photon statistics
Photons are bosons, so they obey Bose-Einstein statistics. The mean number of photons in a mode of frequency at temperature is:
This is the Planck distribution (also called the Bose-Einstein distribution for zero chemical potential, since photon number is not conserved). At low frequencies (), many photons occupy each mode. At high frequencies (), occupation drops exponentially. This statistics also underlies the theory of stimulated and spontaneous emission.
Thermodynamic considerations
Entropy and radiation
The photon gas inside a black body cavity has well-defined thermodynamic quantities. The entropy of black body radiation is:
where is the total internal energy. Since , the entropy scales as (at fixed volume). This result comes directly from the statistical mechanics of the photon gas and governs the irreversibility of radiative processes.
Free energy of radiation
The Helmholtz free energy of the photon gas is:
Since , the free energy is negative, which is consistent with the fact that photons are spontaneously emitted into the cavity to reach equilibrium. The radiation pressure, energy density, and free energy together determine the equation of state for the photon gas: , where is the energy density.
Deviations from ideal behavior
Gray bodies
A gray body has a constant emissivity that doesn't vary with wavelength. Its emission spectrum has the same shape as a black body spectrum but is scaled down uniformly. This is a useful approximation for many engineering materials, though in reality most materials have emissivity that varies with wavelength.
Emissivity factors
Emissivity () quantifies how closely a real surface approximates a black body at a given wavelength. It depends on:
- Material composition
- Surface roughness and condition
- Temperature
- Wavelength
- Viewing angle (non-Lambertian surfaces have directionally dependent emissivity)
Accurate knowledge of emissivity is critical for non-contact temperature measurements (like pyrometry) and for heat transfer calculations in engineering.
Radiation pressure
Momentum transfer
Each photon carries momentum:
When photons are absorbed or reflected by a surface, they transfer momentum, exerting a force. For isotropic radiation in thermal equilibrium, the radiation pressure is:
where is the energy density. At the center of a massive star, radiation pressure can become comparable to or even exceed gas pressure, and it plays a key role in determining the Eddington luminosity limit (the maximum luminosity a star can have before radiation pressure blows away its outer layers).
Solar sail concept
Solar sails exploit radiation pressure from sunlight for propulsion. A large, lightweight reflective surface captures momentum from solar photons, providing continuous, fuel-free acceleration. The force is small but constant, making solar sails viable for long-duration missions. Reflective sails get roughly twice the momentum transfer compared to absorbing sails, since reflected photons reverse their momentum.
Modern applications
Thermal imaging
Thermal cameras detect infrared radiation emitted by objects and convert it into temperature maps. Every object above absolute zero emits thermal radiation, and the intensity and spectral distribution follow (approximately) Planck's law modified by the object's emissivity. Applications include medical diagnostics, building energy audits, electrical fault detection, and night vision.
Incandescent vs LED lighting
An incandescent bulb heats a filament to about 2700–3000 K, producing a broad black body spectrum. Most of the emitted energy falls in the infrared, making these bulbs only about 5% efficient at producing visible light. LEDs, by contrast, emit in narrow wavelength bands through electroluminescence, converting electrical energy to visible light far more efficiently. Understanding black body radiation makes clear why incandescent bulbs are inherently wasteful: the physics of thermal radiation guarantees that most energy goes to wavelengths you can't see.