Antibody Structure and Function
Antibodies are Y-shaped proteins produced by B cells that serve as the adaptive immune system's primary tool for identifying and neutralizing specific threats. Their structure is elegantly matched to their function: one end binds a unique antigen, while the other end communicates with the rest of the immune system to coordinate a response. Understanding how this molecule is built helps explain how the immune system achieves both specificity and versatility.
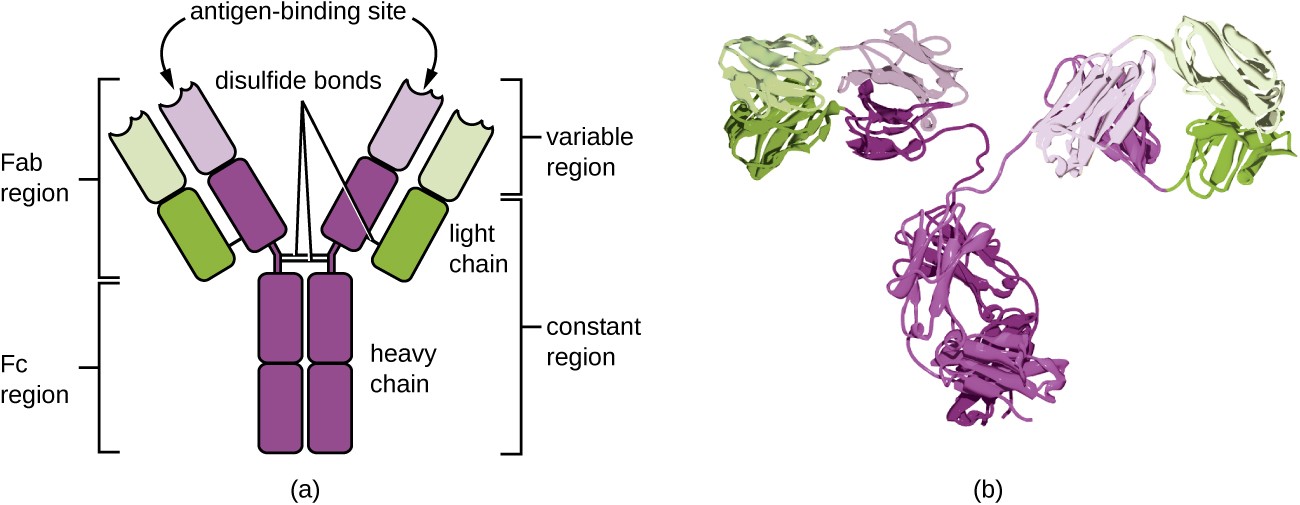
Components of Antibody Structure
Each antibody (immunoglobulin, or Ig) is built from four polypeptide chains: two identical heavy chains and two identical light chains, arranged into the characteristic Y shape.
The molecule divides into two functionally distinct regions:
- Fab (fragment, antigen-binding) region — the two "arms" of the Y. Each Fab contains the variable domains of one heavy chain () and one light chain (). Within these variable domains sit the hypervariable regions, also called complementarity-determining regions (CDRs). The CDRs are the actual contact points that grip the antigen, and their sequence varies enormously between different antibodies. That variation is what gives each antibody its specificity.
- Fc (fragment, crystallizable) region — the "stem" of the Y. It's made up of the constant domains of the two heavy chains. The Fc region doesn't touch the antigen at all. Instead, it interacts with Fc receptors on immune cells and with complement proteins, determining what happens after the antibody binds its target.
Several structural features hold the molecule together and fine-tune its behavior:
- Disulfide bonds link the two heavy chains to each other and each heavy chain to its light chain (interchain bonds), while additional intrachain disulfide bonds stabilize the folding of individual domains.
- The hinge region, located between the Fab arms and the Fc stem, gives the arms flexibility. This lets a single antibody bind two epitopes that may be spaced at different distances on a pathogen surface.
- N-linked glycosylation on the Fc region (particularly at Asn297 in IgG) modulates effector functions. Changes to these sugar groups can significantly alter how well the antibody activates complement or binds Fc receptors.
Functions of Antibody Isotypes
The heavy chain constant region determines the antibody's isotype (also called class). Each isotype has a distinct heavy chain type and is specialized for particular immune tasks.
