Radioactive Decay and Half-Life
Radioactive decay is the process by which unstable atomic nuclei lose energy by emitting radiation. Each radioactive isotope decays at its own characteristic rate, measured by its half-life. Because this rate is predictable, scientists can use it to determine the age of ancient materials through a technique called radiometric dating.

Concept of Radioactive Half-Life
The half-life is the time required for half of a radioactive substance to decay into other products. Two properties make half-life especially useful:
- It remains constant for each specific isotope. Potassium-40, for example, has a half-life of 1.3 billion years, while carbon-14's is only 5,730 years.
- It's independent of external factors like temperature, pressure, or chemical environment. Nothing you do to a sample changes how fast it decays.
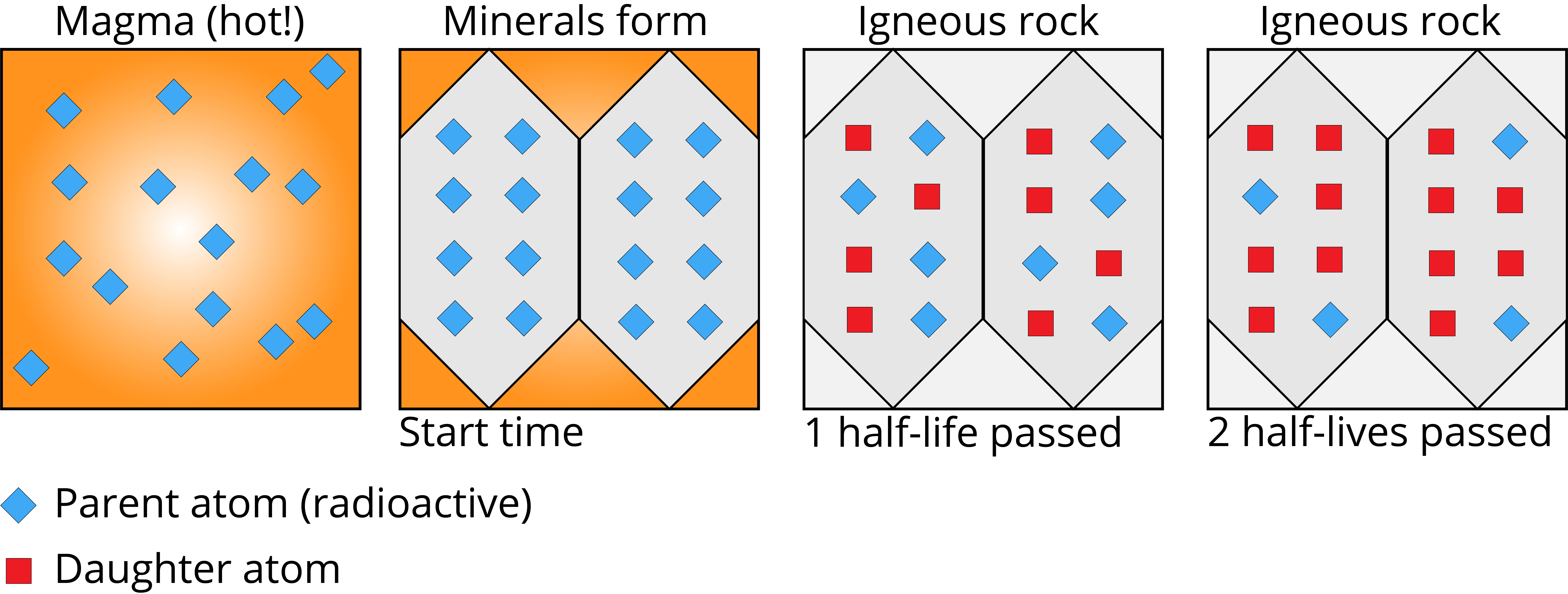
Radiometric dating takes advantage of these predictable decay rates to determine the age of rocks, fossils, and other materials. The basic idea: measure the ratio of the remaining parent isotope (the original radioactive atom) to the daughter isotope (what it decays into). Since you know the half-life, you can calculate how much time has passed since the sample formed.
Several parent-daughter pairs are used depending on the age range of the sample:
- Uranium-lead dating works for rocks billions of years old
- Rubidium-strontium dating covers a similar range and is useful for igneous and metamorphic rocks
- Carbon-14 dating is used for organic materials up to about 50,000–60,000 years old
Calculations for Decay Rates
The quantity of a radioactive isotope remaining after some time is given by the exponential decay equation:
where:
- = quantity of the isotope remaining at time
- = initial quantity of the isotope
- = the decay constant, related to the half-life by
- = elapsed time
You can also think about decay in terms of half-lives directly. After half-lives, the remaining fraction of the original sample is:
where . This form is often faster for problems where the elapsed time is a whole number of half-lives.
Solving a typical half-life problem:
- Identify the initial quantity and either the remaining quantity or the elapsed time .
- Look up (or use the given) half-life of the isotope.
- If you need the remaining quantity, plug into either decay equation above.
- If you need the elapsed time, rearrange the equation. From the exponential form: .
Example: A sample originally contains 80 g of a radioactive isotope with a half-life of 1,000 years. After 3,000 years (that's 3 half-lives), the remaining amount is .

Decay Chains and Equilibrium
Not every radioactive isotope decays straight to a stable product. Some go through a decay chain, a series of successive decays where each step produces a new radioactive isotope until a stable nucleus is finally reached. Uranium-238, for instance, passes through 14 decay steps before ending as stable lead-206.
Radioactive equilibrium occurs within a decay chain when a daughter isotope is produced at the same rate it decays. At that point, the amount of the daughter isotope stays roughly constant even though decay is still happening.
The stable atoms at the end of a decay chain are called radiogenic isotopes. These are the daughter products that accumulate over time and are measured in radiometric dating.
Carbon-14 Dating
Carbon-14 Dating Process and Limitations
Carbon-14 () is a radioactive isotope of carbon continuously produced in the upper atmosphere when cosmic rays strike nitrogen atoms. From there, it enters the carbon cycle: plants absorb during photosynthesis, and animals take it in by eating plants. In living organisms, the ratio of to stable stays roughly constant at about 1 part per trillion, because the organism is constantly exchanging carbon with the environment.
When an organism dies, that exchange stops. The in its remains decays with a half-life of 5,730 years, while the stable stays put. By measuring how much remains relative to , you can calculate how long ago the organism died. This technique works on organic materials like wood, charcoal, bone, and shell.
Limitations to keep in mind:
- Age range: Carbon-14 dating is only effective up to about 50,000–60,000 years. Beyond that, too little remains to measure reliably. This means it cannot be used to date dinosaur fossils (those are tens of millions of years old).
- Contamination: If older or younger carbon gets introduced into a sample, the measured age will be off. Isotopic fractionation (where different organisms or processes absorb at slightly different rates) can also skew results.
- Atmospheric variation: The method assumes the atmospheric ratio of to has been constant over time. In reality, it fluctuates due to changes in solar activity, volcanic eruptions, fossil fuel burning (which adds old, -depleted carbon to the atmosphere), and nuclear weapons testing (which temporarily spiked atmospheric in the mid-20th century). Scientists correct for these variations using calibration curves built from tree rings and other independent records.