IgG is the most abundant isotype in serum (~75% of circulating antibodies). It neutralizes toxins and viruses, promotes opsonization, and mediates antibody-dependent cell-mediated cytotoxicity (ADCC). IgG is the only isotype that crosses the placenta, providing passive immunity to the fetus. It has four subclasses (IgG1–IgG4) with slightly different effector profiles.
IgM is the first antibody produced during a primary immune response. It circulates as a pentamer (five monomers joined by a J chain), giving it ten antigen-binding sites. This high valency makes IgM extremely efficient at agglutinating pathogens and activating the classical complement pathway, even though individual binding sites tend to have relatively low affinity.
IgA is the dominant antibody at mucosal surfaces (respiratory tract, gut, urogenital tract) and is also found in breast milk, saliva, and tears. Secretory IgA exists as a dimer joined by a J chain and wrapped in a secretory component that protects it from enzymatic degradation. It neutralizes pathogens before they can breach epithelial barriers.
IgE is present at very low serum concentrations but plays an outsized role in allergic reactions and defense against parasitic worms (helminths). IgE binds with extremely high affinity to FcεRI receptors on mast cells and basophils. When antigen cross-links bound IgE, these cells degranulate and release histamine and other inflammatory mediators.
IgD is co-expressed with IgM on the surface of naive, mature B cells, where it functions as part of the B cell receptor (BCR) complex. Its role in B cell activation and maturation is still being clarified, and it's found at very low levels in serum.

Antibody-Mediated Effector Mechanisms
Once an antibody binds its antigen through the Fab region, the Fc region triggers downstream immune responses. The major mechanisms are:
- Opsonization — Antibodies coat a pathogen's surface, and phagocytes (macrophages, neutrophils) recognize the exposed Fc regions through their Fc receptors. This dramatically enhances phagocytic uptake compared to uncoated targets.
- Classical complement activation — The complement protein C1q binds to the Fc regions of antibodies clustered on a pathogen surface (IgM and IgG are most effective). This initiates the classical pathway cascade, ultimately forming the membrane attack complex (MAC) that lyses the target cell.
- Neutralization — Antibodies bind directly to toxins or to viral surface proteins required for host cell entry, physically blocking the pathogen from interacting with its receptor. No Fc-mediated signaling is needed here; the Fab does all the work.
- ADCC (antibody-dependent cell-mediated cytotoxicity) — NK cells recognize antibody-coated target cells via their FcγRIII (CD16) receptor. Binding triggers the NK cell to release perforin and granzymes, killing the target.
- ADCP (antibody-dependent cellular phagocytosis) — Similar to opsonization, but specifically refers to phagocytes engulfing antibody-coated cells (such as tumor cells or infected cells) after Fc receptor engagement.
- Mast cell and basophil degranulation — Cross-linking of IgE molecules already bound to FcεRI on mast cells or basophils causes rapid release of histamine, leukotrienes, and other inflammatory mediators. This is the basis of type I hypersensitivity (allergic) reactions.
Antibody Affinity and Avidity in Immune Responses
Affinity refers to the binding strength at a single antigen-binding site for a single epitope. It depends on how well the shapes and charges of the binding site and epitope complement each other, mediated by non-covalent interactions (hydrogen bonds, van der Waals forces, electrostatic interactions, and hydrophobic effects).
Avidity is the overall strength of the antibody-antigen interaction when multiple binding sites engage multiple epitopes simultaneously. A pentameric IgM molecule, for example, may have relatively low affinity at each individual site, but its avidity is high because all ten binding sites can engage at once. Avidity depends on both the antibody's valency (number of binding sites) and the epitope density on the target.
The kinetics of binding are described by three parameters:
- (association rate) — how quickly the antibody and antigen come together
- (dissociation rate) — how quickly they fall apart
- (equilibrium dissociation constant) — a lower means higher affinity
Affinity maturation is the process by which antibody affinity improves over the course of an immune response. During germinal center reactions, B cells undergo somatic hypermutation, introducing random point mutations into the variable region genes. B cells whose mutations happen to improve antigen binding are preferentially selected to survive and proliferate. This is why secondary immune responses produce antibodies with much higher affinity than primary responses.
High-affinity antibodies are also central to modern medicine. Monoclonal antibodies engineered for high affinity and specificity are used in diagnostics (e.g., pregnancy tests, ELISAs) and therapeutics targeting cancer (e.g., trastuzumab for HER2-positive breast cancer) and autoimmune disorders (e.g., adalimumab for rheumatoid arthritis).